Liposarcoma is the most common soft tissues sarcoma in adults. Pleiomorphic liposarcoma (PLS) is a rare subtype of liposarcoma with the highest malignancy rate. In this paper we present a rare case of pleiomorphic liposarcoma of the colon and the management of the disease.
We report a 54-years-old patient diagnosed of pleiomorphic liposarcoma after a segmental colonic resection for colonic ileus.
Liposarcomas are rarely encountered in the gastrointestinal tract. The accurate diagnosis is histopathological. No consensus exists about their management. The radical surgery is the best treatment and the combination with radiation therapy or chemoradiation can be discussed in reference center.
Pleiomorphic liposarcoma of the colon is extremely rare with high recurrence and poor prognosis. The treatment is still controversial. More studies are required to define guidelines for the treatment and the follow-up of the patients.
Liposarcoma, Colon, Pleiomorphic
Liposarcoma is the most common soft tissues sarcoma in adults and represents 20% of mesenchymal malignancies and 50% of retroperitoneal sarcomas [1,2]. It occurs most commonly in the extremities and retroperitoneum and rarely involves the gastrointestinal tract [2-4]. Only a few cases of primary colic liposarcoma have been reported in the literature and there are no treatment guidelines. Pleiomorphic liposarcoma (PLS) is a rare subtype of liposarcoma with the highest malignancy rate [5,6]. We present here a rare case of primary colic pleiomorphic liposarcoma and the discussion of the treatment and follow-up of the patient.
A 54-year-old male patient visited the emergency room with a three-week history of abdominal pain, a fifteen-pound weight loss and a slow transit with sometimes diarrhea and swelling. He had no significant past medical or surgical history. An abdominopelvic CT showed an invagination of the left colon on a solid mass with an associated significant ileus. He underwent emergency surgery by laparotomy. A solid mass of about 4 cm in length was palpated just below the left colic flexure. There were no other particular findings during the surgery: no ascites, the colon was well vascularized, no carcinomatosis. The liver was macroscopically healthy. The caecum and transverse colon were dilated but not the small intestine, so the colic valve was still competent. A segmental resection of the descending colon with a large lymph node dissection has been performed before a mechanical anastomosis. Intraoperative macroscopic examination of the resection revealed a thickening of the colonic wall without any presence of a real solid mass. No extemporaneous examination was done due to the nocturnal care. After the surgery, the patient presented a post-operative ileus at day 7 with a good outcome after a conservative treatment. The wound healing was marked by an abscess of the abdominal wall which needed a drainage and local treatment.
We send the resected bowel to pathologists and the microscopic examination showed no infiltered nodes (17 loco regional nodes were collected). The resected margins were free from lesions. There were no vascular infiltration. The colonic submucosa was infiltrated by the lesion but not the muscularis layer. The first histological report identified a fusocellular polypoid sarcoma as an ulcerated lesion of 4 cm. The case of the patient was then discussed at our tumor board. A work-up for extension included a thoracic CT-scan and PET-CT. The thoracic CT showed a micronodule in the middle lobe a his right lung with a lymph node. The results of the PET-CT were negative for another tumor and the micronodule of the right lung was not suspect. Further examinations for typing of the tumor were done at the University Hospital of Zurich. Several analysis were done on the specimen to achieve the accurate diagnosis: histologically, the tumors cells were very large, pleiomorphic with broad cytoplasmic masses, hyperchromic chromatin and many atypical mitotic Figure 1. The presence of cells with cytoplasmic vacuoles was confirmed so the term of pleiomorphic lipoblasts can be used. By immunohistochemistry, reactions for pancytokeratin, SI 00, SMA, desmin, CD34, CDI 17, STAT6, ALKI were negative. The nuclear expression of MDM2 protein was lacking. A complementary analysis of the MDM2 gene showed no presence of the gene. All these findings lead to the final diagnosis of a high-grade pleiomorphic liposarcoma of the colon.
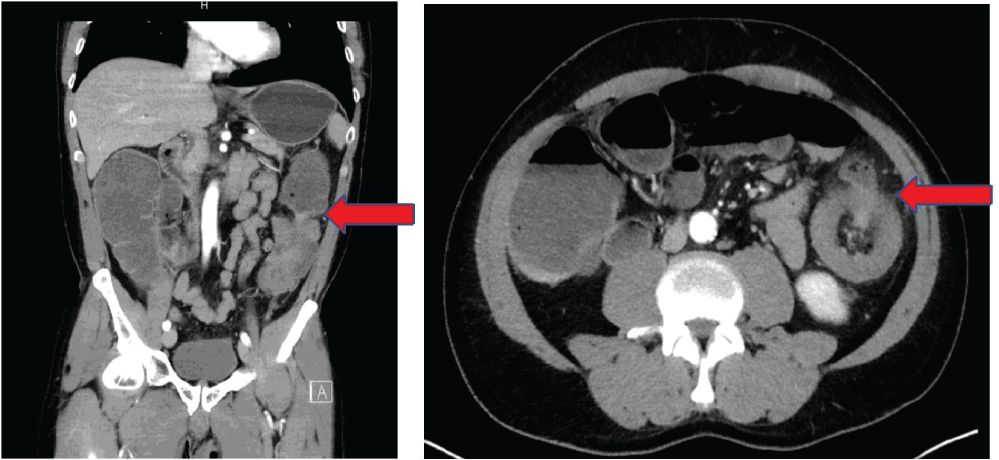 Figure 1: Preoperative computed tomography findings: colonic ileus with a stenosis on the descending colon and thickening of the colonic wall. View Figure 1
Figure 1: Preoperative computed tomography findings: colonic ileus with a stenosis on the descending colon and thickening of the colonic wall. View Figure 1
This report describes an extremely rare case of descending colon pleiomorphic liposarcoma. Pubmed was queried for articles with the terms « liposarcoma » and « colon » for the past 20 years. Seventy-nine articles were returned but only 11 described primary colonic liposarcoma. The pleiomorphic type is rarer with only a few cases in the past 20 years [5,7,8]. This explain more the fact that the treatment and follow-up of the pleiomorphic liposarcoma is based on the knowledges of the retroperitoneal sarcomas.
Liposarcoma is one of the most common soft tissue sarcoma. They are found commonly in the extremities or in the retroperitoneum specially for the dedifferentiated forms [1,3]. According to the World Health Organization guidelines, there are five types of liposarcomas: Well-differentiated, Dedifferentiated, Myxoid, High-grade myxoid, Pleiomorphic [1,9,10]. The classification is based on morphologic cell features and cytogenic aberrations [7-9]. The histologically study of the material and immunohistochemistry tests are required to do the accurate diagnosis. The pleiomorphic liposarcoma is characterised by the presence of lipoblasts and thus the presence of cytoplasmic vacuoles. The presence of mature fat cells must be excluded in this type of liposarcoma. By immunohistochemistry the pleiomorphic liposarcoma shows a negative response to the proteins MDM2, CDK4 and p16. The diagnosis can be separated at this point from the dedifferentiated liposarcoma which are positive for the 3 proteins (MDM2, CDK4, p16) [9-11]. The grade of the tumor is based on different criterias: the tumor differentiation, the mitotic index and the tumor cell necrosis. In this case we have a high-grade tumor with a poor prognosis.
In fact, the survival-rate of the patients and recurrence of the disease are related to different factors: the grading of the tumor, the subtype of the tumor, the margins of the resection, the presence or not of metastasis and the tumor size [12]. Concerning the grading, the highest the score the poorest the prognosis. A high grade tumor is also related to a high risk of recurrence [13]. Histologic subtype has been shown to be the most important predictor of local recurrence and distant metastasis [14,15]. Low-grade liposarcoma (well differentiated and myxoid) have a high rate of local recurrence (approaching 60%) but a very low rate of distant recurrence. High-grade liposarcoma have higher rates of distant metastasis and local recurrence [14,16,17].
In this case, we are interested in the pleiomorphic type which is very rare, and accounts for only 5% to 10% of lipomatous tumors [9,16]. This type is a high-grade and aggressive tumor with a high mortality rate [6,18]. Like the others high-grade liposarcomas the rate of metastasis is about 20% [7,18]. The most common sites for metastasis are lung, liver, bones and pancreas. The prognosis is poor with a survival rate at 5 years of 29% [7]. This type of tumor (PLS) often appears between 40-60 years of age [4,5,16]. The sex ratio is equal [5,9]. In fact the diagnosis is always late due to deep localisations of the tumors and the rarity of early symptoms. The most common clinical signs are the growth of a progressively painless mass, and after, the symptoms due to the compression of this mass such as abdominal pain (50%-80%), vomiting or disturbance of bowel mobility (60%) [7,8].
Like the other types of liposarcomas, PLS is the most often found in the extremities, retroperitoneum, abdominal wall, mesentery, head and neck region. In our case the colonic localisation is really unusual and rare [3,7]. The tumor examinations such CT, MRI, and ultrasound are required to assess the size and the resectability of the tumor but give no information about the type of the liposarcoma [8,18]. The final diagnosis of PLS is always histological [8]. The factors of poor prognosis are the same as for the other types of sarcomas: large tumor size, positive margins, a deep localisation, dissemination of the disease and the presence of necrosis in the tumor [4,6]. The mean treatment is a large radical surgical resection if it can be performed [3,19]. The local recurrence rate of PLS is very high but adjuvant treatment is still controversial [7,16]. Chemotherapy does not have any benefits on the survival rate of the patients or on the metastasis rate. The doxorubicin-based chemotherapy has been suggested to minimize the risk of relapse [6,8,15,16]. Radiotherapy doesn’t seem to have positive effects on the survival rate but limits the risk of recurrence [1,6,8,16]. At this time there are no standardized guidelines for PLS. There are although new recommendations concerning soft tissues and visceral sarcomas from the ESMO-EURACAN group (European Society for Medical Oncology- European Reference Network for rare adult solid cancers) [20]. These guidelines are for the diagnosis, treatment and follow-up of the patients. Regarding the diagnosis, the patient need imagery before operation (MRI, CT) but also a needle biopsy to analyse the subtype of the tumor. To better manage the operation a PET-CT can be added to realize the size of the tumor and the presence of necrosis in the tumor. The pathologist has to come from an expert center. The treatment depends on the grade of the tumor and if the disease is local or not. For grade 1 the surgery is recommended. If the resection is R0 the patients will undergo a follow-up. If the resection is R1, some radiotherapy can be done after the surgery. For grade2 and 3, the surgery is the best treatment if the operation if possible. A R0 resection will lead to a follow-up. R1 resection can be followed by radiotherapy or chemotherapy. For the tumors grade 2/3 and no possibility of surgery a neoadjuvant treatment of chemotherapy or radiotherapy can be proposed in order to decrease the size of the tumor and go to the surgery. For advanced, metastatic or unresectable disease, a chemotherapy protocol with doxorubicin can be used in order to stabilize the disease. The recommendations for the follow-up used for PLS are the same as for retroperitoneal sarcomas: High-risk patients generally relapse within 2-3 years, whereas low-risk patients may relapse later. Relapses most often occur to the lungs. Therefore, routine follow-up may focus on these sites. A practical approach in place at several institutions is as follows: surgically-treated intermediate-/high-grade patients may be followed every 3-4 months in the first 2-3 years, then twice a year up to the fifth year, and once a year thereafter; low-grade sarcoma patients may be followed for local relapse every 4-6 months, with chest X-rays or CT scan at longer intervals in the first 3-5 years, then annually. If the margins are negative [20-23].
The recommendation of the reference center (Zurich) for our patient was to perform do a thoraco-abdominal CT every 3 to 6 months during the first 5 years after surgery. No adjuvant treatment was suggested.
In case of recurrence, as shown in the literature, the best treatment would be a second surgery [19]. If the removal of the tumor is not possible or if the patient is not able to undergo a second surgery, then we would have to discuss about chemotherapy or radiotherapy [21-23].
Pleiomorphic Liposarcoma of the colon is a rare but aggressive neoplasm with poor prognosis. Radical surgery is the best treatment for the survival of the patients. Adjuvant treatment such chemotherapy or radiotherapy still remains controversial but the ESMO study gives new clear recommendation. The patients have to be followed in a reference center as due to the rarity of the PLS.
The authors have no conflict of interest.