It is time to embrace a more nuanced view of what it means to be "diabetic" and how to manage our patients with diabetes. To be clear, this is not a criticism of current doctrine or those that came before us. In fact, this is a call to recognize that time, knowledge, and understanding marches forward and we should not be too slow to appreciate this fact.
To wit, rather than diabetes classifications being fixed in stone, let us recognize that, great variation and overlap of "typical features" exists between the current DM classifications [1]. For example, approximately 30% of patients diagnosed with Type 1 diabetes mellitus (T1DM) have insulin resistance where as T-cell mediated autoimmunity is prevalent in patients diagnosed with Type 2 DM (T2DM) with reports of cellular and humoral islet autoimmunity in 41% and 14%, respectively, of patients with T2DM on metformin in the National Institute of Diabetes and Digestive and Kidney diseases GRADE (Glycemia Reduction Approaches in Diabetes) clinical trial [2,3]. Additionally, it is imperative to realize our ability to detect frank complications is limited and the damage and processes begin early in the disease continuum [4]. Further, it is important to widen our view of the causes of diabetes and its complications to incorporate common pathophysiologic processes beyond "just metabolic" [5,6]. Indeed the pathologic mechanisms that damage the b-cell (i.e. genetic/epigenetics, inflammation, abnormal environment, and insulin resistance) also contribute to cell and tissue damage elevating diabetes-related complications but also link to other common conditions including cancer, dementia, psoriasis, atherosclerotic cardiovascular disease, nonalcoholic fatty liver disease, and nonalcoholic steatohepatitis (Figure 1) [6].
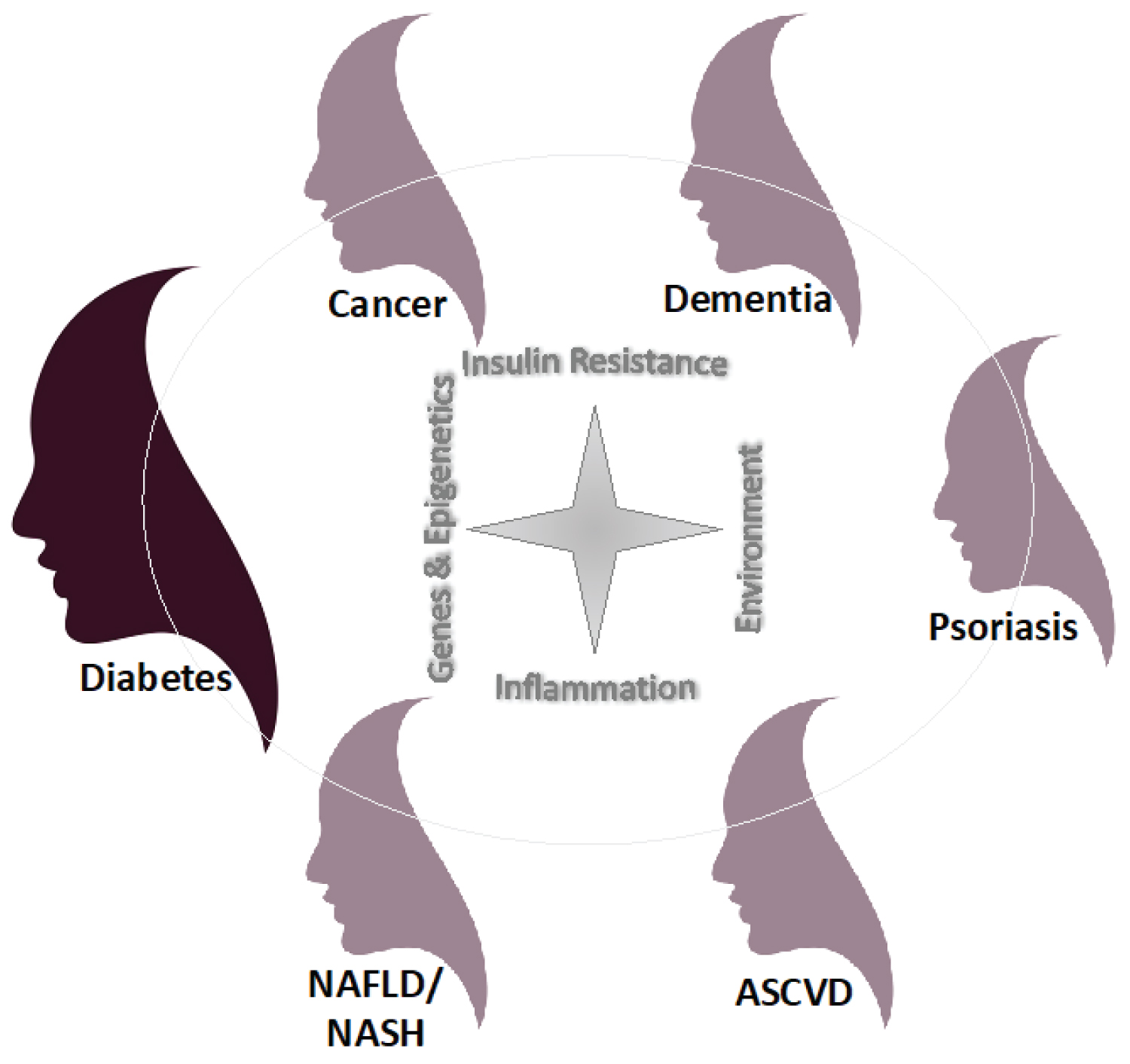 Figure 1: Common pathophysiologic mechanisms and triggers that an individual patient has or is exposed to can determine the clinical presentation or "face" of diabetes and other commonly linked conditions.
Figure 1: Common pathophysiologic mechanisms and triggers that an individual patient has or is exposed to can determine the clinical presentation or "face" of diabetes and other commonly linked conditions.
ASCVD: Atherosclerotic Cardiovascular Disease; NAFLD: Nonalcoholic Fatty Liver Disease; NASH: Nonalcoholic Steatohepatitis.
View Figure 1
With this said, this opens up the potential to begin to think differently with regard to how we manage our patients. Rather than feel stuck in the mud with single medication therapies for DM, we should fell emboldened to approach its management with multimodal approaches. Think of the money wasted trying to determine which drug was "better" when we could be considering drugs with complimentary benefits and mechanisms of action that address the multifaceted etiology of the DM spectrum [6]. In addition, some newer therapeutic classes not only have proven benefits for managing hyperglycemia, but also reduce cardiovascular and kidney risk as well as promote weight loss [6]. Furthermore, moving our understanding of a patient "unlucky enough" to get more than 1 disease besides DM toward recognizing the interconnection between common conditions allows us to further add to our armamentarium of potential drugs to address common etiologies across disease [6]. This broader view brings us closer to precision medicine for our patients. Importantly, as espoused by Dr. Sniderman of the McGill University Health Centre, clinicians must continue to value clinical reasoning and logic as well as evidenced-based medicine in order to move individualized patient care safetyforward [7]. We should endeavor to treat each patient as if they are our own family, recognizing that clinical reason AND science-based approaches are best, as we await evidenced-based corroboration of current therapies and as well as potential new advanced technologies (e.g, CRISPER, artificial intelligence [AI]) and novel drugs [8-10]. Indeed, imagine a future with AI-powered precision medicine building algorithms to classify key pathophysiologic processes and markers enabling physicians to identify those at risk for disease and complications and determine the best therapeutic options for their patients on an individual basis [9,10]. Till then, we should continue to open to the recognition and treatment of diabetes in all its different faces with individualized approaches to deliver the best care for our patients.
Dr. Schwartz received no funding for this work. Dr. Schwartz has no current conflicts of interest. Amy W Rachfal, PhD of Stagegate Partners, LLC, assisted in the consolidation and editing of this article.
SS Schwartz received no funding for this work and has no current conflicts of interest.