Here we present the case of a 17-year-old male patient affected by autosomal-dominant Hyper IgE Syndrome (AD-HIES) who underwent to incision and drainage of recurrent anterior anal abscess. To our knowledge, this is the first reported case about recurrent perianal infection in autosomal-dominant Hyper IgE Syndrome.
Anal abscess, Hyper IgE syndrome
Hyper IgE Syndromes (HIES) are a rare primary immunodeficiencies characterized by elevated serum IgE, with an estimated incidence of 1 in 500000 to 1000000 individuals. The HIES are classified in two forms. Autosomal-dominant Hyper IgE Syndrome (AD-HIES) is clinically characterized by abnormalities in different systems, including the immune system, connective tissue, skeletal and vascular among others. Autosomal-recessive Hyper IgE Syndrome (AR-HIES) manifests as disorders of the immune system, recurrent skin and lung infections, sensitivity to viral infections (such as Molluscum Contagiosum, Human Papillomavirus, Herpes Simplex and Varicella Zoster virus), increase in neurological abnormalities and central nervous system involvement, but does not have musculoskeletal alterations [1-3].
Here we describe the case of a 17-year-old male patient affected by AD-HIES who complained of recurrent anterior anal abscess. To our knowledge, this is the first reported case about recurrent anal abscess in autosomal-dominant Hyper IgE Syndrome.
In March 2019, a 17-year-old male patient arrived at the Emergency Division of Paediatric of our Hospital with fever and cough since five days and severe pain in the anterior perianal area since the previous day.
The patient is affected by a sporadic form of autosomal-dominant Hyper IgE Syndrome, diagnosed when he was 10-year old. Genetic analysis demonstrated STAT3 mutation (1909G→A, V637M), not present in his family. The patient has characteristic facial appearance: prominent forehead, broad nasal ridge, sunken eyes and fleshy nose. Others typical features of our patient are kyphosis, persistence of deciduous teeth and dental crowding in childhood.
Over the past few years he reported many hospitalizations for pneumonia and underwent to multiple incisions and drainages of cutaneous abscesses (armpit, knee, gluteus).
In the last three years, he reported three episodes of perianal abscess. Two cases (May 2016 and August 2017) required ambulatory treatment (incision and drainage under local anaesthesia), one case (September 2018) needed hospitalization, incision and drainage under epidural anaesthesia.
At hospitalization, the thoracic clinical examination revealed diffuse harsh noise with respiratory failure. The abdomen was treatable in all quadrants. In the anterior perianal area (at 12 o'clock in gynaecological position), the clinical examination revealed an area of tenderness without evidence of fluctuating collection. The rectal exploration was very painful without detectable fluctuating collection in perianal area. No residual retracting scar or anal dysfunction were present. Laboratory analyses demonstrated increased inflammatory markers (neutrophilic leucocytosis, C-reactive protein, erythrocyte sedimentation rate) and increase in serum IgE levels (more than 5000 U/mL). Chest X-rays revealed two pneumonia, sited at the left pericardial area and superior lobe of the right lung (Figure 1). Computed tomography of the pelvis showed a multiseptated abscess of the anterior perineum (Figure 2). The patient received a proctologist valuation and underwent to a transanal ultrasonography which confirmed the presence of a deep anterior anal abscess outside the external anal sphincter (Figure 3).
 Figure 1: Chest X-rays: Double pneumonia (left pericardial area and superior lobe of the right lung). View Figure 1
Figure 1: Chest X-rays: Double pneumonia (left pericardial area and superior lobe of the right lung). View Figure 1
 Figure 2: Computed tomography of the pelvis: Multiseptated abscess of the anterior perineum. View Figure 2
Figure 2: Computed tomography of the pelvis: Multiseptated abscess of the anterior perineum. View Figure 2
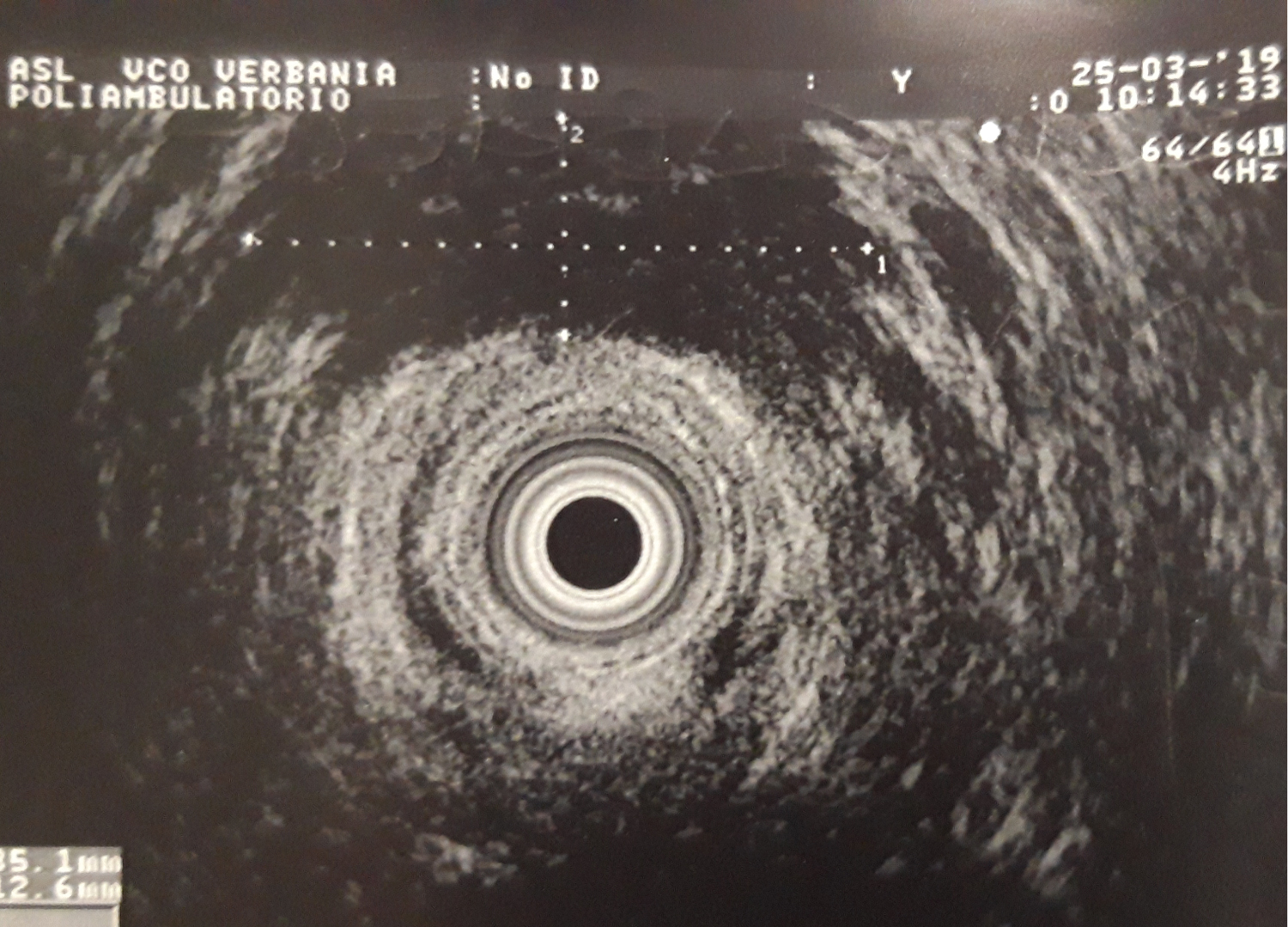 Figure 3: Transanal ultrasonography: Confirmation of the presence of a deep anterior anal abscess outside the external anal sphincter. View Figure 3
Figure 3: Transanal ultrasonography: Confirmation of the presence of a deep anterior anal abscess outside the external anal sphincter. View Figure 3
The patient underwent to incision and drainage of the abscess; an underlying transsphincteric fistula was surrounded by a loose seton. The patient received antibiotics (Ceftriaxone and Metronidazole) and supplemental oxygen with a simple face mask. He had an uneventful clinical course and was discharged 4 days after the operation in good conditions. The cultural exam of the intraoperative pus was negative.
Inborn errors of immunity (IEI) are primary immunodeficiencies characterized by increased susceptibility to infectious and autoinflammatory diseases, allergy and malignancy. They are caused by monogenic germline mutations resulting in loss or gain of function of the encoded protein.The International Union of Immunology Societies (IUIS) expert committee on IEI contribute to the biennial genotypic classification of all these disorders, organized in ten tables [4]. However, due to the growing number of IEI included in this classification (about 20 new immunodeficiencies discovered per year) and the consequent difficulty to use it at the bedside, Bousfiha, et al. proposed a more user-friendly phenotypic classification adapted for the clinician, based on the clinical and laboratory features observed in patients. [5].
Hyper IgE Syndromes (HIES) are inborn errors of immunity characterized by elevated serum IgE. The estimated incidence is of 1 in 500,000 to 100,000 individuals [1,2]. Two types of this syndrome are described: autosomal-dominant and autosomal-recessive [1-3,6,7]. The most prevalent mutation described in the autosomal-dominant Hyper IgE Syndrome is the mutation in the signal transduction and activation of transcription of STAT3 gene, involved in healing and immune mechanisms. Among other things, the mutation lead also to an intensification of the production of IgE by B lymphocytes [1-3,7]. The majority of AR-HIES is consequent to mutations in DOCK8, with absent or reduced levels of protein involved in cytoskeletal rearrangement allowing cell migration, adhesion and growth. Moreover, absence of DOCK8 results in altered immune response which might explain the susceptibility to bacterial and persistent viral infections [6].
Clinical distinction between the two forms is difficult because of the similarity of the first symptoms, such as respiratory tract infections, eczema and skin infections, combined with elevated levels of serum IgE and eosinophilia [3]. Generally, AD-HIES is characterized by various connective tissue, skeletal and vascular abnormalities [2]. Characteristic facial appearance (facial asymmetry, prominent forehead, sunken eyes, broad nasal ridge, fleshy nose, prognathism, craniosynostosis), pneumatoceles, skeletal abnormalities (scoliosis and fractures) and delayed shedding of primary teeth are typical to AD-HIES [3,7]. AR-HIES is a distinct clinical entity characterized by extensive, difficult to control and recurrent cutaneous viral and bacterial infections (Herpes Simplex virus, Human Papillomavirus, Molluscum Contagiosum virus, Varicella Zoster virus), lung infections, eczema (more severe than in AD-HIES) and extreme eosinophilia without skeletal or dental abnormalities. The patients with AR-HIES also have recurrent infections of the gastrointestinal tract, such as Salmonella enteritis and giardiasis [2,3].
Here we reported a case of a young male affected by autosomal-dominant Hyper IgE Syndrome with recurrent anterior anal abscess.
This is the first reported case about this clinical conditions in autosomal-dominant Hyper IgE Syndrome, to our knowledge. The analysis of the case leads us to the conclusion that patients with recurrent perianal abscesses (with or without other risk factors, like as Crohn disease, diabetes, obesity) should be asked about previous abscess histories in order to prevent late primary immunodeficiency diagnosis. Our patient had own diagnosis of autosomal-dominant Hyper IgE Syndrome, but some patients develop multiple anal abscess without immunodeficiency diagnosis. It's widely known that a condition of secondary immunodeficiency (e.g. HIV-related) is a risk factor for recurrence of anal abscess [8-10]. Despite that, we have not found in Literature any description regarding the relationship between primary immunodeficiency and recurrent anal sepsis. This relationship could consequently be a field of application for further studies.
The cultural exam of the intraoperative pus is essential for specific antibiotic therapy after surgery. In relation to different populations of patients, the microorganisms are differentiated in enteric flora, non-enteric flora and skin flora bacteria. The most common pathogen in childhood is Klebsiella Pneumonia, while Escherichia Coli is predominant in adulthood [11]. In our patient, the cultural exam (during all the episodes) was negative, so that we could not take specific treatment. The impossibility to isolate a microorganism could be another field of study about the possible coexistence of a immunodeficiency status.
Supportive therapy (like as good skin care, appropriate treatment and prophylaxis of skin infections and prophylactic antimicrobial therapy for pulmonary infections) were adopted in our patient, without great success. Only the delineation of the pathogenesis of HIES and creation of animal models will lead to a correct therapy of this immunodeficiency [1,3,6].
Lovisetto Federico performed the research and wrote the paper; Andrea Guala contributed to acquisition of data; Zonta Sandro critically revised the research.
None of the authors.