A 31-year-old woman who carried a laparoscopic adjustable gastric banding for morbid obesity presented chronic postprandial vomiting and lack of efficacy with weight recovery three years after surgery. The adjustable gastric band was removed but symptoms did not improve. Morphologic and functional tests demonstrated a functional obstruction 5 to 6 cm below the oeso-gastric junction, probably related to a fibrous scare, which could explain symptoms. Laparoscopic procedure confirmed the diagnosis, the scare was surgically removed and food intolerance symptoms resolved rapidly.
Bariatric surgery, Laparoscopic adjustable gastric banding, Morbid obesity, Fibrous scare, Chronic vomiting
The adjustable gastric band (AGB) is a restrictive technique that creates a gastric compartment with a capacity of about 20 ml, by placing a circular device at the upper part of the stomach below oeso-gastric junction [1]. AGB used to be widely performed [2], but due to secondary effects and frequent weight recovery after removal, this technique tends to be supplanted by gastric bypass or sleeve gastrectomy [3].
In comparison to other bariatric surgical techniques, AGB has the lowest mortality and morbidity rates but some complications are common. The most frequent early complications are acute gastric occlusion, infection of the device, and gastric perforation. The most frequent late complications are: Gastric erosion (7% of cases) with a risk of AGB intragastric migration [4]; the slippage of the band (2-4% of cases) which can lead to a dilation of the gastric pouch, a volvulus and a gastric necrosis [5,6]; and oesophageal dilation, or pseudo-achalasia, (10% of cases) that can be complicated by an oesophageal atony [7,8].
In this case report we describe for the first time a case of a gastric fibrous scare which induced food intolerance with chronic vomiting, persistent after surgical removal of the AGB.
A 31-year-old female patient with morbid obesity underwent a laparoscopic gastric banding in 2013. Before surgery her weight was 105 kg for a size of 164 cm, with a BMI at 40 kg/m2. Surgery has allowed a loss of 30 kg, equivalent to 28.5% of the initial weight in 18 months.
Upper gastrointestinal (UGI) symptoms progressively appeared 3 years after surgery, with vomiting, dysphagia, associated with weight recovery to 105 kg. The AGB was thus removed in April 2017, without any preoperative explorations.
Despite the withdrawal of the AGB, the upper digestive symptomatology worsened with food intolerance, persistent vomiting and dysphagia even with a liquid diet. Routine laboratory workup and an abdominal ultrasound were normal. An eso-gastroduodenal endoscopy revealed an aspect of mega esophagus with fluid stasis. After aspiration a stenosis was revealed just below the cardia, on a height of 2.5 cm, with fragility of the mucosa. However, the endoscope easily crossed the stenosis, without any sensation of blocking or projection.
In June 2017, given the lack of improvement in the clinical condition of the patient, two months after AGB removal, the patient was referred to another secondary center for further explorations. A new morphological assessment was performed. Barium esophagram demonstrated a complete interruption of radiocontrast agent passage, with stasis beyond the gastric pouch (Figure 1), followed by vomiting. During high resolution manometry, esophageal motility was normal: Normal integrated relaxation pressure of the lower esophageal sphincter, lack of premature contraction. However, manometry demonstrated an area of hyper pressure, 5-6 cm below the lower sphincter of the esophagus (Figure 2). Eso-gastroduodenal endoscopy was repeated and demonstrates a stenosis just below the cardia (Figure 3). However, endoscopist injected contrast product through the endoscope and it was identified a stasis of the contrast product in the fundic pouch upstream of cardial stenosis. An endoscopic dilatation with an 18 mm diameter hydraulic balloon was performed during the endoscopy and improved the passage of the contrast agent. However, no clinical improvement was observed after the endoscopic dilatation.
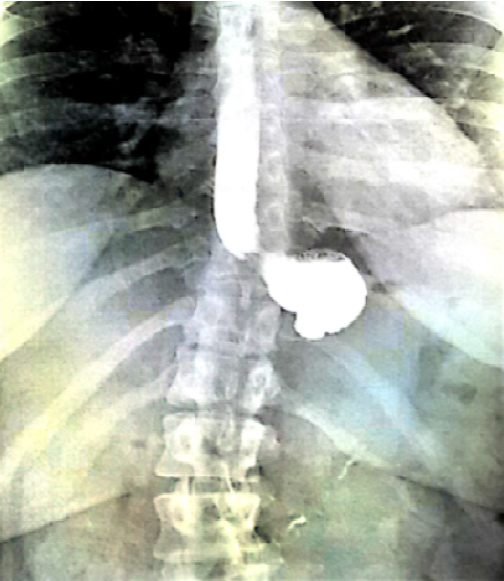 Figure 1: Esogastroduodenal transit before fibrous scare surgery.
View Figure 1
Figure 1: Esogastroduodenal transit before fibrous scare surgery.
View Figure 1
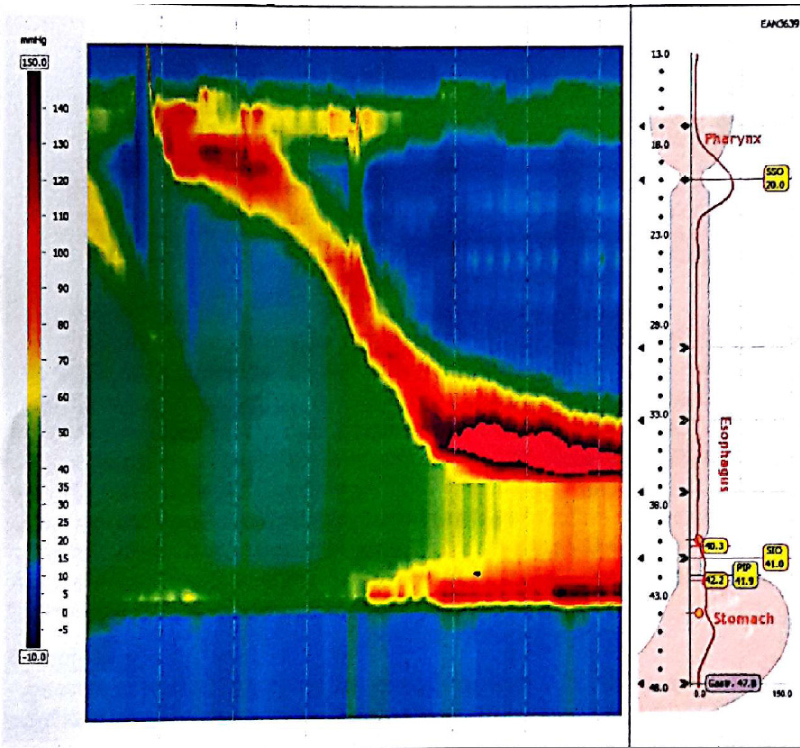 Figure 2: High resolution esophageal manometry before fibrous scare surgery.
View Figure 2
Figure 2: High resolution esophageal manometry before fibrous scare surgery.
View Figure 2
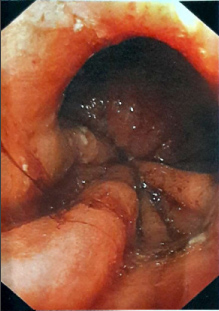 Figure 3: Esogastroduodenal endoscopy before fibrous scare surgery.
View Figure 3
Figure 3: Esogastroduodenal endoscopy before fibrous scare surgery.
View Figure 3
The patient was referred to our unit in July 2017 for further explorations and management of immediate and alimentary post-prandial vomiting. Her weight was 94 kg, with 10 kg weight-loss since the removal withdrawal of the AGB. A new esogastroduodenal transit demonstrated a stenosis (1.5 mm diameter) at the junction between the fundus and the gastric body over a length of 2.8 cm, with upstream gastric dilation.
Laparoscopy was performed and demonstrated the persistence of a very hard perigastric fibrous scare located on the initial site of the AGB. The surgeon released the anterior face of the stomach, incised and removed the scare. A 36 french Faucher tube was inserted and passed easily without residual stenosis.
The symptoms were immediately improved after surgery. The patient was able to orally feed quickly and normally under normal diet without vomiting nor dysphagia. Postoperative eso-gastroduodenal transit demonstrated a normal passage of the radiocontrast agent (Figure 4). Six months after the surgery the patient still remained asymptomatic with a normal diet.
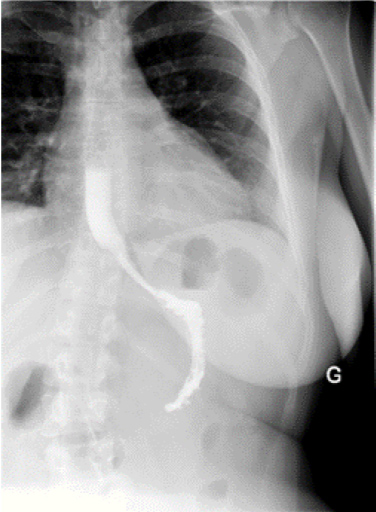 Figure 4: Postoperative esogastroduodenal transit.
View Figure 4
Figure 4: Postoperative esogastroduodenal transit.
View Figure 4
This case report describe a case of food intolerance with chronic vomiting, 3 years after a gastric band surgery, persistent after surgical removal, related to perigastric fibrous scare that was not improved to endoscopic dilation.
Fibrous scare formation has been described in the gastric wall at the level of the initial location of an AGB and is usually associated with infection, migration of the band [9] and as a risk factor of fistula after band removal and sleeve gastrectomy [10].
Vomiting is a common complication after bariatric surgery. It occurs in one third to two thirds of patients undergoing bariatric surgery [2], and is often linked to poor food hygiene with over-sized meals, ingested too quickly or without being chewed. Gastric emptying does not appear to be impaired by the AGB [11]. In case of persistent vomiting occurring several months or years after AGB a intra-gastric migration of the AGB must be ruled out by endoscopy [6]. If it is normal, a diagnosis of pseudo-achalasia, which symptoms are dysphagia, vomiting or retrosternal pain, should be suggested. Pseudo-achalasia mainly occurs when the gastric band is located next to the lower esophageal sphincter [12]. The diagnosis is made by esophageal manometry which demonstrates abnormal esophago-gastric junction relaxation in association with a decrease or a lack of esophageal peristalsis [13]. Usually, AGB removal may allow an improvement of symptoms and manometric anomalies but, in some cases, abnormal esophageal motility may persist [14]. Endoscopic treatment by dilation should be considered in some cases of esophageal dysmotility disorders [15].
In our patient the esophageal motility and lower esophageal relaxation were normal during high resolution manometry. However, an area of hyper pressure 5-6 cm below the lower sphincter of the esophagus was revealed, in connection with the gastric fibrous scare. In our patient, endoscopic dilatation was not effective, probably because the balloon diameter (18 mm) was not sufficient enough to dilacerate the scare. A 30 mm pneumatic dilatation, like during achalasia, could be tested, but due to the intensity of symptoms with rapid weight loss and after medico-surgical consultation we decided a laparoscopic approach which confirmed the diagnosis and allowed resection of the scare. After an extensive literature research, this complication has not been reported before.
In conclusion, chronic vomiting after AGB removal is probably mainly related to pseudo-achalasia, but if esophageal manometry is normal, the hypothesis of persistent gastric fibrous scare located few centimeters below the usual position of the AGB must be evocated. In our case, gastric pressure measurement during high resolution manometry, which is not usually performed, allowed the diagnosis.
The authors declare that they have no conflicts of interest in relation to this article.