Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is new genus of beta coronavirus that produces diverse neurologic manifestations like myalgia, headache, delirium, anosmia, ageusia, acute ischemic or hemorrhagic stroke, seizures, encephalopathy and rarely Guillain Barr syndrome (GBS), meningoencephalitis and acute disseminated encephalomyelitis (ADEM) [1,2]. 20% of severe COVID-19 intensive care unit admissions are because of some neurological deficits [3]. SARS-CoV-2 virus are very resemblance with SARS-CoV-1 and MERS and can have potential to damage the central and peripheral nervous systems via direct or systemic ways [4,5]. The initial critical steps in pathogenesis of COVID-19 infection are binding of SARS-CoV-2 with angiotensin converting enzyme 2 (ACE-2) [6]. The role of ACE-2 is to control blood pressure through inhibiting of angiotensin-renin-aldosterone pathways. It helps in conversion of angiotensin II to angiotensin (1-7) [7]. High level of angiotensin II may cause vasoconstriction, renal failure, cardiac injury, apoptosis and oxidative insults [8]. ACE-2 are present in cell membrane as a receptor in multiple organs (like respiratory, kidneys, liver, blood vessels, immune system, skeletal muscles and brain) [9]. After binding with ACE-2 receptors and endocytosis of SARS-CoV-2 virus in respiratory ciliated epithelial cells, virus force cells to produce more copies of its and infect more cells and triggers innate immune system to excessive release of proinflammatory signaling molecules (cytokines) [10].
Hypercytokinemia (cytokine storm) causes systemic hyperinflammation, increase vascular permeability, hypercoagulation and edema [11]. Cytokine storm contributes to ARDS, kidney failure, liver injury, heart failure, myocardial infarction as well as multiple neurological conditions. This may also trigger vasculitis/endothialitis in and around nerves and muscles [12]. A direct invasion of SARS-CoV-2 to the muscles may cause viral myositis [13]. The early time course of severe muscle weakness in COVID-19 infection may suggestive of viral myositis and may be complicated by rhabdomyolysis [14,15]. Skeletal muscle injury and high creatine kinase (CK) in critical COVID-19 patients in intensive care units may contributes to critical illness myopathy with other risk factors like used of steroid, neuromuscular blocking agent and SIRS/sepsis. In patient with myocardial injury, there is evidence that both myocarditis due to SARS-CoV-2 virus as well as myocardial infarction due to cytokine storm, hypercoagulability, and ischemia can happen at the same time [16]. In published studies of COVID-19 in China, myalgia or fatigue affected 44-70% of hospitalized patients and increased creatine kinase (CK) was present in up to 33% of admitted patients [17]. In postmortem muscles biopsies of COVID-19 patients, there were no SARS-CoV virus identified [18].
Exact mechanism of skeletal muscle injury in covid 19 patients is not fully recognize. Direct SARS-CoV-2 viral invasion of skeletal muscle through ACE2 receptor, hematogenous dissemination and immune-mediated skeletal muscles injury secondary to activation of immune cells and inflammatory response with cytokine storm syndrome may be possible mechanism of skeletal muscle injury, muscle fiber proteolysis and fibrosis [19]. The available data at present suggest that COVID-19 associated muscle injury is most likely para-infectious and or immune-mediated and can manifest as dermatomyositis-like interferonopathy-1, myositis, or immune-mediated necrotizing myopathy [20]. Direct SARS-CoV-2 viral invasion of diaphragm muscle may contribute to diaphragm dysfunction and lead to difficulty to weaning from mechanical ventilation and respiratory failure [21-24].
This is retrospective observational study with IRB Protocol ID # MRC-01-20-523. This study was done at Hamad medical cooperation (HMC), Doha, Qatar. There were allocated tertiary care multidisciplinary hospitals by government to manage patients with COVID-19 infection. 413 COVID-19 patients were retrospective evaluated from January, 01, 2020 to December, 31, 2020. A positive confirmed COVID-19 infection defined as a result of real time reverse transcription polymerase chain reaction (RT-PCR) test positive on both nasopharyngeal and oropharyngeal specimens simultaneously. Routine laboratory testing (CBC, CMP, PT/APPT/INR, RFT, LFT, CRP, Ferritin, LDH, D-dimer, IL-6, lactic acid, CPK, myoglobin, troponin etc.), radiologic examination included chest X-rays, chest CT, EMG/NCS studies and lumbar puncture (if clinical indicated) all was performed according to standard clinical practice and CDC Qatar guidelines. Study was approved and written informed consent was waived by IRB # MRC-01-20-523 of HMC, Doha, Qatar, on October, 07, 2020, due to pandemic of COVID-19 infection and urgent requires of new data collection and analysis result for improves clinical practice. We retrospectively evaluated electronic medical records including laboratory and radiologic findings of all patients with confirmed SARS-CoV-2 infection and collected data on age, gender, symptoms (fever, cough, SOB, myalgia, headache etc.), comorbidities (hypertension, diabetes, obesity, smoking, ischemic heart disease, stroke and chronic kidney disease etc.). All neurologic manifestations were reviewed and confirmed by 2 trained neurologists. Skeletal muscles injury was defined as when patient had myalgia and high serum creatine kinase level > 300 U/L or myoglobin level >72 ng/ml.
Rhabdomyolysis was defined as when patients had elevated serum creatine kinase level > 1000 U/L. The primary objective is to determine the incidence of skeletal muscle injury in Covid-19 patients and secondary objective to determine the association of other multisystem organ injury including acute myocardial injury and acute kidney injury in Covid-19 patients. The incidence of skeletal muscle injury in Covid-19 patients and to determine the association of other multisystem organ injury including acute myocardial injury and acute kidney injury with skeletal muscle injury in covid-19 patients estimated and presented with 95% CI. Descriptive statistics used to summarize and determine the sample characteristics and distribution of various considered parameters related to demographic, diagnostic, clinical features, follow-up outcome measures and other related features of such patients. The normally distributed data and results reported with mean and standard deviation (SD) with corresponding 95% confidence interval (CI); the remaining results reported with median and interquartile range (IQR). Categorical data summarized using frequencies and percentages. Associations between two or more qualitative variables examined and assessed using Pearson Chi-square and Fisher Exact tests as appropriate.
Unpaired t and ANOVA used to compare mean values of different quantitative parameters between two or more than two groups. Correlation between various outcomes measured evaluated quantitatively calculated by Pearson or Spearman rank-order correlation. An appropriate regression analysis performed to assess the effects of various factors and covariates on primary outcome measures. Pictorial presentations of the key results made using appropriate statistical graphs. A two-sided P value < 0.05 statistically significant. All statistical analyses done using statistical packages SPSS 24.0 (SPSS Inc. Chicago, IL) and Epi Info 2000 (Centers for Disease Control and Prevention, Atlanta, GA).
A total 413 patients that hospitalized with confirmed COVID-19 infection were included in this study. Their demographic shown in Table 1 and clinical characteristics are shown in Table 2 and Bar Chart 1, Bar Chart 2. Their Age mean (SD-14) was 52 (range from 22 to 86 year) years, and 94% were men (389) and 6% were female (25). 57% were less than 55 years age, 43% were greater than 55 years age and 5.6% were greater than 75 years of total patients. Different nationality with majority were 20% of Indian (84), 12.5% of Bangladeshi (52), 10% of Qatari (42), 9% of Nepalis (38) and 8.7% (36) of Filipino and Pakistanis. In Table 3 shown Underlying risk factors. Of these all patients, following underlying risk factors including 47.6% hypertension (197), 46.9% diabetes (194), 21% obesity (88), 10% newly diagnosed (42), 10% chronic kidney disease (43), 9.7% ischemic heart disease (40), 6.8% smoker (28), 3.6% asthma (15) and 2.91% malignancy (12). Neurologic manifestations were categorized into three major categories: Central nervous system (CNS), peripheral nervous system (PNS) and skeletal muscles. Skeletal muscle injury was 47.8% and rhabdomyolysis was 15.7% (as shown in Bar Chart 3, Bar Chart 4 and Bar Chart 5). CNS manifestations were 26.7% and PNS were4.3%. 48.5% patients (201) had nervous system symptoms: The most common neurological symptoms at onset of illness including myalgia were 28% (116) shown in Bar Chart 2, headache was 10.4% (43), dizziness was 5.8% (24), anosmia was 2.9% (12) and dysgeusia were 1.4% (6) as shown in Bar Chart 1. In patients with skeletal muscle injury the most common reported symptoms were 28% myalgia. 29% (120) patients admitted in MICU were intubated and ventilated while 16.2% (67) patients had noninvasive ventilation. 8.5% (35) patients died in MICU with covid complication and septic shocks. 66.7% (276) patients received steroids, 21.5% (89) took convalescent plasma. Multiorgan systems involvement of covid-19 patients (shown in Bar Chart 6, Bar Chart 7, Bar Chart 8 and Bar Chart 9) including 47.8% (198) had skeletal muscle injury (serum creatine kinase > 300U/L or myoglobin > 72 ng/ml), 15.7% (65) had rhabdomyolysis (serum creatine kinase > 1000 U/L), 36.7% (152) had acute kidney injury, 27.5% (114) had acute liver injury (> 200 ALT or AST levels), 23.9% (99) acute myocardial injury (high tropThs, male > 30, female > 20 ng/l), 11.4% (47) had acute heart failure (high proBNP > 1800 pg/ml), 11.1% (46) had acute pancreatitis (serum lipase > 180 u/l), 5.8% (24) had previous uncontrolled blood sugar (with HbA1c > 12) and 3.1% (13) had post ICU critical illness neuromyopathy supported by clinical and NCS/EMG study. 11% (46) had AKI associated with rhabdomyolysis, 9.9% (41) had cytokine storm syndrome associated with rhabdomyolysis, 9.2% (38) had acute liver injury associated with rhabdomyolysis, 6.7% (28) had acute myocardial injury associated with rhabdomyolysis. Table 4 showed the laboratory, Echocardiography and Nerve conduction study/Electromyographic findings. Laboratory features associated with severe COVID-19 including 39.4% (163) had > 300 u/L serum creatine kinase (Bar Chart 3), 35.7% (148) had serum myoglobin > 72 ng/ml (Bar Chart 4), 15.7% (65) had rhabdomyolysis (serum creatine kinase > 1000 U/L) (Bar Chart 5), 71% (295) had > 500 mcg/L ferritin level, 66.2% (274) had LDH > 245 units/L, 61% (253) had > 100 mg/l C-reactive protein levels, 53.9% (223) had > 1000 ng/mL D-dimer, 37% (153) had > 70 pg/m of IL-6, 22.2% (90) had > 2 ng/ml procalcitonin, 6.3% (26) had > 4 mmol/l lactic acid. 7.7% (32) patients had echo showed 3.4% (14) were mild low EF, 1.7% (7) were severe low EF, 0.5% (2) were moderate low EF. 2.7% (11) patients had NCS/EMG studies showed 1.7% (7) patients were CINM and 0.7% (3) patients were CIM.
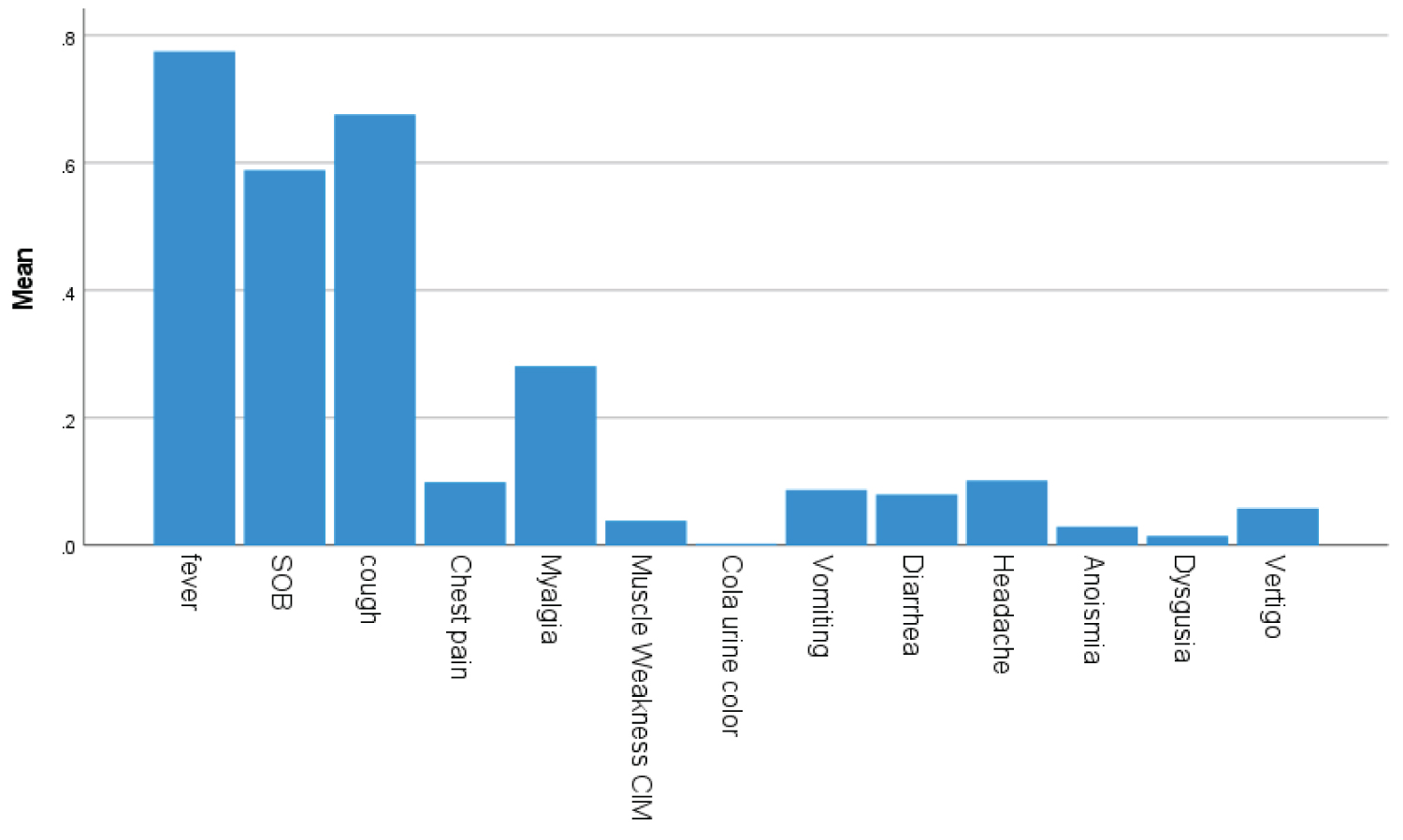 Bar Chart 1: Bars showed frequency of Covid and neurological symptoms of fever, cough, SOB, myalgia and headache with descending frequencies.
View Bar Chart 1
Bar Chart 1: Bars showed frequency of Covid and neurological symptoms of fever, cough, SOB, myalgia and headache with descending frequencies.
View Bar Chart 1
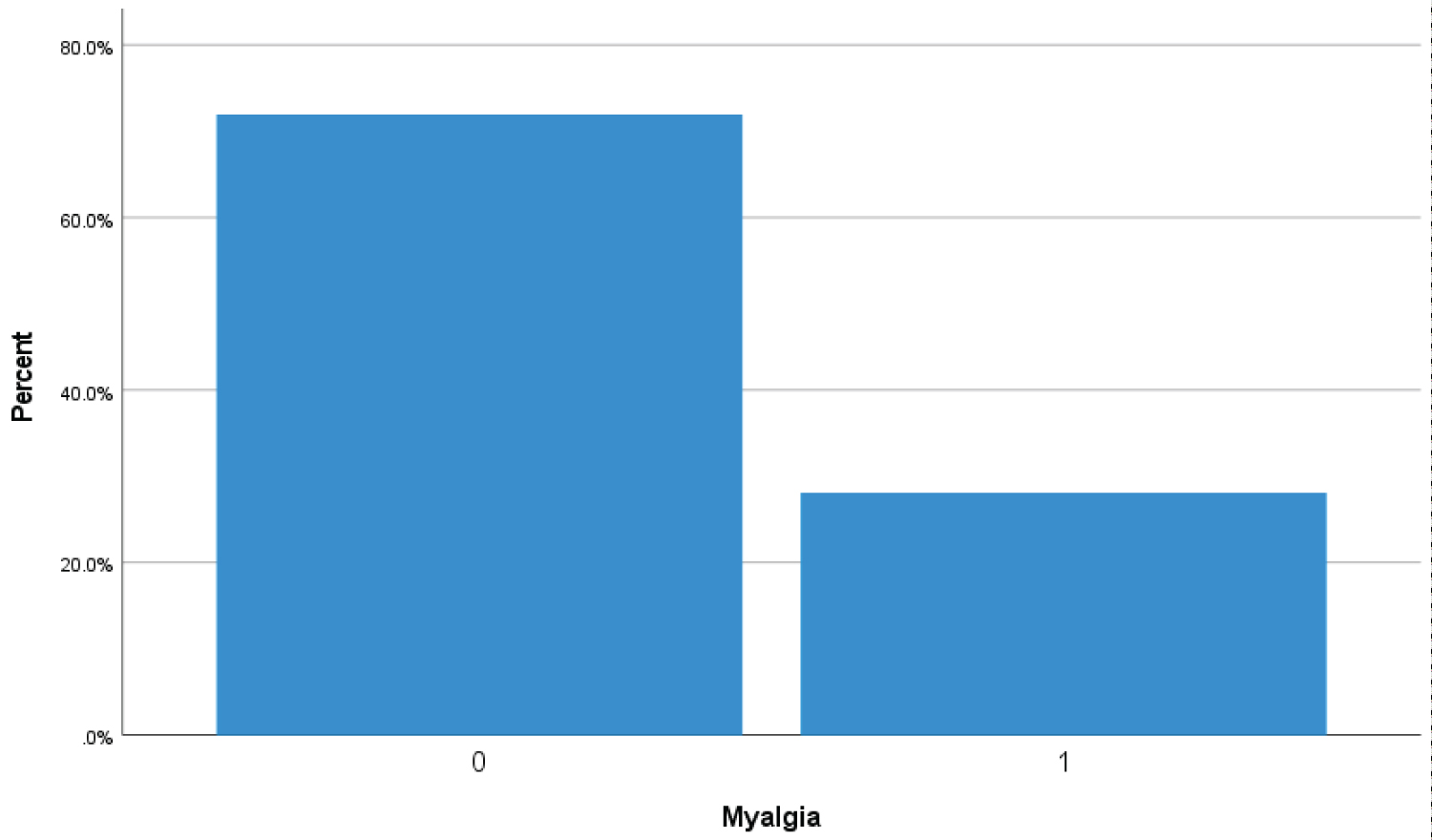 Bar Chart 2: Bar chart showed 28% (116 covid-19 patients) presenting with symptom of myalgia (1 represents myalgia symptoms while zero represents no myalgia). View Bar Chart 2
Bar Chart 2: Bar chart showed 28% (116 covid-19 patients) presenting with symptom of myalgia (1 represents myalgia symptoms while zero represents no myalgia). View Bar Chart 2
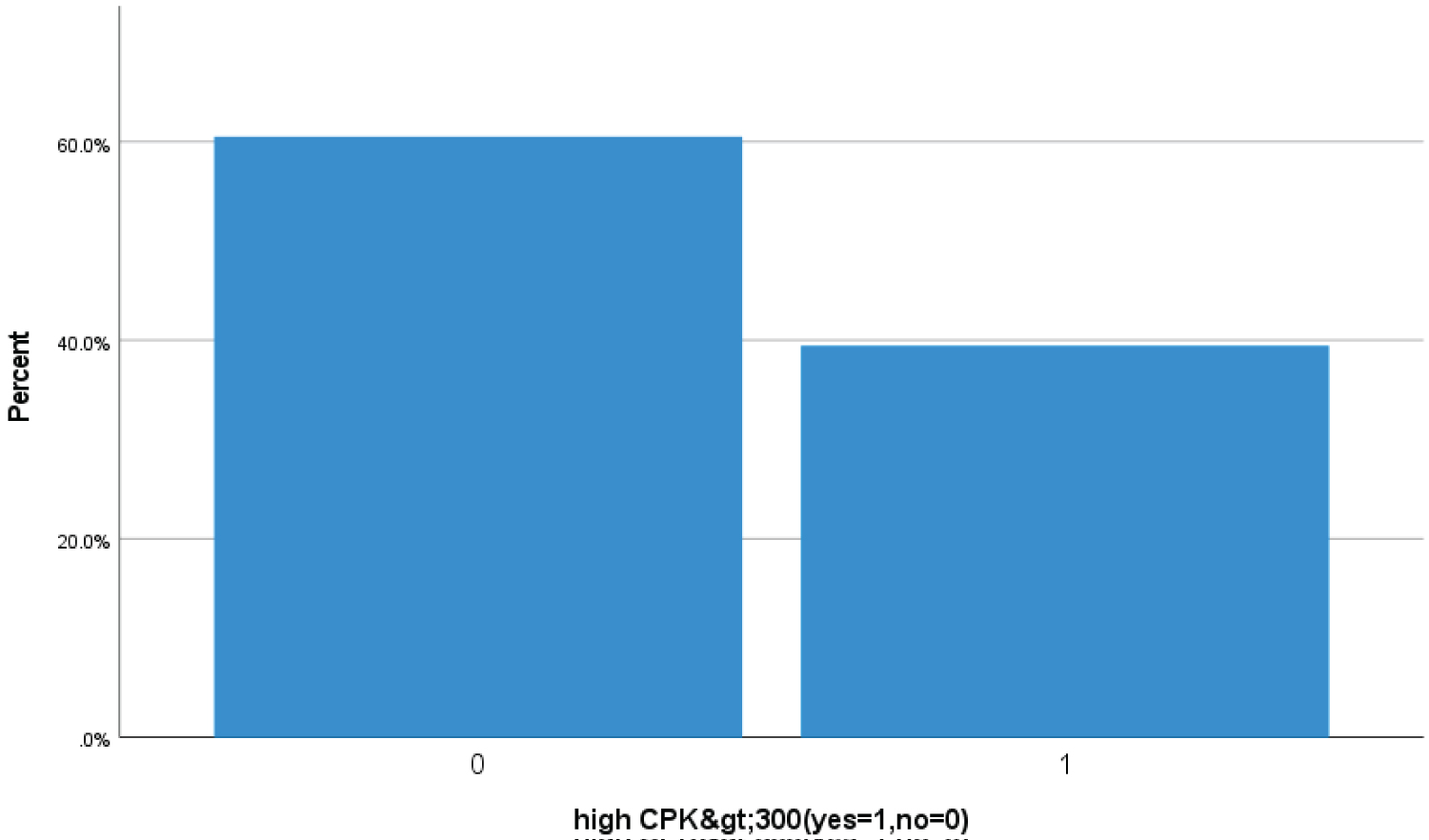 Bar Chart 3: Barchart showed 39.4% (163 Covid-19 patients) had high > 300 u/L serum creatine kinase (CPK) suggestive of severe Covid skeletal muscles injury (1 represents high > 300 u/L CPK level zero represents less then < 300 u/L).
View Bar Chart 3
Bar Chart 3: Barchart showed 39.4% (163 Covid-19 patients) had high > 300 u/L serum creatine kinase (CPK) suggestive of severe Covid skeletal muscles injury (1 represents high > 300 u/L CPK level zero represents less then < 300 u/L).
View Bar Chart 3
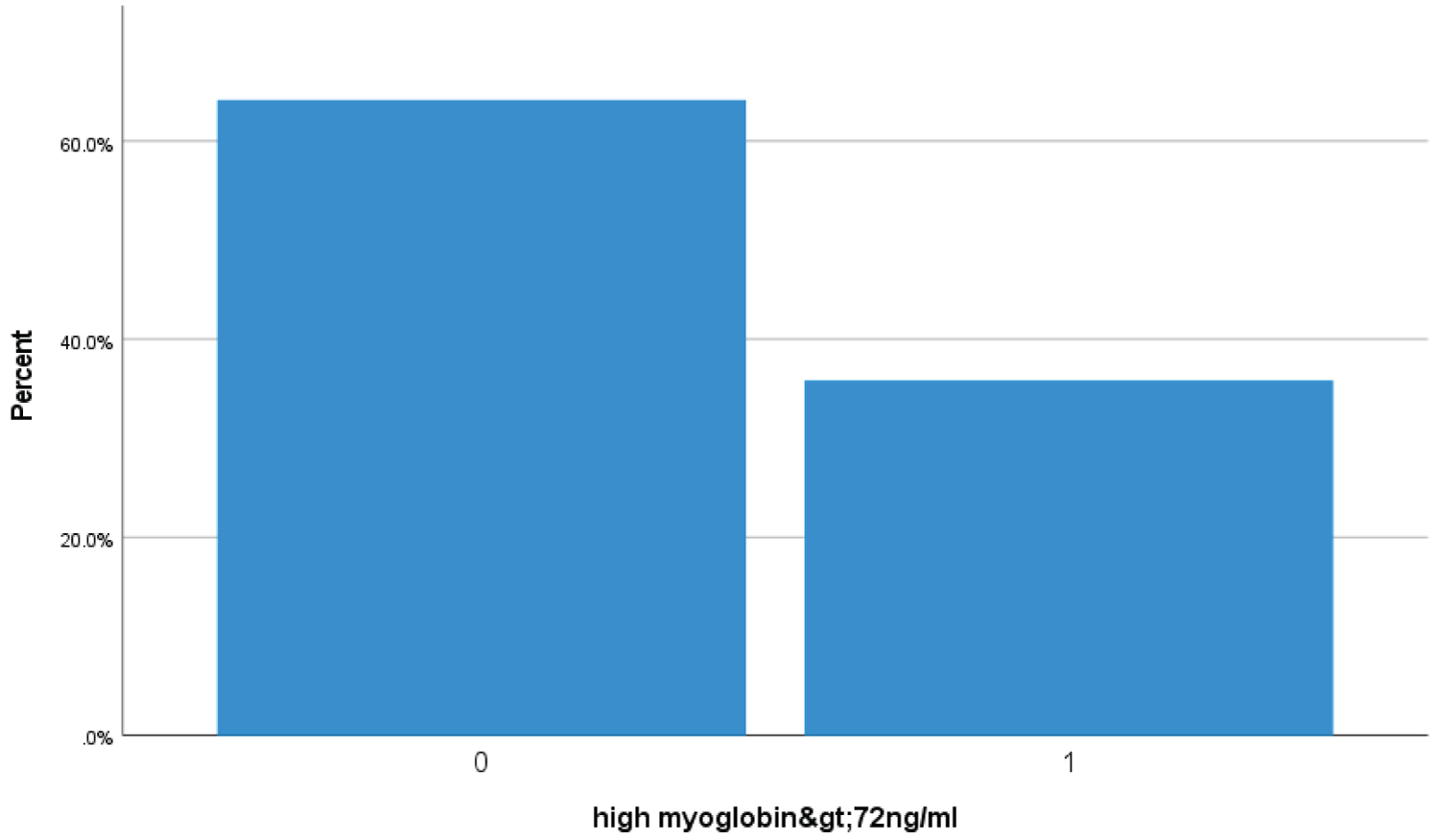 Bar Chart 4: Bar chart showed 35.7% (148 Covid-19 patients) had high serum myoglobin > 72 ng/ml suggestive of severe Covid skeletal muscles injury (1 represents high > 72 ng/ml myoglobin level zero represents less then < 72 ng/ml).
View Bar Chart 4
Bar Chart 4: Bar chart showed 35.7% (148 Covid-19 patients) had high serum myoglobin > 72 ng/ml suggestive of severe Covid skeletal muscles injury (1 represents high > 72 ng/ml myoglobin level zero represents less then < 72 ng/ml).
View Bar Chart 4
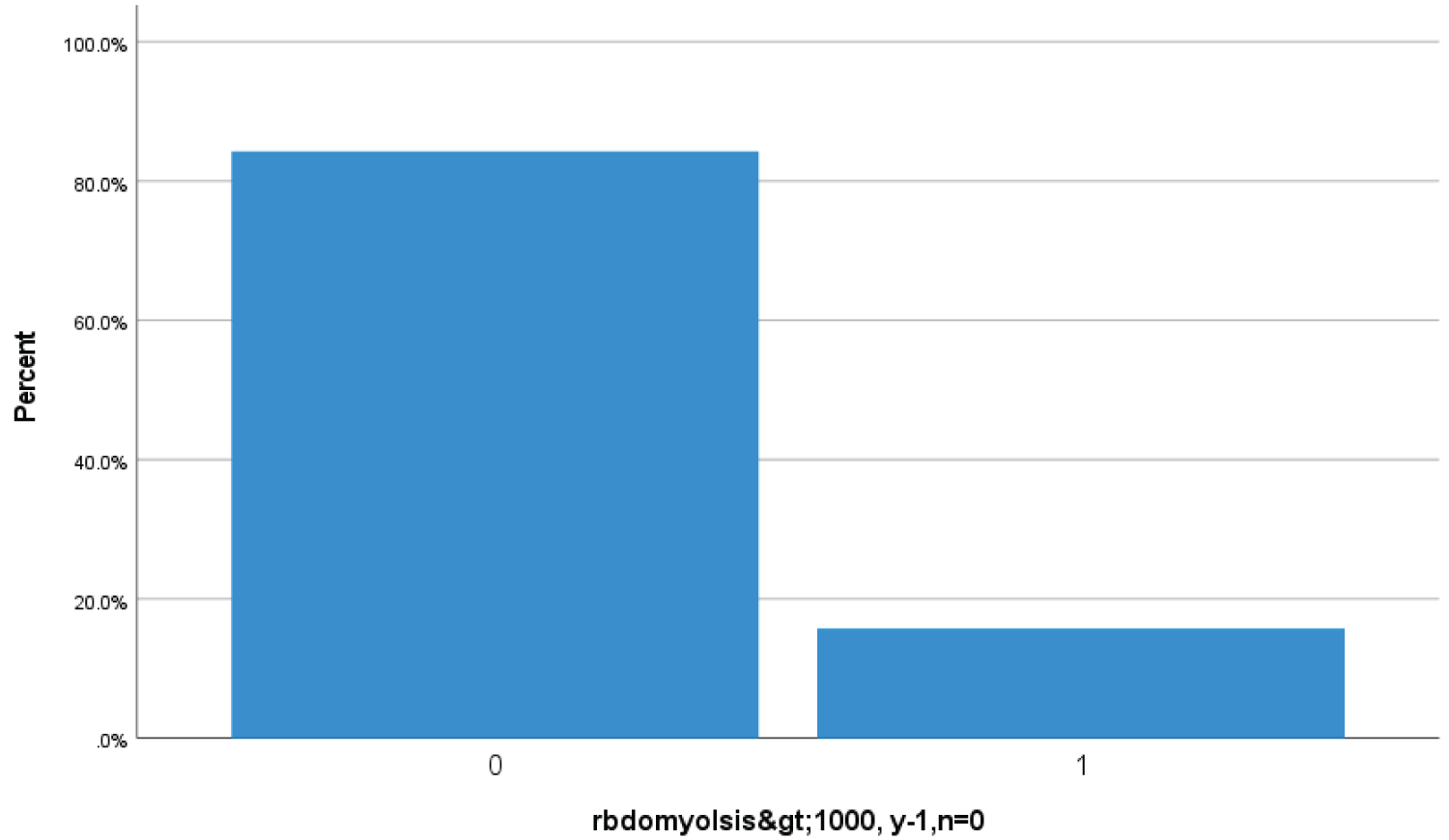 Bar Chart 5: Bar chart showed 15.7% (65 Covid-19 patients) had high serum creatine kinase > 1000 u/L suggestive of rhabdomyolysis (1 represents > 1000 u/L serum creatine kinase while zero represents less then < 1000 u/L).
View Bar Chart 5
Bar Chart 5: Bar chart showed 15.7% (65 Covid-19 patients) had high serum creatine kinase > 1000 u/L suggestive of rhabdomyolysis (1 represents > 1000 u/L serum creatine kinase while zero represents less then < 1000 u/L).
View Bar Chart 5
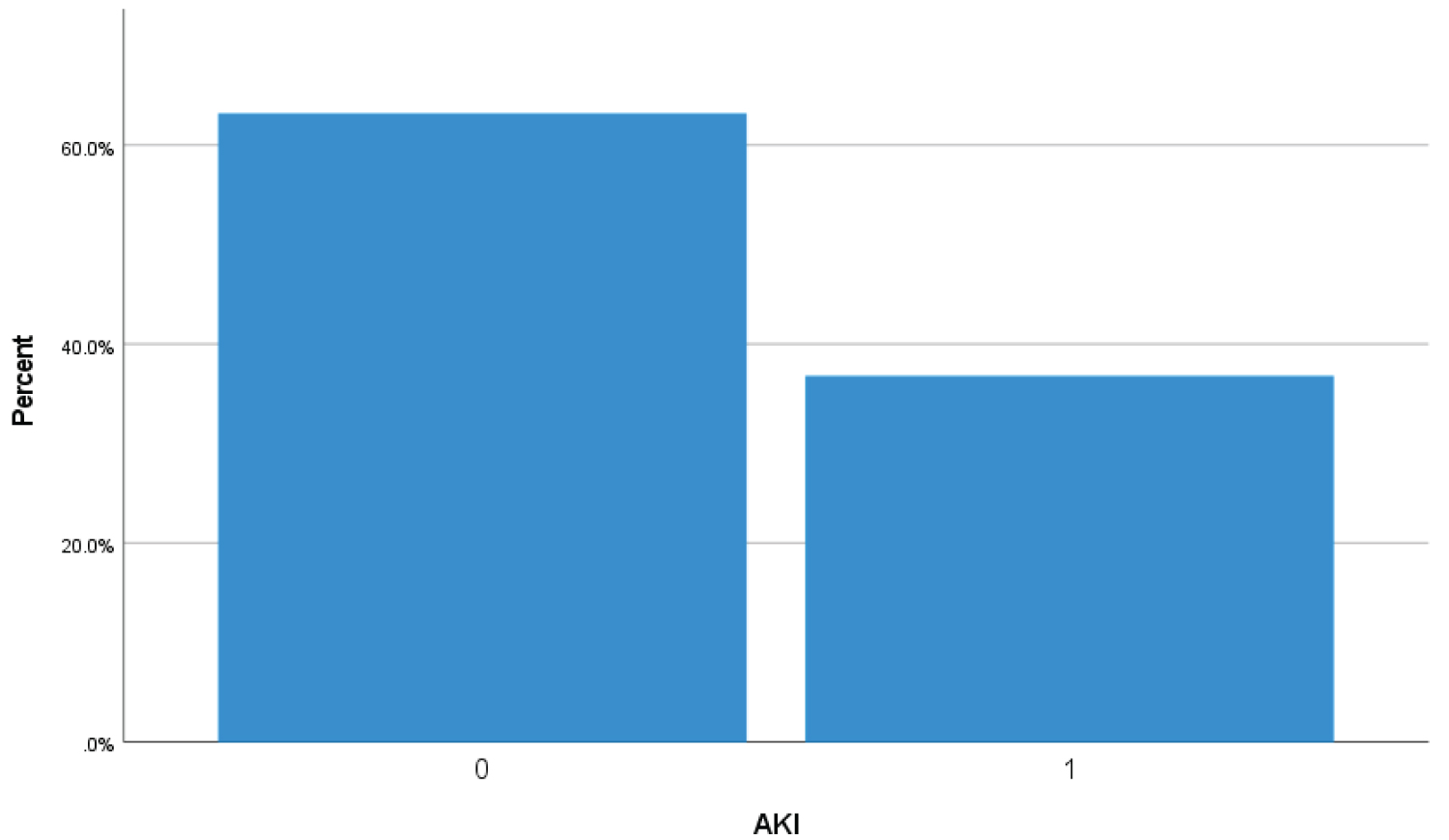 Bar Chart 6: Bar chart showed 36.7% (152 Covid-19 patients) higher urea > 8 mmol/L and creatinine > 106 umol/L on admission or during hospital stay suggestive of acute kidney injury (1 represents acute kidney injury while zero represents no acute kidney injury).
View Bar Chart 6
Bar Chart 6: Bar chart showed 36.7% (152 Covid-19 patients) higher urea > 8 mmol/L and creatinine > 106 umol/L on admission or during hospital stay suggestive of acute kidney injury (1 represents acute kidney injury while zero represents no acute kidney injury).
View Bar Chart 6
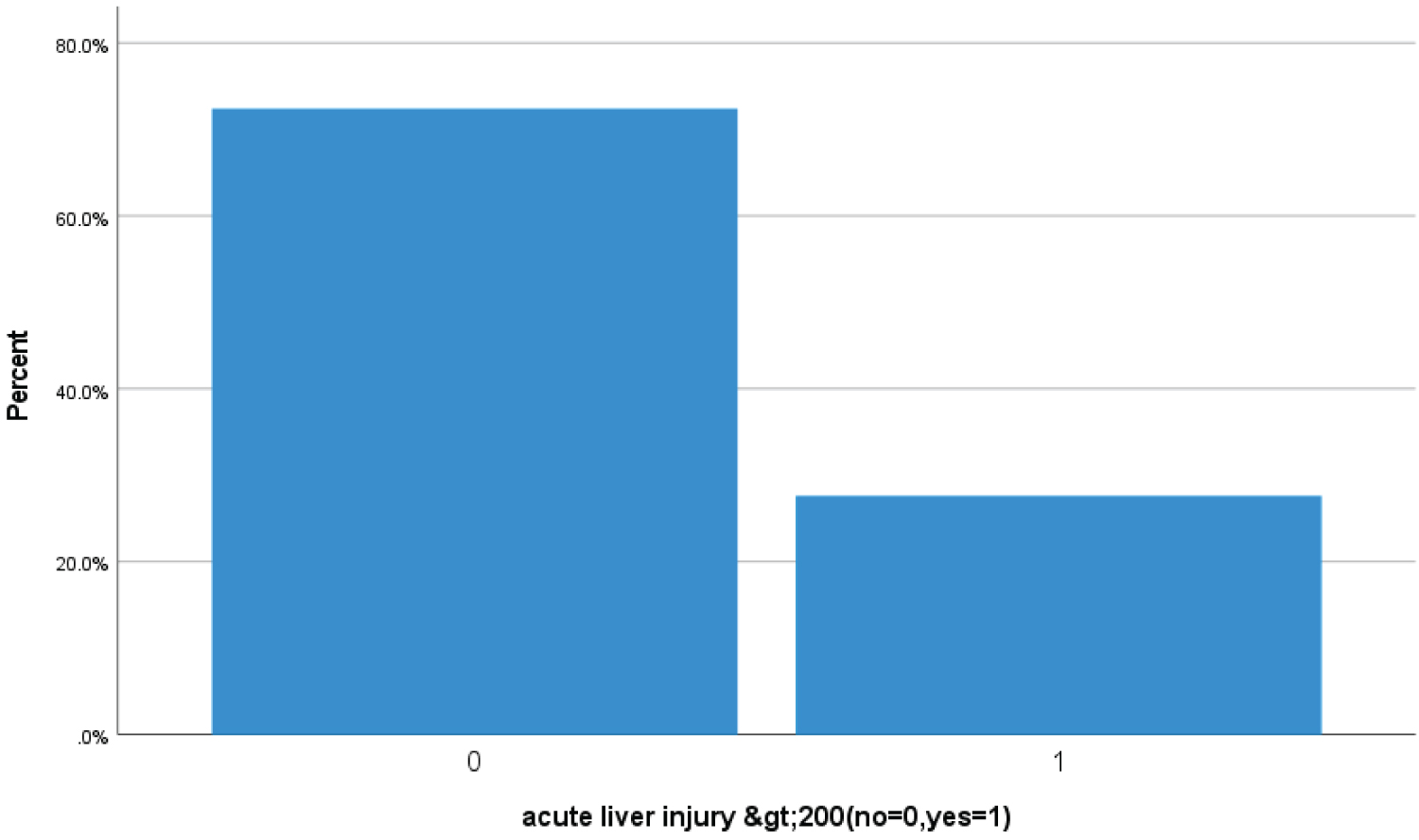 Bar Chart 7: Bar chart showed 27.5% (114 Covid-19 patients) had > 200 ALT or AST levels suggestive of acute liver injury (1 represents acute liver injury while zero represents no acute liver injury).
View Bar Chart 7
Bar Chart 7: Bar chart showed 27.5% (114 Covid-19 patients) had > 200 ALT or AST levels suggestive of acute liver injury (1 represents acute liver injury while zero represents no acute liver injury).
View Bar Chart 7
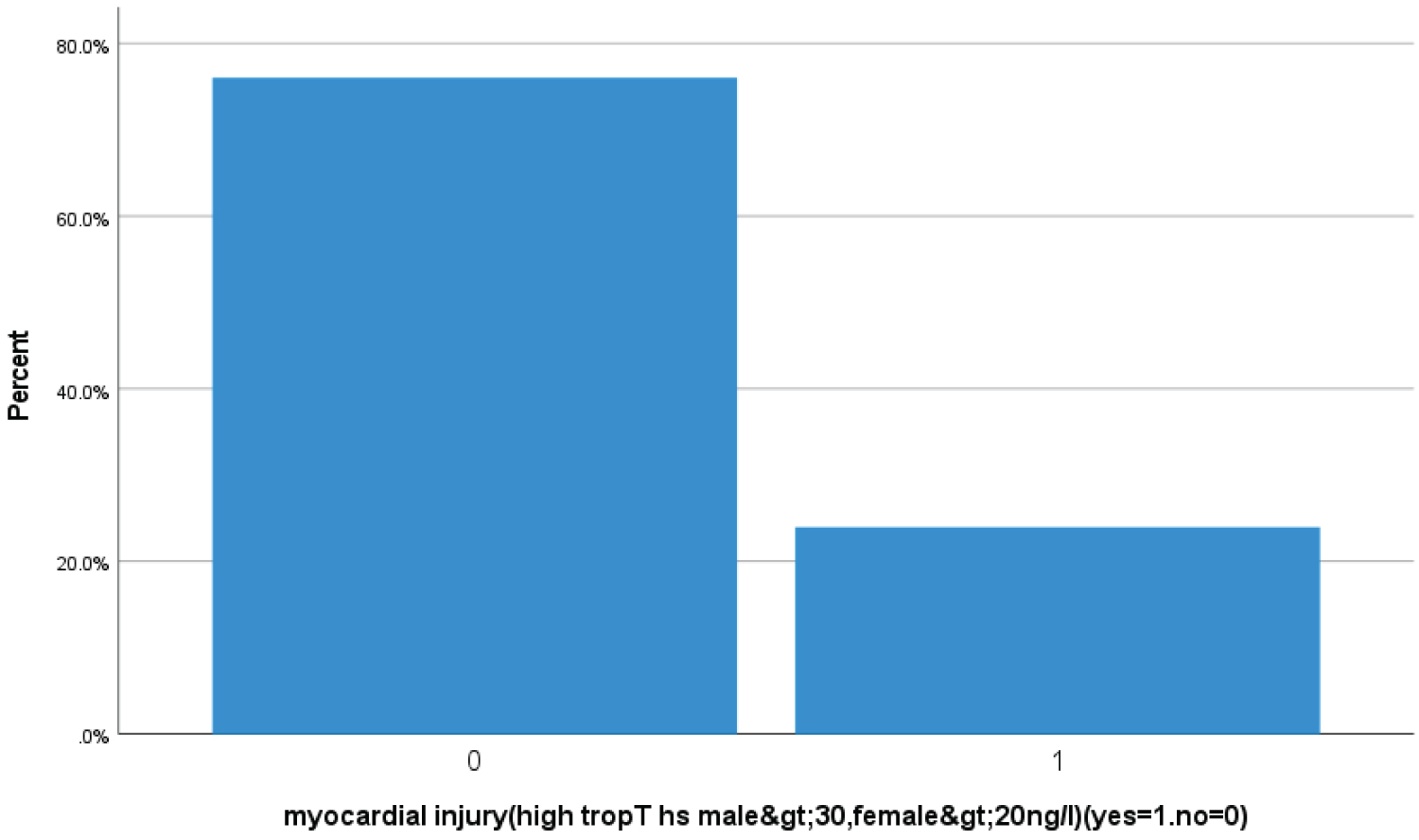 Bar chart 8: Bar chart showed 23.9% (99 Covid-19 patients) had high trop T hs, for male > 30, for female > 20 ng/l suggestive of acute myocardial injury (1 represents acute myocardial injury zero represents no acute myocardial injury).
View Bar Chart 8
Bar chart 8: Bar chart showed 23.9% (99 Covid-19 patients) had high trop T hs, for male > 30, for female > 20 ng/l suggestive of acute myocardial injury (1 represents acute myocardial injury zero represents no acute myocardial injury).
View Bar Chart 8
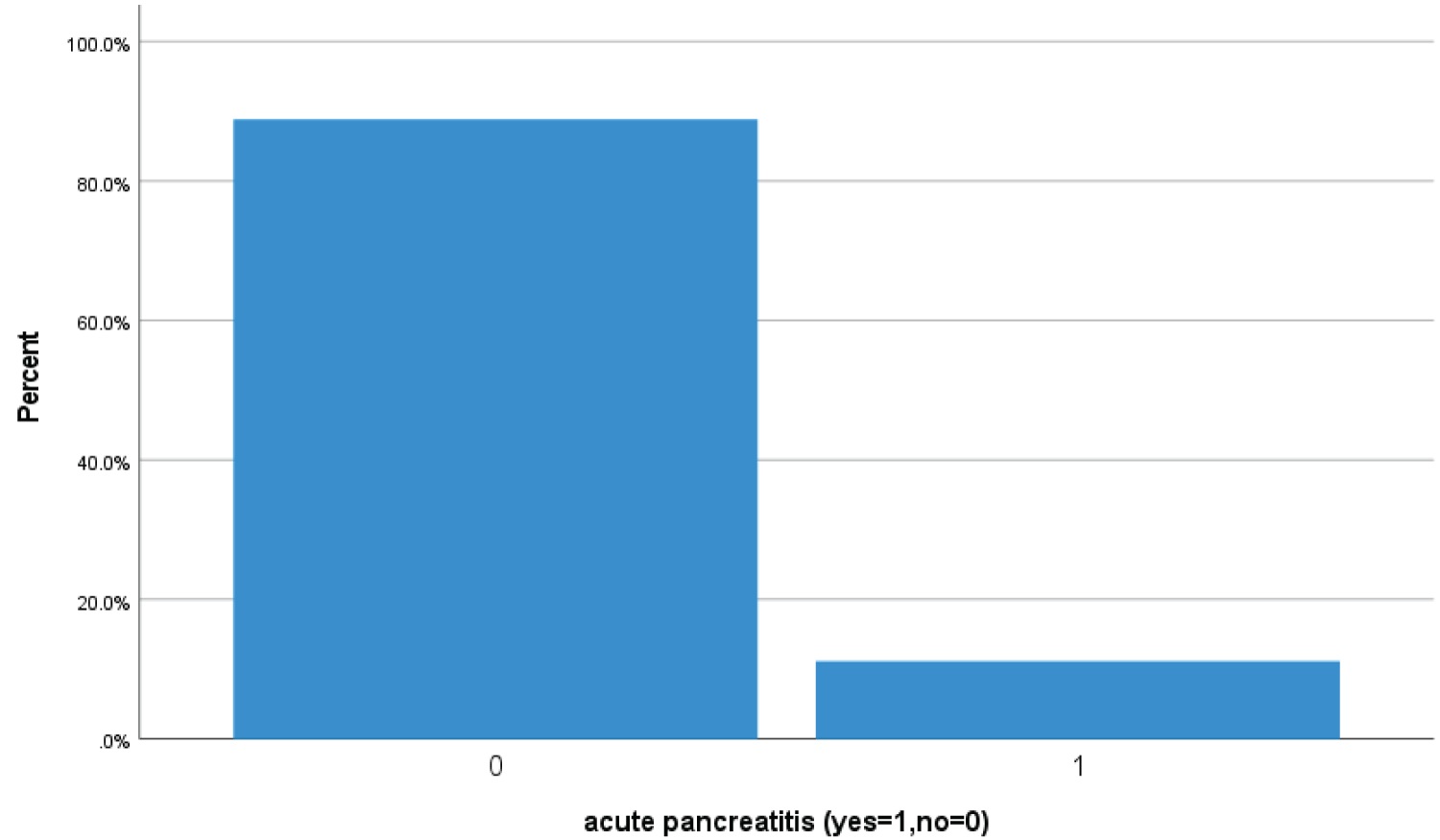 Bar Chart 9: Bar chart showed 11.1% (46 Covid-19 patients) had high serum lipase > 180 u/l suggestive of acute pancreatitis (1 represents acute pancreatitis while zero represents no acute pancreatitis acute pancreatitis).
View Bar Chart 9
Bar Chart 9: Bar chart showed 11.1% (46 Covid-19 patients) had high serum lipase > 180 u/l suggestive of acute pancreatitis (1 represents acute pancreatitis while zero represents no acute pancreatitis acute pancreatitis).
View Bar Chart 9
Table 1: Demographics. View Table 1
Table 2: Clinical manifestations. View Table 2
Table 3: Underlying risk factors. View Table 3
Table 4: Laboratory, Echocardiography and Nerve conduction study/Electromyographic findings. View Table 4
This is the first research article on skeletal muscles injury and rhabdomyolysis of the COVID-19 hospitalized patients from Qatar. From January, 1, 2020 to 31, December, 2020, total 413 patients were included in this study.
Majority of patient were less than 55 years age (57%) with different nationality. The most common risk factors were found Hypertension, Diabetics and Obesity. In total of 413 patients, 48.5% (201) were diverse neurological symptoms that involved central, peripheral nervous system and skeletal muscles. Most common neurological symptoms including myalgia, headache and dizziness are in order of descending frequency. The most common neurological involvement was skeletal muscle injury (47.8%). In addition to respiratory system, other extrapulmonary multiorgan systems involvement of covid patients including skeletal muscle injury (47.8%), acute kidney injury (36.7%), acute liver injury (27.5%), acute myocardial injury (23.9%), rhabdomyolysis (15.7%), acute heart failure (11.4%), acute pancreatitis (11.1%) and post ICU critical illness neuromyopathy (3.1%) that supported by clinical and NCS/EMG study. Multiorgan system involvements association with rhabdomyolysis including AKI, cytokine storm syndrome, acute liver injury and acute myocardial injury are in order of descending frequency. Exact mechanism of skeletal muscle injury in covid 19 patients is not fully recognize. Direct SARS-CoV-2 viral invasion of skeletal muscle including diaphragm, hematogenous dissemination and immune-mediated skeletal muscles injury secondary to activation of immune cells and inflammatory response with cytokine storm syndrome may lead skeletal muscle injury including muscle fiber proteolysis and fibrosis. Severe COVID-19 infection in patients with muscular dystrophic like Duchenne muscular dystrophyare at higher risk of respiratory failure due to skeletal and diaphragm muscle injury. In COVID-19 there has been case reports of rhabdomyolysis that may lead to acute renal failure, compartment syndrome and disseminated intravascular coagulation (DIC).
All patient's electronic data were retrieved retrospectively and mostly of patients were from dedicated Covid facility for predominantly male. During COVID-19 spike waves times, there were high rate of hospital admissions, therefore skeletal muscles symptoms were subjective and neuroimaging like CT scan or MRI or Neurodiagnostic procedures like EMG/NCS were limited to decrease the chance of acquire COVID-19 infection.
In conclusion, the most common neuromuscular manifestation of COVID-19 were skeletal muscle injury and rhabdomyolysis. In critical COVID-19 infection other multiorgan dysfunctions including renal, hepatobiliary, cardiac, cerebrovascular accident (CVA) and venous thromboembolism. Patients with severe Covid-19 infection with concomitant muscular dystrophic are at higher risk for skeletal muscle injury and rhabdomyolysis including diaphragmic injury and respiratory failure. Skeletal muscles injury with SARS-CoV-2 infection manifest as myalgia, generalized fatigue, myositis, rhabdomyolysis, dermatomyositis-like interferonopathy 1, immune-mediated necrotizing myopathy and diaphragmic or respiratory failure. Thus, we postulated that skeletal muscles injury may be peculiarly affected by SARS-CoV-2 via direct viral invasion through ACE2 receptor, immune-mediated and proinflammatory signaling molecules of cytokines and hematogenous dissemination. Further studies will berequired with focus on short- and long-term outcomes of musculoskeletal and neuromuscular health of patients improving from critical covid illness and benefits of using post Covid rehabilitation programs.
Mary Metreenj, Ansaf Ahmed, Jessy Jacob, Lucy Karugu, Neelima John, KaziMuntaha
Nothing to disclose or no funding for this study.
There is no conflict of interest.
Liaquat Ali: Data collection, data analysis, manuscript writing & review literature; Adnan Khan: Manuscript writing, manuscript review; Mohammad Alhatou: Manuscript writing & review literature; Osama Elalamy: data analysis, manuscript review; Beatriz Canibano: Data analysis, manuscript writing & review; Golam Adeli: Data analysis, manuscript writing & review; Ahmed Mohammad: Data collection, data analysis; Khawaja Haroon: manuscript writing, manuscript review; Suhail Hussain: Data analysis, manuscript writing; Suha Elmaki: Data analysis, manuscript writing; Yahya Imam: Data collection, data analysis; Ambreen Iqrar: Manuscript writing & review literature; Mirza Baig: Data collection.
The authors declare that data supporting the findings of this study are available within the article.
Entitled "Incidence of skeletal muscle injury with SARS-CoV-2 infection in tertiary care hospitals in Qatar." has been approved by IRB.