The use of kidney allografts with multiple renal arteries (MRAs) in kidney transplantation is no longer considered a contraindication to renal transplantation. Traditional decision on best management of accessory renal vessels has been guided by surgeons' surgical experience and institutional experience. The role of inferior epigastric artery (IEA) as an inflow vessel has often been relegated as a secondary option for allografts with MRAs due to perception of greater technical challenge and worse outcomes. However, there is limited information on patient selection and description of intra-operative application of this technique. Adequate description of this technique in literature for preservation, dissection and anastomosis options have been scarcely described. The authors would like to highlight two critical factors in using the IEA as in-flow source for accessory vessels: Firstly, value of careful preservation of this vessel during initial dissection when status of deceased donor allograft vessel anatomy may be an unknown. Secondly, we highlight a reproducible and descriptive attempt at safely performing the IEA anastomosis for accessory vessels.
Inferior epigastric artery, Renal transplant, Vascular surgery
Multiple renal arteries occur due to the embryonic migration of developing metanephros from the pelvis to the abdomen in-utero, and sequential change in perfusion from the iliac arteries to the abdominal aorta. An inferior polar accessory artery can be present in up to 35% of cases [1]. Most commonly, these are inferior polar vessels, and diameters can vary. As renal arteries are physiological end-arteries, anastomosis of these additional vessels is critical. Most importantly, it is also important to reduce potential morbidity associated with ureteric ischemia [2] due to the absence of perfusion from an accessory renal artery supplying the renal pelvis and proximal ureter. Accessory vessels greater than 2 mm in diameter are generally considered for preservation to reduce ischemic complications [3].
The use of kidney allografts with multiple renal arteries (MRAs) was traditionally seen as a relative contra-indication in kidney transplantation. For living donation, the contralateral (right) kidney may still be favoured in place of (left) kidney with MRAs. For deceased donor allografts, the finding of an accessory polar vessel may have led to non-utilisation of the precious allograft in the past. Most of the reluctance stemmed from perception that there was increased complexity of vascular anastomosis, placing the recipient at higher risk of complications. In-fact a recent meta-analysis has suggested that utilisation of deceased allografts with MRAs may be associated with delayed graft function [4]. This is in contradiction to a separate multi-institution paper looking at live donation, which had shown no difference in use of ‘complex' kidney allografts in terms of one-year graft survival [5].
Indeed, the history of vascular anastomosis techniques in renal transplantation has evolved over time. Surgeons' and institution's surgical experience, has largely guided decisions on how best to incorporate main and accessory vessels. Traditionally, the anterior division of the internal iliac artery was utilised as an end-to-end anastomosis to the main renal artery (RA). However, overtime, the utilisation of an end to side anastomosis of RA to external iliac artery became mainstream [6]. With this, several variations in managing the accessory polar vessel were developed including [6,7] (Figure 1):
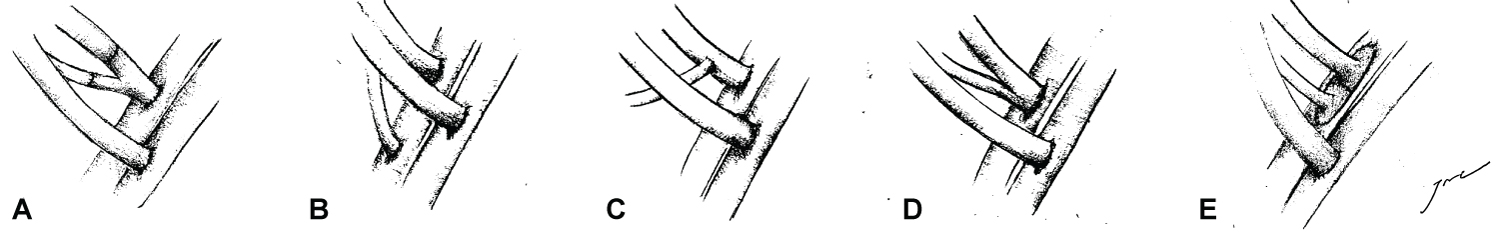 Figure 1: (A) End to side anastomosis to EIA utilising interposition Y-graft; (B) End to side anastamosis to EIA inferior to MRA anastamosis; (C) End to side anastamosis to MRA, with subsequent MRA-EIA anastomosis; (D) Side to side anastamosis to MRA, with subsequent end to side anastamosis to EIA; (E) End to side anastamosis to EIA utilising carrell patch.
View Figure 1
Figure 1: (A) End to side anastomosis to EIA utilising interposition Y-graft; (B) End to side anastamosis to EIA inferior to MRA anastamosis; (C) End to side anastamosis to MRA, with subsequent MRA-EIA anastomosis; (D) Side to side anastamosis to MRA, with subsequent end to side anastamosis to EIA; (E) End to side anastamosis to EIA utilising carrell patch.
View Figure 1
• Back-table anastomosis to main renal artery (RA): Side to side with RA, end to side to RA. Subsequent RA end to side onto external iliac artery
• Direct implantation above or below main renal artery onto external with end to side
• Ex-situ back-table vascular reconstruction of diseased vessels including: Renal orifice endarterectomy, reconstruction of aorta to maintain carrel patch with RA and accessory vessel; and various donor interposition grafting of accessory vessel.
The inferior epigastric artery as a source of in-flow for accessory vessels (upper or lower pole) was first described several decades ago [8]. The IEA is branch arising from medial aspect of the external iliac artery, ascending just above the inguinal ligament. It ascends along the medial border of the deep inguinal ring, punctures the transversalis fascia and ascends to enter the rectus sheath just beneath the arcuate line. It anastomoses with the superior epigastric artery in the substance of the rectus muscle above the umbilicus.
Utilisation of the IEA for anastomosis for accessory vessel has several potential advantages. The utilisation of IEA to independently anastomose the accessory vessel avoids additional manipulation of the main renal artery. This may avoid the complications associated with main renal artery thrombosis, dissection or bleeding which can have catastrophic complications. Furthermore, the utilisation of IEA will minimise the size mismatch with inflow vessel for the polar vessel. In particular, it offers the ability to anastomose small accessory vessels (2-3 mm), which may otherwise be inadvertently sacrificed. There are several single-institution publications supporting the utilisation of IEA anastomosis over other reconstructions for accessory vessels in live donors. In particular, there is association with reduced acute tubular necrosis, delayed graft function (DGF) and even lower rates of rejection [3,9-12]. The mechanism remains unclear however, it may relate to sequential anastomosis reducing cold/warm ischemia to allograft and ischemic preconditioning. Despite the small numbers overall, unpublished data of a systematic review of all studies comparing IEA anastomosis to other techniques, supported a trend for reduced DGF [13].
Whilst most of the literature relates to live donor cases, we believe it has strong value in deceased allograft donation. In particular, it is not infrequent to encounter accessory vessels in allografts, which may be missed during retrieval. By allowing for preservation of the IEA during initial dissection, the option of IEA anastomosis is preserved. By following the IEA anastomosis technique highlighted, we believe, this will allow for safe and reproducible accessory vessel implantation whilst potentially reducing DGF rates in allograft.
Dissection: The patient is positioned in supine. Standard Rutherford-Morrison incision is performed to gain access to the retroperitoneum. Following division of external oblique fascia, the lateral border of the rectus muscle is identified. Babcock's are used to lift the lateral border of the rectus muscle until the inferior epigastric vascular bundle is visualised.
The inferior epigastric artery, vein and branches/tributaries of both perpendicular to IEA course are carefully dissection and ligated as shown. It is essential this performed in a controlled fashion to avoid avulsion of branches. In particular, paying close attention to branches proximally, near IEA origin is vital. The IEA should be followed as the IEA enters the substance of the rectus muscle. The IEA vessel is marked to ensure orientation and then can be divided to provide good length of IEA.
Anastomosis (Figure 2): Sequential anastomosis following the main renal artery reperfusion is performed. This often reveals small area of ongoing ischemia in region supplied with polar vessel. Often evidence of back bleeding from accessory vessel is noted.
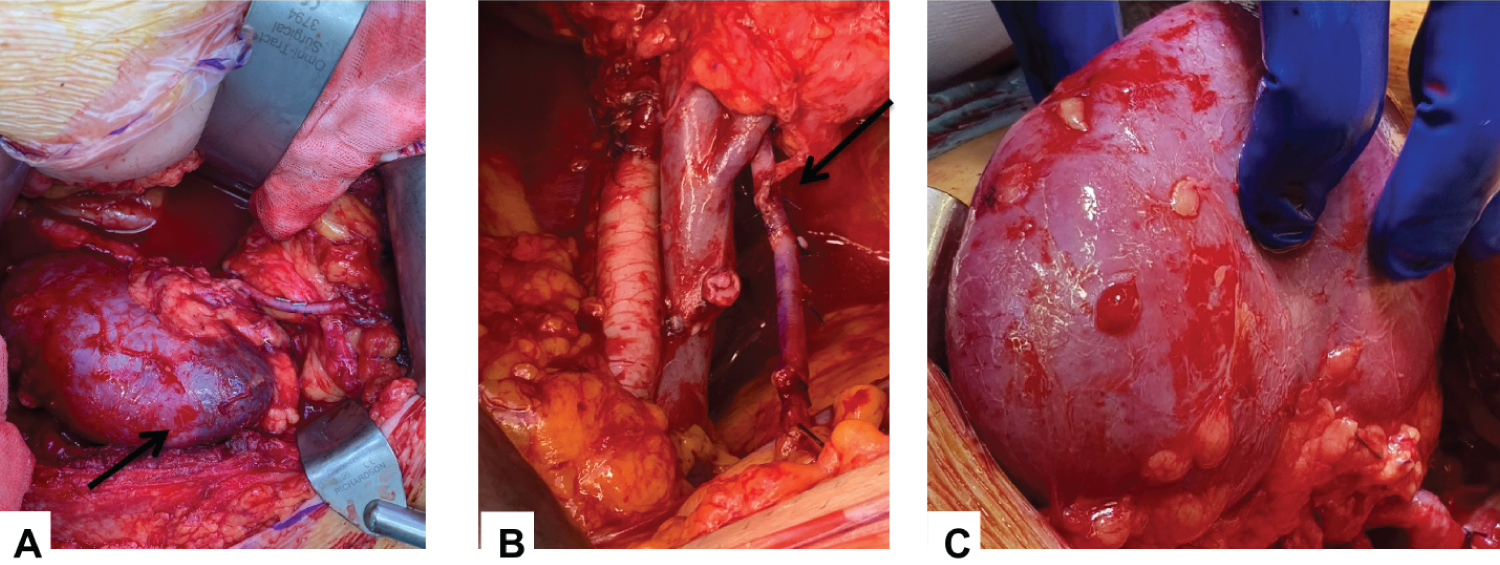 Figure 2: (A) Intra-operative appearance of dusky lower pole before accessory vessel anastamosis but after MRA reperfusion; (B) EIA anastamosis; (C) Post accessory vessel reperfusion and uniform appearance of cortex.
View Figure 2
Figure 2: (A) Intra-operative appearance of dusky lower pole before accessory vessel anastamosis but after MRA reperfusion; (B) EIA anastamosis; (C) Post accessory vessel reperfusion and uniform appearance of cortex.
View Figure 2
After checking length of IEA, a small cannula (22G) with 10 ml syringe of heparinised saline can be utilised to confirm orientation and gently hydro-distend the IEA. A mini-bulldog clamp is placed near IEA take-off.
The IEA is cut to appropriate length to avoid kinking. Small degree of spatulation to the IEA and/or accessory polar vessel can be performed. Interrupted in-to-out stitches are placed with double armed 7-0 prolene stitches at 0 and 6 o'clock. Using these as orientation stitches, further 1-3 interrupted stitches are placed inside out on donor vessel and IEA.
The sutures are sequentially tied down and the clamps released prior to tying the final sutures to incorporate growth factor of vessel. Intra-operative ultrasound can support visual inspection of vessel patency and anastomosis adequacy.
Limitations
Patient selection: Several vessel, recipient and implantation factors play a role in selecting the IEA as an appropriate inflow target. Recipient factors which may make IEA less than ideal inflow target include: Older age, history of diabetes, history of ipsilateral pre-existing vascular disease near iliac/femoral region. In addition, intra-operative evidence of heavy calcification, diminutive inferior epigastric size or in-sufficient IEA length may preclude it's use. Finally, ensuring patency of accessory vessel on back-table is crucial to assess target for IEA anastomosis. Use of a fine cannula (22G) with 10 ml syringe of cold perfusate can be used to flush the accessory vessel and confirm washout of any residual blood. The utilisation of machine perfusion technology for accessory vessel allografts has limited evidence in literature but may be possible by antegrade or retrograde techniques [14].
Surveillance: Most institutions perform ultrasound within 24 hours and first few days post-operatively. The identification of polar vessel and anastomosis via a trans-abdominal window can be hampered by presence of large abdominal pannus, bowel gas and post-operative fluid status. The indirect observation of good perfusion to the inferior pole and presence or absence of urological complications has previously been reported in literature as surrogate markers [2]. Nuclear medicine imaging and magnetic resonance imaging (with time of flight) may provide alternative avenues to assess perfusion via IEA anastomosis in medium to long-term [15]. However, no long-term data exists comparing different surveillance techniques and outcomes.
The utilisation of an end-to-end anastomosis of the accessory polar vessel to the inferior epigastric artery is a safe, replicable and reliable technique in managing allografts with MRAs (particularly those 2-3 mm). Due to limited long-term literature evidence, it is likely that the surgeons' clinical experience will remain a key factor in appropriate selection and management of MRAs. However, IEA anastomosis, with appropriate technique and adjuncts, should no longer be considered as secondary choice in managing MRAs. In particular, ensuring careful IEA dissection during renal transplant exposure will preserve this option until back-table preparation of deceased allografts has been performed. Intra-operative ultrasound may be of value in ensuring quality control and developing surgeon's confidence in this vascular technique.
This has not been previously published or submitted elsewhere for publication. The first two authors contributed to write up, review of literature and editing of the paper. All other authors contributed significantly to supervision, editing and revision of the paper.
There are no conflicts of interest for any authors involved in this study.
Informed consent was obtained from patient in written and verbal.
No external funding was used in the writing of this paper.