Diabetes mellitus is a serious global concern due to its long-term health-related effects and the financial burden it poses to healthcare systems and families around the world. Excess consumption of sugar is one of the main causes of the issue. There are many sweetening ingredients that claim to be the solution to substitute normal sugar while maintaining the blood sugar level at a low level, yet the evidence is lacking and not always substantiated. Sweetch Natural Sugar Replacement is a proprietary natural sugar alternative product composed of a blend of different plant-derived sweetening ingredients. It has zero calories, no added flavor, non-GMO, and the same taste profile and measure as sugar (1:1 replacement). It is appropriate in drinks as well as any form of cooking. The objective of this study is to evaluate changes in blood sugar levels, as well as the safety of human volunteers after oral absorption of Sweetch Natural Sugar Replacement, substantiating Sweetch claim on zero glycemic response. The efficacy and safety analyses were performed using the principles of a fasting blood sugar level test on 22 randomized subjects practicing a healthy lifestyle. Participants were divided into 2 groups: An active group using the investigational product (IP; 50 g of the IP dissolved into mineral water.) versus a controlled group using a placebo (plain mineral water). After 12 hours of overnight fasting, volunteers took their drink orally and their blood samples were taken at specific time points (T0, T30 min, and T2 h) to measure blood glucose levels. Results indicate that there is no significant difference in the average blood glucose level at various time points between the active group and the placebo group. This study confirms that Sweetch Natural Sugar Replacement does not cause any noticeable changes in blood sugar levels post-oral administration. No adverse health events were observed throughout the study. Through this study, Sweetch Natural Sugar Replacement's claim on zero glycemic index (GI) is proven legitimate.
Diabetes, Glycemic response, Blood glucose dynamics, Natural sugar replacement
GI: Glycemic Index; FBG: Fasting Blood Glucose; FAO: Food and Agriculture Organization; WHO: World Health Organization; T30: 30 Minutes-Post Load; T2 hr: 2 Hours Post-Load; Sweetch: Sweetch Natural Sugar Replacement; IP: Investigational Product; S-Day: Screening Day; T-Day: Testing Day; CRO: Contracted Research Organization
Diabetes mellitus refers to a series of autoimmune, metabolic, and genetic disorders that are caused mainly by hyperglycemia [1] - an event where there is an excessive amount of glucose circulating in the blood plasma [2]. According to the WHO, diabetes mellitus was ranked in the top 10 causes of death globally in 2019 [3]. The number of diabetic cases globally experience a continuous growth from 171 million in 2000, 285 million in 2010, and 415 million in 2015 [4,5]. It is suggested, however, that the data is most likely underestimated due to the sheer number of unreported cases in developing and emerging countries where testing methods are not always accessible. Regardless, simulations of the global diabetic rate indicate no sign of slowing down Figure 1 [6].
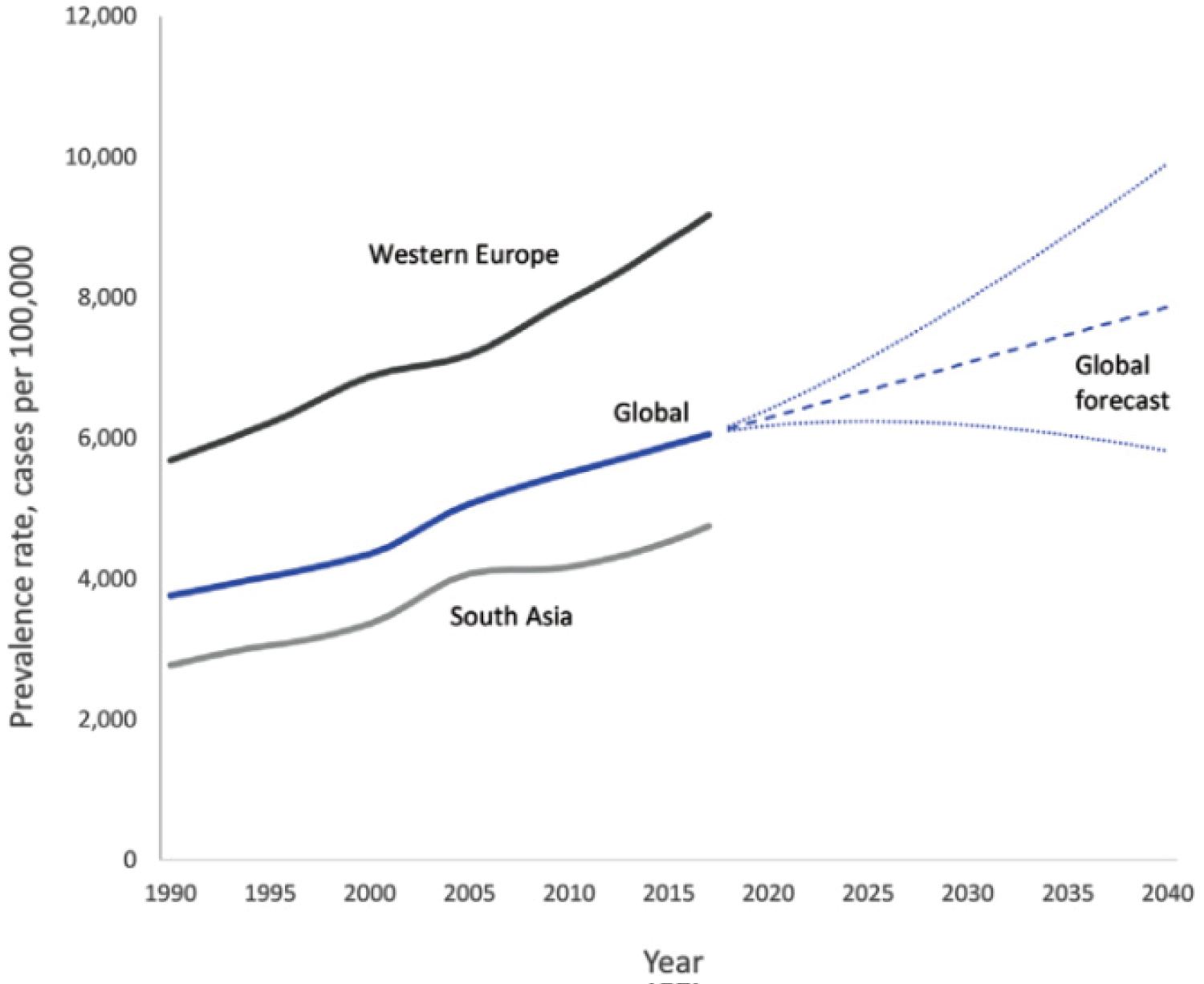 Figure 1: Trends in the global forecast for the prevalence of diabetes [29].
View Figure 1
Figure 1: Trends in the global forecast for the prevalence of diabetes [29].
View Figure 1
A sedentary lifestyle can be considered as one of the biggest causes of diabetes aside from genetic disorders. A positive relationship between the consumption of sugar, converted from carbohydrates and fat, in excessive amounts and a higher risk of diabetes type 2 has been reported [7]. Another study [8], carried out over 6 months in Nordic countries, demonstrated the negative impact of sugar-containing beverages on blood triglycerides and total cholesterol at a statistically significant level.
The world consumed 171.69 million metric tons of sugar in 2019/2020 and this is projected to be approximately 178.84 million metric tons by 2023 [9]. This is in line with the growth of the world's sugar production from 188.25 million metric tons in 2018/2019 increasing by 21 million in 3 years. India, the world's largest sugar manufacturer, reported an increase in national consumption [10]. In addition, observations [7] showed that high sugar content can also be found in fast food, making diabetic very prominent for a nation where fast-food culture has been and is on the grow. Sugar consumption may be associated with inevitable cultural and hedonic relevance [11]. For instance, it brings Indians to a way of joyful life, and it is mandatory to offer sweets to ancestors and gods [8]. Furthermore, the speedy pace of urbanization has led to a bigger portion of the population having access to more food choices, often unhealthy and filled with sugar.
Parallel to the rise in sugar intake observed in India, the US is also witnessing a similar trend. America's national sugar consumption per capita ranked first in the world with 126.4 grams of daily use [12]. This is 3 to 4 times higher than the recommended intake from the World Health Organization [13] and 10 times more than the recommended intake by the United Kingdom National Healthcare System [14]. In addition, the total sugar consumption in America increased by 1 million metric tons in 2021, compared to 2019 according to the latest report [6].
From a different standpoint, despite the studied relationship between sugar consumption [15], diabetes mellitus, obesity, and cardiovascular disorders, some evidence [16] also show that this relationship expands beyond obesity as 40% of patients suffering from diabetes are non-obese. Increasing sugar consumption over a short period of time may be responsible for the development of this unusual relationship [17]. As a result, solutions to reduce sugar consumption are essential. The need for an alternative, healthy source, of sweeteners is more relevant than ever considering that the cost of diabetes mellitus treatment has risen by 25% in a 5-year period since 2012 [18]. With the financial burden, there has been a promotion for diabetic prevention by reducing the daily sugar intake to control the blood sugar index [15]. This is further supported by another piece of evidence that suggests diabetes mellitus is a chronic disease that needs consistent treatment to maintain a healthy level of blood sugar [19]. However, completely replacing sugar might not be achievable for all diabetic patients. Findings from research [20] on diabetic patient behaviors have indicated that lowering sugar intake could be associated with negative moods and feelings, such as frustration, and depression. This craving for mood-lifting energy has led to an increased dependency on sugar [21]. From an industrial manufacturing standpoint, cutting sugar out of formulations seems to be a challenge for many food and beverage manufacturers as it will affect the appetizing taste and texture, the key drivers for their product sales [15]. Regardless, changes need to start taking place, and the technological advance in sugar substitute product is the start.
Sweetch is a proprietary natural sugar substitute, replacing regular refined sugar for daily consumption. Developed with all-natural plant-based GRAS ingredients, adding the benefit of maintaining the blood glycemic index and having no calories while ensuring the taste, texture, and usage measure of sugar. The product does not contain any preservative, artificial flavor, and/or color. All ingredients are approved GRAS and meet non-GMO requirements as well.
Fasting blood sugar (FBG) is an essential test to determine the blood glycemic level after an overnight fast. The test is commonly used for detecting diabetes mellitus type 2, with or without symptoms, or pre-diabetes [22]. The role of this test has recently been determined as a parameter to diagnose the development of cardiovascular disease in healthy subjects [23]. The fasting period is required to allow the body time to fully process all consumed carbohydrates or sugars prior to testing, allowing a more accurate result.
The purpose of the reported study is to examine whether Sweetch Natural Sugar Replacement is effective in not causing any increase in blood sugar levels after oral administration. Data is collected at various time points, following the principal protocol set for an FBS test.
Hypothesis 1:
H0: There is no difference between the two treatments1
H1: There is a difference between the two treatments1
1: The treatments of Sweetch against the placebo
Hypothesis 2:
H0: There is no difference between the two timepoints2
H1: There is a difference between the two timepoints2
2: The T30 min and T2 h time points
1. To assess the efficacy of Sweetch, a natural sugar replacement, on healthy human volunteers by measuring the blood sugar dynamics at different time points (T30 min and T2 h) post load.
2. To assess the safety of the test product by monitoring any potential adverse health event.
This study was conducted in compliance with the applicable international and local regulatory requirements as well as applicable ICH guidelines, Helsinki Declaration (2013), other applicable regulatory guidelines, and in respect of the Contracted Research Organization (CRO) internal Standard Operating Procedures (SOPs) and Work Instructions (WIs).
Sweetch Natural Sugar Replacement does not contain any added sugar and based on its studied composition, has an expected glycemic index of zero. The study is meant to confirm this by comparing the blood sugar dynamics following intake of dissolved Sweetch Natural Sugar Replacement with a control taking plain, mineral water.
Twenty-two subjects (11 males and 11 females) who practice a healthy lifestyle were recruited to take part in the study. The first visit was reserved for health screening. Subjects who failed to meet all required biometric health parameters were rejected. Basic selection criteria for suitable participants include age, height, weight, BMI, and FBG. An additional medical examination was taken to reveal any details on relevant surgical history or medical risks including allergies or drug sensitivity. During the health screening, subjects were asked to complete a Habitual Questionnaire. The answers given would allow a good assessment of the subjects to meet the requirements for living a healthy lifestyle with insight into existing health conditions, exercise, and diet routines. The study was performed in New Delhi, India on 24 September 2022.
The twenty-two participants were randomly assigned into two groups: the active group orally received 50 g of the investigational product (IP) dissolved in mineral water and the controlled (placebo) group orally received plain mineral water.
Inclusion criteria were as follows: 20-60 years-old (both age inclusive), practice a healthy lifestyle, body mass index (BMI) less than 30 Kg/m2, no known pre-existing metabolic disorders, willing to remain in 12 hours fasting overnight before blood sample collection, agreeing for blood sample collection multiple times, informed about the study objectives and procedures, and able to understand them, willingness to come for regular follow up visits if required, willingness to give a voluntary written informed consent and being able to understand English or Hindi, willingness and being able to fulfill the study requirements and schedule, willingness to undergo urine pregnancy test (applicable for female of childbearing potential).
Non-inclusion criteria were pregnant or lactating individuals, FBG higher than 110 mg/dl, systolic blood pressure less than 90 mm or higher than 140 mmHg, having an unusual diet revealed by habitual questionnaire (for example binge eating, Air diet and juice diet…), history of allergy and known hereditary Phenylketonuria (PKU), participating in any other therapeutic trial in the last 4 weeks, history of underlying uncontrolled serious chronic medical illness including diabetes, hypertension, metabolic disorders, liver disease or history of alcoholism, HIV, or any other serious medical/surgical condition or medication from past three months that would interfere with the participation in the trial, starting, stopping, or changing his/her hormonal treatment in the previous one month prior to the study, history of severe alcohol intake and/or smoking habits, being on any topical or systemic medicine, supplement or treatment that could interfere with the study treatments/assessments within the last 4 weeks prior to participation in the study, and during the study, being unable to be contacted by phone in case of emergency.
Each subject attended 2 visits to the study center for S-day and T-day. On the S-day, the subjects were asked to sign an informed consent form (ICF) for blood sample collection for FBG measurement. The inclusion and exclusion criteria assessment were evaluated by the principal investigator, along with the demographic data, medical history interview, and habit questionnaire.
In preparing for the T-day instruction, subjects were asked to restrict their alcohol the day before the test. They were also asked not to eat or drink anything for 12 hours before the test, and until blood sampling for T2 h is done. On T-day, the sample was taken for FBG (T0 timepoint - Preload). The participants were randomly allocated to either the active group or the placebo group according to the randomization schedule and asked to drink the glass (water for placebo, water with 50 g of Sweetch for active group). Blood sampling was conducted at T30 Min and T2 hour time points post-load. Throughout the test, all participants were closely monitored for any adverse health event.
The pre-established study protocol is used to guide the measurement of the blood sugar level. A Glucometer was used the first time on the health screening visit, while on the T-day, approximately 3-4 ml of blood sample was collected intravenously from each subject at each set time points (T0, T30, T120) under sterile conditions.
Descriptive statistics for all numerical variables are presented as mean ± SD (standard deviation). Microsoft Excel (Office 365) and IBM SPSS version 19.0 were used for statistical analysis purposes. P-values of less than 0.05 were considered statistically significant, insignificant differences are defined as P-values greater than 0.05.
The study protocol was developed and conducted in compliance with the ethical principles adopted by the 18th World Medical Congress in Helsinki in 1964 and amendments adopted in 1975, 1983, 1989, 1996, and 2000, and clarifications in 2002, 2004, 2008, and 2013.
The test was performed in accordance with guidelines issued for Good Clinical Practices (GCP) published by ICH Topic E6, Ethical guidelines for biomedical on human participants published by ICMR, and in compliance with local regulatory requirements.
The protocol, Subject Information Note and ICF, and other requested documentation were reviewed and approved by an appropriately constituted ethic committee as per Indian Regulations NDCT rules 2019.
On the screening day, an evaluation for inclusion and non-inclusion criteria was done. 22 healthy subjects out of a total of 27 screened subjects (14 males and 13 females) were enrolled and randomized for the T-day. All completed the study without any protocol deviation. 5 subjects were not qualified due to failure in meeting the screening requirements and were excluded from the data. The biometric characteristics of the 22 studied subjects gathered from the screening are present in Appendix.
On the T-day, following the testing protocol, each group was administered either the testing product dissolved in water or pure water as a controlled placebo. Following the FAO guidelines, which states that any food portion testing for the glycemic response, should contain 50 grams of carbohydrates [23,24] the test was done giving 50 grams of Sweetch Natural Sugar Replacement to the volunteers assigned to the IP group.
At designated time points T30 minutes and T2 hours, blood samples from each group were collected for the measure of the glycemic response. The results presented in Table 1 and Figure 2 show that there is no significant difference in the mean blood sugar levels between the testing group and the placebo group at the designated period starting from T0 to T30 minutes. Specifically, from T0 to T30 minutes, the testing group has an average blood sugar level difference of 4.6 and for the placebo group, 3.2, p-value = 0.921. At T0-T2 hr, the mean blood sugar difference for the testing group is 3.2 and the mean difference for the placebo group is 7.5, p-value = 0.15.
 Figure 2: Blood sugar levels of the two groups. Error bars 95% CI.
View Figure 2
Figure 2: Blood sugar levels of the two groups. Error bars 95% CI.
View Figure 2
Table 1: Evolution across time and treatment comparison for "Blood Sugar" (n = 11 for both groups). View Table 1
This study was conducted to assess the effectiveness of Sweetch Natural Sugar Replacement in not raising blood sugar levels in healthy human volunteers as well as its safety.
In the past, when the FAO standards were applied, subject non-compliances were recorded [15]. However, throughout this study, the result indicates good compliance with the administering amount of 50 g/testing subject, proving the study protocol was comfortably accepted by the volunteers, ensuring the accuracy of data.
Blood sugar level will increase gradually as carbohydrates are digested in the stomach [25]. This process could last up to 2 hours before the blood glucose level would return to normal [26]. Contrary to the principal statement above, the result of the study shows that the 11 testing subjects have a significantly decreased mean blood sugar level after 30 minutes of consuming Sweetch sample dissolved in mineral water, from 86.8 mg/dL to 82.1 mg/dL (p = 0.041). This indicates the use of Sweetch sugar replacement allows testing subjects to absolutely maintain their blood sugar level at a level equivalent to or slightly below zero consumption of sugar. This decrease can be explained by the fact that the body still needs the energy to run the internal metabolism and support physical activities, hence a portion of the existing blood sugar level was used. The decrease in blood sugar level post-load could be subjected to further investigation in a larger sample pool to validate repeatability.
From T30 minutes to T2 hrs, there was a slight increase in the mean blood sugar level of the testing group from 82.1 mg/dL to 83.5 mg/dL (p = 0.077). However, the increase is not significant.
Finally, when comparing the result between the two groups, no statistically significant differences were observed between the glycemic responses of the testing group (solubilized Sweetch Natural Sugar Replacement) and placebo (plain mineral water) at time points between T0 and T30 min (p = 0.921), and T0 and T2 h (p = 0.153). This demonstrates that Sweetch Natural Sugar Replacement does not cause any modification in the blood glucose dynamics after consumption and carries the same glycemic effect to the users as if they were to drink plain mineral water.
Various in-vitro studies suggest that natural sugar-replacing ingredients including stevia could have several antidiabetic properties [27]. The antidiabetic property of Stevia is [28,29] explained by the increase of the peroxisome proliferator-activated receptor-γ (PPARγ) levels leading to improved insulin sensitivity [30] and a faster rate of glucose breakdown. Another piece of evidence suggests that stevia may have an insulinomimetic effect by increasing the expression of the transmembrane glucose transporter type 4 proteins (GLUT4) [31], which allows glucose uptake by body cells at a faster rate. Further research is still necessary to gain a better understanding of the exact working mechanisms that allow stevia and other non-caloric sweetening ingredients to be processed in the body in ways that it will not affect the blood sugar level. The negative aspects of stevia glucosides are in the bitter aftertaste. The sweetness is roughly 600 times the sugar sweetness, which means that recipes must be adjusted and, the quantity being so small it is not suitable for baking, as the texture is different.
This in-vitro evidence strongly aligns with the result of this study by providing an explanation of how the components of Sweetch Natural Sugar Replacement could have been metabolized in the bodies of the volunteers, leading to non-significant difference between the blood glucose dynamics of the active group and the placebo group.
From a clinical standpoint, the non-glycemic property of numerous natural sugar-replacing ingredients like stevia has been clinically evaluated. For instance, a study was conducted on the effectiveness of stevia on 34 type-2 diabetic patients for 2 months [32] indicating no statistically significant differences. Results from this study correlate the finding with Sweetch Natural Sugar Replacement, indicating a potential non-interfering impact on blood glucose parameters (FBG included) in diabetic patients.
A point of emphasis is the duration of the study. The study conducted by Aiami, et al. [32] was over the span of eight weeks. Another meta-analysis [29] of Stevia's therapeutic effectiveness against diabetes was also demonstrated with a long-term study period (from 3 days to 3 months). This suggests that an additional study could be conducted to test for the long-term effect of Sweetch natural sugar replacement on human blood sugar levels, which will give a holistic view of the performance of Sweetch in not raising blood sugar levels in both the short-term and long-term. This could be achieved by implementing a hemoglobin A1C methodology, which will reveal one's blood sugar level over the course of 90 days [33].
Safety is one of the most important aspects of any food form product. The safety topic of various sugar replacement products on the market has been heavily debated. The use of saccharin, which is an artificial sweetener with no calories, has been tested on rats and weight gain leading to obesity was reported [34]. This ingredient later has been banned by the US FDA. In addition, evidence studying the toxicity of aspartame on participants who were sensitive to this low-calorie sweetener indicated headache frequency, blood pressure parameter, blood histamine concentrations, and allergic symptoms against control groups [35], proving potential adverse health effects. Just because a product claims to be a sugar replacement, it does not mean it is safe and healthy. In this study, volunteers of the testing group were closely monitored throughout the duration of the study by health professionals. At the conclusion of the study, no adverse event was reported from any volunteers. This data strongly suggests that Sweetch can be considered safe. This statement is further strengthened by knowing that every ingredient used in the formulation of Sweetch, has been granted generally accepted as safe (GRAS) by the FDA.
This study was conducted to assess the efficacy and safety of Sweetch as a natural sugar replacement, on healthy human volunteers by measuring changes in blood glucose dynamics at different time points (T30 min & T2 h) post load. The results support the acceptance of H0 of hypothesis 1, indicating no significant changes in blood sugar levels after the consumption of Sweetch Natural Sugar Replacement between the testing group and the placebo group. The product was well tolerated with the perceivable taste being the same as regular refined sugar. This study also confirms the safety of the product with no adverse event witnessed in the testing group. The information gathered in this study can be considered as a reference point to guide the application of Sweetch Natural Sugar Replacement as a healthy and safe replacement for sugar in foods and beverages where sweetening ingredients are required.
All authors have read and agreed to the published version of the manuscript and have contributed equally to its content.
The clinical study was conducted according to the guidelines of the Declaration of Helsinki, OECD Good Laboratory Practices (GLP), and within the national guidelines of India.
Informed consent was acquired from all participants involved in this study.
The authors declare that they do not have any conflict of interest.