Angiotensin-converting enzyme inhibitors (ACE-I) are medications within the antihypertensive class that are used by nearly 108 million patients worldwide [1]. A rare but possibly life-threatening adverse effect of ACE-I is angioedema, which occurs due to elevated levels of bradykinin [2]. In this case report, we discuss a patient case where the use of tranexamic acid (TXA), an antifibrinolytic agent, prevented impending intubation due to ACE-I induced angioedema.
ACE-I, a common antihypertensive medication, regulates blood pressure by inhibiting angiotensin-converting enzyme (ACE). This inhibition prevents the formation of angiotensin II from angiotensin I, a peptide hormone that causes vasoconstriction [3]. ACE is also involved in the degradation of bradykinin, a potent mediator of vasodilation. When ACE is inhibited, the arteriolar smooth muscle relaxes and increases blood flow, leading to increased vasodilation of blood vessels, vascular permeability, and possible development of angioedema with or without the need for intubation. The development of ACE-I induced angioedema (ACE-AE) originates with kallikrein, which cleaves high molecular weight kininogen to generate bradykinin [4,5]. The initial production of bradykinin can be decreased by a known antifibrinolytic agent, TXA. TXA has been shown to improve ACE-AE and prevent impending intubation by reducing the amount of bradykinin needed to be degraded by ACE (Figure 1).
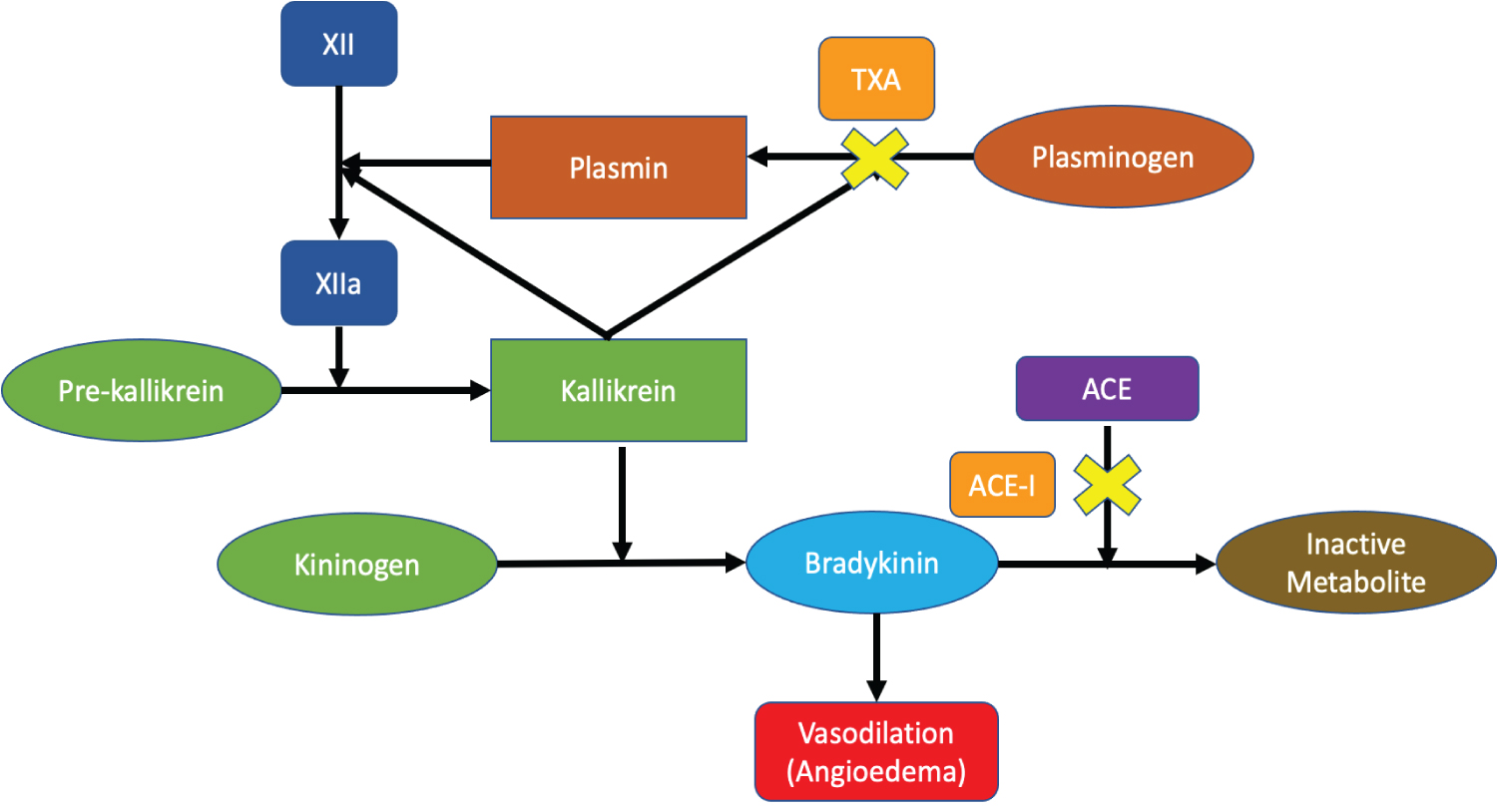 Figure 1: Mechanism of action of TXA in ACE-I angioedema. TXA reduces the circulating bradykinin by inhibiting the conversion of plasminogen to plasmin, an important enzyme for kallikrein-activation and bradykinin formation. View Figure 1
Figure 1: Mechanism of action of TXA in ACE-I angioedema. TXA reduces the circulating bradykinin by inhibiting the conversion of plasminogen to plasmin, an important enzyme for kallikrein-activation and bradykinin formation. View Figure 1
Our patient has a prominent past medical history of hypertension, coronary artery disease status post myocardial infarction, diabetes, and chronic kidney disease. Forty-five minutes prior to arrival at the emergency department (ED), the patient woke up in the morning with a swollen tongue after eating Chinese food the night before. The patient had no known drug or food allergies. The spouse noted that the patient seemed to be having difficulty breathing, thus they came to the ED. Upon arrival, the patient had an O2 saturation of 98%, temperature of 36.6 °C, respiratory rate of 17 bpm, heart rate of 65 bpm, and a blood pressure of 144/52 mmHg. The patient was initially treated for allergic reaction and received dexamethasone 20 mg, methylprednisolone 125 mg, diphenhydramine 50 mg, famotidine 20 mg, epinephrine 0.3 mg IM, and inhaled racepinephrine 0.5 mL. Thirty minutes later, the patient was also given TXA 1g due to worsening symptoms. Prior to administering TXA, the patient consented and was scheduled for intubation by the anesthesiologist in the operating room (OR). However, shortly after receiving TXA, the patient was re-evaluated by the surgeon, who noted that tongue swelling had decreased and the patient began to have clearer speech. At this time, it was deemed no intubation was needed. The patient was admitted to the intensive care unit with an O2 saturation of 98% and observed for 24 hours without any interventions (Figure 2). Counseling was provided and ACE-I allergy was documented on electronic medical records prior to discharge.
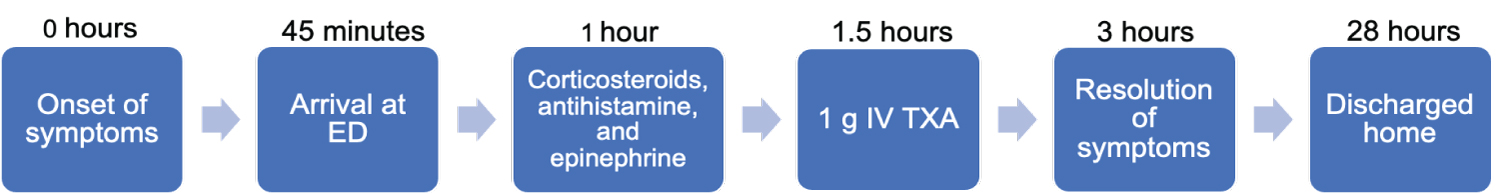 Figure 2. Timeline of events. The progression of the patient's symptoms from onset to hospital discharge is depicted. View Figure 2
Figure 2. Timeline of events. The progression of the patient's symptoms from onset to hospital discharge is depicted. View Figure 2
While ACE-AE is rare, unfortunately, it is estimated that between 0.1 to 0.7% of patients on ACE-I develop angioedema [2]. This arises most commonly within the first year of treatment. However, it may also develop after years of use. Angioedema presents as asymmetric swelling of the face, lips, larynx, genitalia, or extremities. It occurs due to elevated levels of bradykinin, leading to vasodilation of blood vessels, increased vascular permeability, and plasma extravasation into the submucosal tissue. The most severe cases include failure to secure the airway requiring intubation [4]. There are also some genetic and environmental risk factors for development of ACE-AE. These include age over 65, African American decent, smoking, and history of ACE-I induced dry cough [6,7].
Currently, there is no standard treatment for ACE-AE. Typical first line management includes a combination of histamine targeted therapies (e.g. epinephrine, antihistamines, and corticosteroids), which are likely ineffective.5 Intubation is also considered as swelling of the mucosa can lead to airway obstruction with concomitant hypoxia and potentially death. Fresh frozen plasma (FFP), icatibant, and ecallantide have all been explored in the use of ACE-AE. FFP contains ACE which can catalyze the degradation of excessive bradykinin [8]. Icatibant, a selective antagonist of the bradykinin β2 receptor, was shown in a phase II trial to decrease the time of complete resolution of ACE-AE when compared to antihistamine and corticosteroid therapy [9]. Ecallantide, a plasma kallikrein inhibitor, was studied in another phase II trial where it was thought to decrease ACE-AE by inhibiting kallikrein, the enzyme responsible for converting kininogen to bradykinin [10]. Both icatibant and ecallantide are well studied in the treatment of hereditary angioedema (HAE), which are associated with C1esterase inhibitor deficiency [5]. However, the uses of these bradykinin targeted therapies are limited in ACE-AE due to cost and efficacy [11].
A more novel approach in the acute management of ACE-AE involves TXA. As seen in Figure 1, TXA inhibits the conversion of plasminogen to plasmin, which is responsible for activating factor XII to factor XIIa. Factor XIIa stimulates kallikrein activation and cleaves kininogen, which produces bradykinin [4,6]. By reducing the amount of bradykinin, TXA can improve ACE-AE and prevent impending intubation. The average cost of TXA for 1g is $6, which makes this pharmacotherapy significantly more affordable compared to treatment options like icatibant and ecallantide [12].
Currently, there are no large, randomized, controlled trials published regarding the use of TXA in ACE-AE. However, there are limited retrospective studies and case reports justifying its role in. In 2010, a retrospective study analyzed the treatment of TXA given to 25 patients for sporadic idiopathic bradykinin angioedema. It was found that 18 patients had complete remission of symptoms, corresponding to 72%, after administration of TXA [13]. Another retrospective observational study in 2014, which included 37 patients who had either HAE or idiopathic non-histaminergic angioedema, reported that an average of 2.5g of tranexamic acid taken for six months was able to reduce the number of angioedema attacks by 75% [14]. Lastly, a 2018 retrospective review reported improvement in 27 of the 33 patients studied with ACE-AE after receiving 1 gram of TXA within 1 hour of symptom onset. In this review, none of the patients required intubation [15].
Our case report shows that TXA is a cost effective antifibrinolytic agent that reduces the symptoms of ACE-AE. The effect seen in our case is confounded by the concomitant use of histamine targeted therapies. However, TXA was considered only after the patient failed to show improvements with the aforementioned therapies.
The accessibility and affordability of TXA makes it a viable option for patients presenting to the ED with ACE-AE. Our case supports the need for further investigation to assess the role of TXA in ACE-AE.
No data were used to support this study.
The authors state no conflicts of interest.
The authors have not declared a specific grant for this research from any funding agency in the public, commercial, or not-for-profit sectors.