Acute intermittent porphyria (AIP) is a rare inherited disorder of heme metabolism caused by a heterozygous, loss-of-function mutation affecting the enzyme porphobilinogen deaminase (PBGD). The remaining enzymatic activity is sufficient to avoid accumulation of intermediate metabolites until hepatic heme production is further activated, i.e. by a precipitant.
Clinically, it is characterised by severe abdominal pain, neuropsychiatric manifestations and hyponatremia from SIADH. Typically, attacks occur after exposure to a precipitating factor. Clinical diagnosis is often difficult. Despite its autosomal dominant pattern of inheritance, there is only 10-20% penetrance and therefore often a poor family history [1]. A large scale case-control study identified a multitude of possible precipitating factors [2], including many antibiotics, anaesthetic agents and alcohol. A comprehensive list compiled by the European Porphyria Network is freely available [3]. Furthermore, reference texts often list further causes such as calorie restriction and weight loss [4,5], although we are unaware of any clinical evidence to support this.
We present the diagnosis of a patient with AIP, precipitated by severe iron deficiency that led to appetite suppression and weight loss.
A 35-year-old Asian woman was seen in A&E with her first presentation of intense, colicky abdominal pain, worsening over 4 days. Her pain was severe and difficult to localize. It did not radiate to the groin, flank or back, and there were no alleviating or exacerbating factors. She was vomiting and her bowels had not opened for a week. She had a reduced appetite and had lost over 8 kg in body weight over 6 months, corresponding to about 20% of her body mass (BMI = 17.8 on admission).
She was otherwise a fit and healthy mother of two, with no past medical, surgical, gynaecological or psychiatric history. She did not have any allergies, and did not take any regular medications, nor any recreational substances. She did not drink or smoke, and worked as a community carer. Her last menstrual period was four weeks prior to her presentation. Her periods were normally regular, and she did not experience heavy or irregular bleeding prior to her admission. She had not noticed any symptoms of anaemia.
On examination, she was writhing on the bed in agony. She was emaciated, pale, and clinically dehydrated. Her abdomen was soft, diffusely tender, and distended. Bowel sounds were sluggish. Cardiovascular, respiratory, neurological, skin and joint examinations were unremarkable. A urine dipstick was entirely negative, including for β-HCG.
Laboratory investigations on admission showed a new microcytic anaemia and significant iron deficiency. Inflammatory markers were not elevated. She was also mildly hyponatremic and moderately hypophosphataemic (Table 1).
Table 1: Laboratory investigations. View Table 1
CT abdomen and pelvis showed bright enhancement around her stools, the appearance of which suggested that she had ingested oral radiological contrast (Figure 1). Otherwise, no acute pathology was found. The patient had not been given oral contrast, and denied ingestion of any medications, bismuth antacids, or solutions of heavy metals. On closer questioning, the patient admitted to ingesting chalk. This likely represents pica resulting from severe iron deficiency, and explains the CT findings and the constipation.
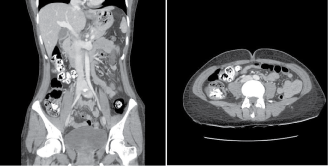 Figure 1: CT abdomen and pelvis showed bright enhancement around her stools, the appearance of which suggested that she had ingested oral radiological contrast.
View Figure 1
Figure 1: CT abdomen and pelvis showed bright enhancement around her stools, the appearance of which suggested that she had ingested oral radiological contrast.
View Figure 1
Her serum sodium continued to drop to a trough level of 106 mmol/L. Serum osmolality was low (233 mosm/L, reference range: 275-295 mosm/L) and a paired urine osmolality was inappropriately raised (491 mosm/L). Urinary sodium was also high (75 mmol/L, reference range: < 20 mmol/L). The hyponatraemia did not respond to fluid restriction, nor intravenous normal saline.
A random urine sample, protected from light, was analysed within 24 hours for urine porphobilinogen (PBG), which was strongly positive (94.3 μmol/L). Faecal samples tested negative for coproporphyrins. She was provisionally diagnosed with acute intermittent porphyria. Haem arginate infusion was bridged with cautious 10% glucose infusions. The patient was given intravenous iron. Symptoms and laboratory values, including electrolytes, quickly improved.
We present a unifying diagnosis of severe iron deficiency leading to pica, appetite suppression, weight loss, leading to an acute presentation of AIP. There were no other disturbances to the patient’s function, medical baseline or lifestyle, and no other discernible precipitants could be identified.
This patient was severely iron deficient on admission, the cause of which remains unknown. Indeed, roughly one-third of South Asian women living in the UK are reported to be iron deficient, compared with 8% of the general population [6]. Iron deficiency is well known to suppress appetite and lead to weight loss. Severe iron deficiency has been show to cause elevated levels of zinc and cobalt metalloporphyrins, which are linked to appetite suppression [7]. Furthermore, subjective appetite scores appear to correlate with serum ferritin [8]. Iron deficiency alone is not thought to be a trigger for AIP [1].
CT appearances were explained by the ingestion of calcium as chalk. This is likely to represent pica, which is associated with iron deficiency. Calcium salts are safe in AIP so her chalk ingestion is highly unlikely to have been the trigger.
Some classical features of AIP are seen in this case. Severe pain is characteristic. SIADH and hyponatraemia are also commonly seen. Features such as constipation, abdominal distension, vomiting, and hypophosphataemia can be attributed to the ileus that may accompany AIP [1]. Constipation may also result from ingestion of large quantities of chalk.
After administration of haem arginate and glucose, the patient recovered well. She underwent iron replacement therapy and her haemoglobin, weight and appetite subsequently recovered. She went on to have an OGD and colonoscopy as an outpatient to investigate the weight loss and anaemia. Both tests were unremarkable. A Coeliac screen was also negative. She was followed up by a specialist porphyria centre (National Acute Porphyria Service, Cardiff & Vale UHB) where her diagnosis was confirmed through further investigation. She also sought genetic screening for herself and her family.