Primary cutaneous aspergillosis (PCA) is one of several invasive fungal infections that have increased in incidence in the last four decades. We present a case of an extremely low birth weight infant born at 24 weeks gestation diagnosed with cutaneous aspergillosis, highlighting the risk factors and clinical findings associated with neonatal PCA. Additionally, we discuss use of serum galactomannan testing as well as combination amphotericin B and voriconazole therapy. Early recognition of lesions characteristic of PCA is required to ensure prompt diagnosis and treatment, potentially preventing dissemination with favorable outcomes.
Primary cutaneous aspergillosis, Extremely low birth weight, Prematurity, Serum galactomannan, Amphotericin, Voriconazole
A 24-week gestation male, birth weight 620 gram, was born to a 27-year-old mother by vaginal delivery secondary to placental abruption and preterm labor. Following initial resuscitation, his first week of life was complicated by mechanical ventilation, placement of a central venous catheter, hypotension, leukopenia, and concern for sepsis. Treatment included ampicillin, gentamicin, changed to vancomycin and meropenem, as well as dopamine and hydrocortisone. Despite negative cultures, he was treated for 7 days for clinical sepsis.
On day of life 9, physical examination revealed a violaceous bullous lesion on the infant's back with surrounding erythematous papules. Blood cultures as well as surface swabs for herpes simplex virus (HSV) polymerase chain reaction (PCR) testing were obtained. Antimicrobial therapy with vancomycin, gentamicin, fluconazole, and acyclovir was initiated. The lesion quickly flattened, forming a necrotic plaque measuring 4 cm × 2 cm (Figure 1). Wound and tissue cultures revealed Aspergillus fumigatus; mold blood cultures were no growth. Herpes simplex virus PCR testing was negative and bacterial cultures were sterile. A punch biopsy was performed on the lesion; presence of Aspergillus was confirmed by histopathology which revealed invasive septate fungal elements and leukocytoclastic debris (see Figure 2) and polarizable material (see Figure 3). This confirmed the diagnosis of primary cutaneous aspergillosis (PCA).
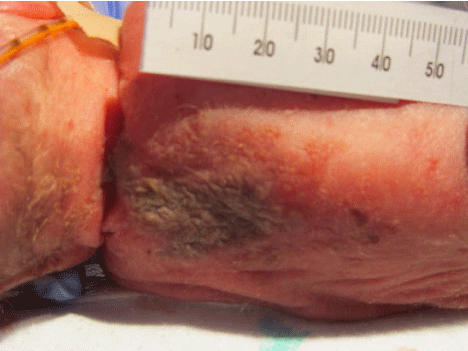 Figure 1: Appearance of the 4 cm × 2 cm necrotic lesion on the neonate's back two days after it first appeared. View Figure 1
Figure 1: Appearance of the 4 cm × 2 cm necrotic lesion on the neonate's back two days after it first appeared. View Figure 1
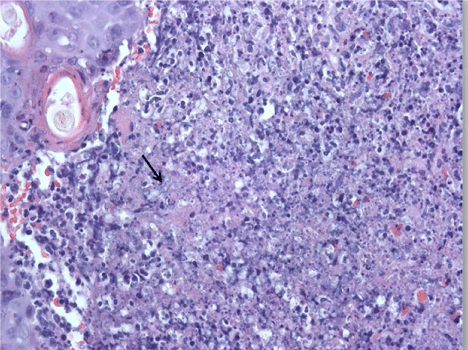 Figure 2: Biopsy with area of dermal infiltrate containing neutrophils, leukocytoclastic debris and fungal elements- (arrow) (H&E Stain) Original magnification ×400. View Figure 2
Figure 2: Biopsy with area of dermal infiltrate containing neutrophils, leukocytoclastic debris and fungal elements- (arrow) (H&E Stain) Original magnification ×400. View Figure 2
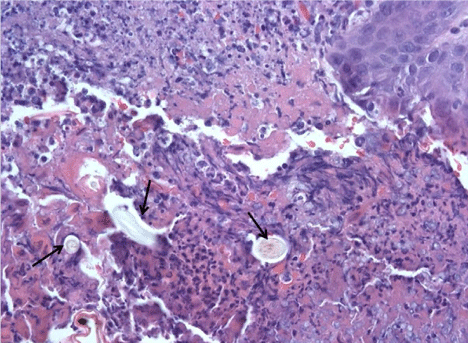 Figure 3: Biopsy with area of overlying scab containing polarizable material (arrows). (H&E stain) Original magnification ×400. View Figure 3
Figure 3: Biopsy with area of overlying scab containing polarizable material (arrows). (H&E stain) Original magnification ×400. View Figure 3
Treatment was changed to amphotericin B, with addition of voriconazole when serum galactomannan antigen was detected (3.664 index with reference range: < 0.5 index). Transthoracic echocardiogram, renal ultrasound, and eye exam did not reveal evidence of A. fumigatus dissemination; due to risk of fungal inoculation from the rash location during a lumbar puncture, no cerebrospinal fluid was obtained. Following 6 weeks of antifungal treatment and frequent excisional wound debridement, the lesion healed, leaving pink tissue with minimal scarring. At the end of treatment, serum galactomannan antigen was not detected.
Primary cutaneous aspergillosis (PCA) is an invasive fungal infection with increasing incidence in the last four decades [1,2]. Extremely low birth weight (ELBW) infants, due to their immature skin/lack of barrier, receipt of broad-spectrum antibiotics, and poor immune function, are at risk for invasive aspergillosis of the respiratory tract, sinuses, or skin [1-3]. Pathogenesis occurs when Aspergillus conidia are deposited on mechanically disrupted skin after which subsequent invasion by fungal hyphae leads to infection [4]. Skin lesions are classically described as erythematous patches and/or plaques with necrotic ulcers [3].
A review of neonatal PCA highlights the risk factors and clinical presentation: 1) Premature delivery, especially ELBW, 2) Use of support devices, such as mechanical ventilation and central venous catheter, 3) Exposure to corticosteroids, 4) Receipt of broad-spectrum antibiotics, and 5) Environmental exposure to Aspergillus [1-3]. Hospital renovation, as well as exposure to Aspergillus-contaminated latex gloves, has been linked to PCA outbreaks in neonatal intensive care units (NICUs) [4,5]. Renovations were being conducted at remote sites in our institution, but not directly in our NICU; environmental testing of the infant's NICU pod unit as well as ventilation duct work did not demonstrate environmental Aspergillus contamination. No other cases of PCA occurred in the 24 months following this case.
The use of serum galactomannan, a cell-wall product of Aspergillus, is a potential diagnostic marker of invasive aspergillosis, in addition to its usefulness to monitor treatment response [6,7]. Pediatric studies have demonstrated high sensitivity and specificity of galactomannan, 89% and 85% respectively, for aide in diagnosing invasive aspergillosis [6]. A limitation to the use of galactomannan in neonates includes a lack of studies performed as well as the inability to calculate the positive- and negative-predicted values of galactomannan, given the unknown incidence of infection in the neonatal population. A neonatal factor which may influence false positive galactomannan testing includes gastrointestinal colonization with Bifidobacterium in these small patients [2]. It has been suggested, however, that in the clinical setting of PCA, a positive galactomannan test is diagnostic of disseminated disease [1,3-5,8-13]. Disseminated Aspergillus infection carries mortality approaching 77%; therefore the usefulness of galactomannan may be timely and lead to prompt diagnosis and treatment of PCA.
Successful treatment of PCA is accomplished by combined medical and surgical approaches [2]. Based largely on adult studies of pulmonary aspergillosis, guidelines recommend voriconazole as a first line agent for invasive aspergillosis, while most of the reported cases of PCA in the literature have been treated with amphotericin B [7,9-11]. Our patient was initially treated with amphotericin B, however when galactomannan levels were found to be elevated, combination therapy with voriconazole was successfully added. This case illustrates that studies are needed to delineate the role of serum galactomannan in suspected neonatal aspergillosis, as well as to provide definitive treatment guidelines of Aspergillus infection in neonates.