When there are multiple vague positive findings and a plethora of possible answers, as well as multiple possible consultants with whom to work, it can sometimes be difficult to decide on the right course of action. A full and through history becomes especially more important in cases like this to help direct work up and therapy. Our case report illustrates that a consult that adds obtains more history and rules out a disease process adds helpful information in patient management. The input of consultants that exclude diseases may have an input in potentially complicated patient with a relatively rare disease, transforming the case and clinical picture in a way that helps to generate a management plan. In our patient with a complicated recurrent skin rash, positive antinuclear antibody test, and history of cancer, the right answer was the multiple consultations that allowed for rapid assessment. It ensured prompt and appropriate medical care that may have been lifesaving in this unusual case of Necrolytic Migratory Erythema (NME). In fact, it should be remembered that a negative consultation allows the medical team to reject one hypothesis and move on to a different theory of the symptoms, and provide appropriate treatment relatively quickly.
A 38-year-old female presented to the Emergency Department with a painful rash on her right earlobe, cheeks, and left hand, as well as a fever of 102 ℉ for three days prior to presentation. She had similar symptoms in the past both three and five months prior to this admission. At those times, she was diagnosed with cellulitis and improved with antibiotic treatment. On this admission, she was afebrile with normal vital signs. There was an erythematous, dyspigmented, ovoid annular lesion with a raised border on the left cheek, an erythematous lesion with purulent discharge on the right ear lobe, and a 2 mm flat circular area of erythema on the lateral aspect of her left hand at the base of the fifth metacarpal (Figure 1). She also had left periorbital edema and right submandibular and left supraclavicular lymphadenopathy.
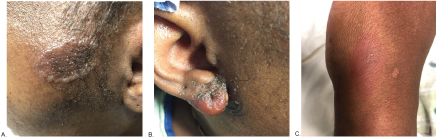 Figure 1: A) Skin finding on left cheek; B) Right ear lobe; C) Left lateral hand.
View Figure 1
Figure 1: A) Skin finding on left cheek; B) Right ear lobe; C) Left lateral hand.
View Figure 1
Laboratory tests showed erythrocyte sedimentation rate (ESR) 41 mm/HR (reference range 0-20), C-reactive protein (CRP) 2.11 mg/dL (reference range < 1.0), see Table 1. Prior to her admission, an antinuclear antibody (ANA) and other serology had been performed (Table 2). Based on her history, physical, and serological tests, rheumatology and dermatology consultations were requested.
Table 1: Relevant components of comprehensive metabolic panel and complete blood count. View Table 1
Table 2: Antinuclear antibody Screen, Immunofluorescence Assay, Reflex Titer/Pattern. View Table 2
Maxillofacial Computed tomography (CT) scan with contrast demonstrated stranding of the subcutaneous fat anterior to the left maxillary sinus and along the anterior/inferior margin of the left orbit suggestive of periorbital cellulitis.
Systemic lupus erythematous (SLE) was considered to be less likely by the rheumatology team, and the dermatology team performed a skin biopsy of the lesion on the left cheek.
The pathology result of the skin biopsy confirmed a diagnosis of necrolytic migratory erythema (NME) based on upper epidermal confluent necrosis associated with pale keratinocytes and foci of acute folliculitis (Figure 2). Typical features of discoid lupus erythematosus were not seen, including no plugging hyperkeratosis, no thickening of the basement membrane, no periadnexal and perivascular lymphocytic infiltrate, and no dermal mucin.
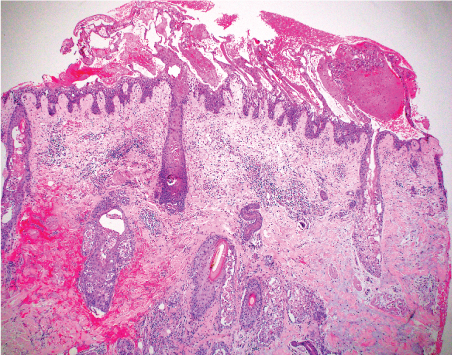 Figure 2A: 4X (40X magnification) - H&E showing superficial epidermal confluent necrosis and pale epidermal keratinocytes with relative preservation of the lower epidermal keratinocytes and basilar layer in a psoriasiform pattern, features of necrolytic migratory erythema (NME). Note the pilosebaceous folliculitis, a feature also described in NME (40 magnification).
View Figure 2A
Figure 2A: 4X (40X magnification) - H&E showing superficial epidermal confluent necrosis and pale epidermal keratinocytes with relative preservation of the lower epidermal keratinocytes and basilar layer in a psoriasiform pattern, features of necrolytic migratory erythema (NME). Note the pilosebaceous folliculitis, a feature also described in NME (40 magnification).
View Figure 2A
 Figure 2B: Epidermal confluent necrosis associated with pale keratinocytes and focus of acute folliculitis at the arrow (H&E 100 magnification (10X)).
View Figure 2B
Figure 2B: Epidermal confluent necrosis associated with pale keratinocytes and focus of acute folliculitis at the arrow (H&E 100 magnification (10X)).
View Figure 2B
With this new information, the patient's past history was explored further. She did have prior history of a T1m N0 pancreatic neuroendocrine tumor 17 months prior to presentation. She did have a pancreatectomy and splenectomy at that time as part of her treatment. Pre-operatively, serum vasoactive intestinal peptide (VIP), serotonin, chromogranin A, pancreatic polypeptide, and glucagon were normal. Post-operative non-fasting insulin level was 25.2 uIU/mL (reference range < 24) and C-peptide was 4.03 ng/mL (reference range 0.50-2.00).
The patient was treated with antibiotics for left periorbital cellulitis and instructed to follow-up with her oncologist to evaluate the possibility of residual or recurrent pancreatic neuroendocrine tumor. The patient did not need follow up with rheumatology or dermatology upon discharge.
As she had a recent a computed tomography (CT) of her abdomen and pelvis three months prior, and a CT angiogram of her chest was done one month prior to the most recent presentation, these were not repeated by the oncology team in the outpatient clinic. Both the CT abdomen and pelvis and CT angiogram were negative for recurrence of her malignancy or metastatic disease. She is planned for octreotide scan to further evaluate her for recurrence.
NME is a rare dermatosis that can be difficult to diagnosis clinically. It can be associated frequently with gastrointestinal autoimmune diseases, solid organ malignancies, as well as endocrine tumors, predominantly glucagonomas. With a suggestive history of an appropriate malignancy, a biopsy of the skin lesions in the hands of an experienced dermatopathologist is the best way to make the diagnosis. NME is an important example of a dermatologic manifestation or an indicator of underlying systemic disease.
In our case given the relative frequency and recurrent nature of the rashes, her sex and race and the antecedent history of a positive ANA, cutaneous lupus was the obvious answer. Her prior history of pancreatic cancer was not initially obtained, and NME was therefore not considered in the differential diagnosis. A rheumatology consult was requested for the presumed cutaneous lupus. Further history confirmed the history of a metachronous nonfunctional pancreatic neuroendocrine tumor extirpated 17 months prior to presentation. This lead to the other differentials including paraneoplastic syndromes and the confirmation of NME on biopsy.
The lesions of NME appear as scaling erythematous patches that may be painful, pruritic [1], and classically arise on the distal extremities, lower abdomen, and periorificial regions [2,3]. They occasionally appear on the face [4] as was seen in this case (Figure 1). Several histologic patterns can be seen in NME, the most distinctive being upper epidermal pale, vacuolated keratinocytes with either focal or confluent necrosis (Figures 2a and Figure 2b). The confluent upper epidermal necrosis has similarities to toxic epidermal necrolysis which shows complete loss of the epidermis, hence the term "necrolytic" in NME. The erythema portion of NME corresponds to the annular erythema surrounding areas of superficial necrolysis leading to either bullae or crusted erosions (Figure 1). An uncommon, but known, histologic finding in NME is suppurative folliculitis, also present in this case (Figure 2a and Figure 2b). Laboratory workup for glucagonoma syndrome may reveal a normocytic anemia [5] which was true of this case. NME has been associated with liver cirrhosis, intestinal malabsorption, and inflammatory bowel diseases [1]. Other rarely reported associations with NME include zinc deficiency, opiate abuse [3,7], pancreatic insufficiency, jejunal adenocarcinomas [8], iatrogenic glucagon administration.
The initial differential diagnosis for this case included facial cellulitis, which was the eventual discharge diagnosis, but rarer disease like SLE, subacute cutaneous lupus erythematosus, sarcoidosis based on the appearance of the lesions, and even recurrence of her malignancy had to be considered in the differential. Other diagnosis to consider include drug reactions and various dermatitis, and nearly always would necessitate a dermatology referral.
In our case, the consideration of cutaneous lupus led to a rheumatology and subsequently to a dermatology consult for a biopsy. Though her symptoms were not lupus related based on the lack of photosensitivity of her rash, she did have an elevated CRP, ESR, reactive thrombocytosis, with positive ANA making the rheumatology consult worthwhile.
NME should be part of the differential diagnosis when considering cutaneous lupus in a patient with a rash, especially if there is a history of malignancy.
Most importantly, we should be include a full and detailed history including past medical history as well as a complete physical examination in the evaluation of every patient. An exhaustive list of differential diagnosis should be considered as possible causes of symptoms if supported by the history, including less likely rarer diagnosis.