Transcatheter aortic valve implantation (TAVI) has become a novel therapeutic option for patients with severe aortic stenosis who have been considered inoperable or high risk for conventional surgical aortic valve replacement. The aim of this study was to determine the effect of TAVI on neutrophil to lymphocyte ratio (NLR).
A total of 97 patients with severe aortic valve stenosis undergoing TAVI in our clinic were included in the study. The patients were divided into two groups based on valve type; patients with CoreValve bioprosthesis (n = 56) and patients with Edwards SAPIEN-XT bioprosthesis (n = 41). Blood samples were drawn before TAVI and were performed at 1 and 6 months after TAVI.
The patients in the Edwards SAPIEN group were older than the patients in the CoreValve group (76.7 ± 6.6 years, p = 0.019). Eurologistic score (p = 0.002), choronic obstructive pulmonary disease (p = 0.006), and pulmonary arterial pressure-systole (p = 0.001) were significantly higher in patients in the CoreValve group than those in the Edwards SAPIEN group. Aortic valve area had increased in both groups after the procedure (p < 0.001 and p < 0.001, respectively). In addition, interventricular septal wall thickness, posterior wall thickness, and left ventricular mass index regressed in both groups at 6 month follow-up (p < 0.001, p < 0.001, and p < 0.001, respectively). When compared with the baseline NLR, a significant decrease was observed in the values at 1 and 6 months in both groups (p < 0.001 and p < 0.001, respectively).
A decrease in the NLR can be both a sign of regression of valvular inflammation and myocardial hypertrophy in patients with degenerative aortic stenosis after TAVI. Our results show that it may be used during the follow-up of patients with TAVI.
Aortic stenosis, Neutrophil to lymphocyte ratio, Transcatheter aortic valve implantation
The prevalence of aortic stenosis is growing in the older population. Increases in longevity due to advances in health care have resulted in an increase in the diagnostic rate of aortic stenosis. Although the prevalence is low in patients < 60-years of age, it increases to approximately 10% in patients > 80-years of age [1]. Calcific aortic valve disease (CAVD) occurs when there is an obstruction to the ejection of blood from the left ventricle, generally at the valvular level in adults [2]. CAVD presents the proliferative and inflammatory changes that occur with lipid accumulation [3]. Degenerative disease of the aortic valve (aortic sclerosis) most likely represents an early stage in the disease process that eventually results in aortic stenosis. Transcatheter aortic valve replacement or implantation (TAVI) has become a viable and durable therapy for patients with severe aortic stenosis who have been considered "inoperable" or "high-risk" for conventional surgical aortic valve replacement (SAVR) [4].
The white blood cell count (WBC) and its subgroups, neutrophil and lymphocyte counts, are among the indicators of systemic inflammation [5]. The neutrophil to lymphocyte ratio (NLR) has been proposed as prognostic marker of the systemic inflammatory response. The important role of inflammation in cardiovascular disorders has been well established. Several studies have shown that a high NLR is associated with a poor clinical outcome in various cardiac diseases, including coronary artery disease (CAD), coronary artery ectasia, heart failure, and acute coronary syndrome [6-8]. The relationship between the NLR and the severity of aortic stenosis has been discussed in previous studies [9,10]. However, there are limited data regarding the NLR according to the implanted valve type. In the present study, we investigated the NLR in patients undergoing a TAVI procedure according to the implanted valve type.
We retrospectively analyzed data from two different centers in patients undergoing transfemoral TAVI between October 2013 and December 2015. A total of 97 consecutive patients with symptomatic, severe aortic stenosis underwent TAVİ using the self-expandable CoreValve (n = 56; Medtronic, Inc., Minneapolis, MN, USA) or the Edwards SAPIEN-XT (n = 41; Edwards Lifesciences, Irvine, CA, USA) bioprosthetic valves in this study. The patients were divided into two groups according to the valve type. The TAVI procedure was performed on patients who had severe symptomatic aortic stenosis, an aortic valve area (AVA) < 1 cm2, a mean gradient > 40 mmHg, a status of being unsuitable for SAVR due to high surgical risk, and a logistic EuroScore > 20% or a logistic EuroScore < 20% with significant co-morbidities. Dobutamine stress echocardiography was used to discriminate severe calcific aortic stenosis in those with left ventricular (LV) systolic dysfunction co-existing with a mean peak gradient of 30-40 mmHg and an AVA ≤ 1 cm2 (low-flow low-gradient). Severe aortic stenosis is present if there is an increased mean gradient > 40 mmHg with AVA < 1 cm2 at any flow rate.
The exclusion criteria included clinical evidence of infective endocarditis or any other active infection, cancer, hematopoietic disease, autoimmune or inflammatory disease, current steroid or chemotherapy treatment, or acute coronary syndrome on admission. Congenital or rheumatic aortic stenosis was also excluded. Initially, 118 patients were eligible for this study. Patients who had clinical adverse events, including death from any cause (four patients), myocardial infarction (four patients), stroke (two patients), life-threatening bleeding (three patients), acute kidney injury (three patients), valve-related dysfunction requiring a repeat procedure (three patients), or major vascular complications (two patients) after the procedure were also excluded from the study. The exclusion criteria were applied to the two groups, and consequently 21 patients were excluded from this study.
All patients underwent a complete medical assessment, physical examination, and laboratory investigation; electrocardiogram, chest X-ray, transthoracic echocardiography (TTE), transesophageal echocardiography (TEE), a coronary assessment with invasive angiography or computed tomography (CT), and a vascular access assessment with CT imaging. The local heart team consisting of cardiologists, cardiovascular surgeons, and anesthesiologist conducted the qualification process. Informed consent for diagnostic treatment procedures was received from all patients. The institutional ethics committee approved this study protocol (Protocol No: 2018/12-5).
TTE was performed on all patients prior to the procedure, after the procedure, and during the follow-up period. Echocardiograms, including Doppler measurements, were performed according to current guideline recommendations [11]. Echocardiographic measurements and hemodynamic calculations were performed by echocardiographers experienced in the quantitative assessment of valvular heart disease. The examinations were performed using the IE33 device (Philips Medical Systems, Bothell, WA, USA). Diastolic interventricular septum thickness (IVS), diastolic posterior wall thickness (PWT), left atrial (LA) diameter, left ventricle end systolic (LVESD) and end diastolic dimensions (LVEDD) were measured from parasternal long-axis view. The LV end-diastolic volume and LV end-systolic volume were calculated in apical four- and two-chamber views using the biplane method of disks (modified Simpson's method). The LV outflow tract (LVOT) and aortic annulus diameter were measured in the parasternal long-axis view. The LVOT velocity and LVOT velocity time integral (VTI) were measured on a spectral Doppler display using pulsed-wave Doppler. The continuous wave (CW) Doppler method was used to obtain aortic valve peak velocities and to search for an acoustic window with the highest value. Maximum and mean aortic valve pressure gradients were estimated by the modified Bernoulli equation using the flow VTI over the ejection period on CW-Doppler recordings with a 50 mm/s timescale on the x axis. The AVA was calculated according to the continuity equation. Valvular stenosis was assessed semi-quantitatively and/or quantitatively according to current guidelines [12]. LV mass was calculated as following [11]:
LV mass (g) = 0.8 × 1.04 [(IVS + PWT + LVEDD)3-LVEDD3] + 0.6
LV mass was divided by body surface area to obtain the LV mass index (LVMI, g/m2), which cut-off values of 115 g/m2 and 95 g/m2 for men and women respectively. Body surface area (m2) was calculated using the Du Bois formula [weight (kg)0.425 × height (cm)0.725 × 0.007184]. Relative wall thickness (RWT = 2 × PWT in end diastole/LV diastolic diameter in end diastole) was calculated.
After TAVI, valvular regurgitation was defined according to the Valve Academic Research Consortium criteria [13]. Early and long-term TAVI effects were assessed using complete preprocedural TTE and complete TTE before discharge and during the follow-up period, respectively.
TAVI was performed in a hybrid operating room. The procedures were conducted under general or local anesthesia with sedation [14]. The valves were implanted via a transfemoral approach. The type of valve and the implantation access route was established based on the TEE results and CT imaging. Device success was defined as proper prosthesis placement with an aortic mean gradient < 20 mmHg, without significant (i.e., moderate/severe) aortic regurgitation [13]. Procedural success was defined as an effective valve implantation procedure with no death within 24 h and no need for conversion to surgery.
Blood samples were obtained through the antecubital vein using a 21G sterile syringe without stasis, following a 12-hour fasting period. To assess complete blood count (CBC), a Coulter LH 780 Hematology Analyzer (Architect plus ci16200 Abbott Medical, Abbott Park, IL, USA) was used to measure the hematological parameters, including WBCs, hemoglobin level, and lymphocyte counts.
After TAVI, follow-ups were performed at 1 and 6 months. Routine physical examinations, echocardiography, and laboratory tests were performed during the follow-up.
All analyses were performed using the SPSS for Windows 15.0 (SPSS Inc. Chicago, IL, USA) software package. Categorical variables are presented as numbers and percentages. Categorical variables were compared using the chi-square test. Continuous variables were tested for normality with the Kolmogorov-Smirnov test. Continuous variables are presented as mean ± standard deviation, and were compared using the paired t-test if the data were normally distributed. If the distribution differed from the normal, Wilcoxon signed rank test was performed. Differences between the two groups were tested with the Student's t-test. To test the group differences for the variables mean gradient, NLR, and LVMI repeated measures analysis of variance (MANOVA) was conducted. Mean gradient, NLR, and LVMI were measured at baseline, 1 month, and 6 months later. A p value < 0.05 was considered significant.
Of 97 patients (mean age 74.6 ± 7.4 years; 50 males and 47 females) underwent TAVI in our clinic. The baseline clinical, demographic, characteristic, and laboratory values of the patients are shown in Table 1. No significant differences were observed between the groups in terms of sex, smoking status, body mass index, CAD, hypertension, peripheral artery disease, diabetes, coronary artery bypass grafting, stroke, hemoglobin, creatinine, or WBC count. Most of patients were in the New York Heart Association class III-IV, and there were no significant differences between the groups (Table 1).
Table 1: Demographical and clinical characteristics of patients. View Table 1
The patients in the Edwards SAPIEN group were older than the patients in the CoreValve group (73.1 ± 7.6 vs. 76.7 ± 6.6 years, p = 0.019, respectively). The Eurologistic score (24.7 ± 5.0 vs. 21.8 ± 3.1, p = 0.002), chronic obstructive pulmonary disease status (24 (42.9%) vs. 6 (14.6%), p = 0.006)), and systolic pulmonary arterial pressure (49.6 ± 12.4 vs. 40.3 ± 6.2 mmHg, p = 0.001) were significantly higher in patients in the CoreValve group than those in the Edwards SAPIEN group. However, the neutrophil count (5.5 ± 2.1 vs. 7.0 ± 1.6, p = 0.001) and leukocyte count (2.0 ± 0.8 vs. 2.4 ± 0.9, p = 0.013) were higher in patients in the Edwards SAPIEN group than those in the CoreValve group (Table 1).
As expected, the echocardiographic assessments of aortic valve obstruction improved after TAVI. The AVA increased from 0.77 ± 0.12 to 1.64 ± 0.15 cm2 (p < 0.001) in patients in the CoreValve group and from 0.80 ± 0.11 to 1.65 ± 0.13 cm2 (p < 0.001) in patients in the Edwards SAPIEN group (Table 2). The improvement in the AVA was accompanied by a significant decrease in the maximal and mean gradient in both groups (p < 0.001 and p < 0.001, respectively; Table 2).
Table 2: Comparison of echocardiographic variables before and after TAVI. View Table 2
At the 6-month follow-up, the IVS regressed from 13.9 ± 1.6 to 11.5 ± 0.9 mm and the PWT decreased from 13.0 ± 1.4 to 11.3 ± 0.8 mm (p < 0.001) in the CoreValve group. The IVS regressed from 14.5 ± 1.4 to 10.8 ± 0.9 mm and the PW decreased from 12.7 ± 0.7 to 11.6 ± 0.7 mm (p < 0.001) in the Edwards SAPIEN group. In addition, the RWT decreased from 0.55 ± 0.08 to 0.49 ± 0.06, LV mass regressed from 265.9 ± 59.1 to 195.3 ± 39.0 g, and the LVMI regressed from 144.5 ± 31.6 to 106.0 ± 21.1 g/m2 (p < 0.001) in the CoreValve group. The RWT decreased from 0.52 ± 0.09 to 0.45 ± 0.07, LV mass regressed from 289.1 ± 48.1 to 210.4 ± 39.7 g, and the LVMI regressed from 160.9 ± 35.7 to 117.0 ± 27.1 g/m2 (p < 0.001) in the Edwards SAPIEN group (Table 2). Improvements in functional capacity were observed in all patients during the follow-up, and there were no between-group differences.
A comparison of the NLR parameters were shown in Table 3. A significant decrease was observed between the baseline NLR values and those at months 1 and 6 in both groups (p < 0.001 and p < 0.001, respectively) (Table 3 and Figure 1).
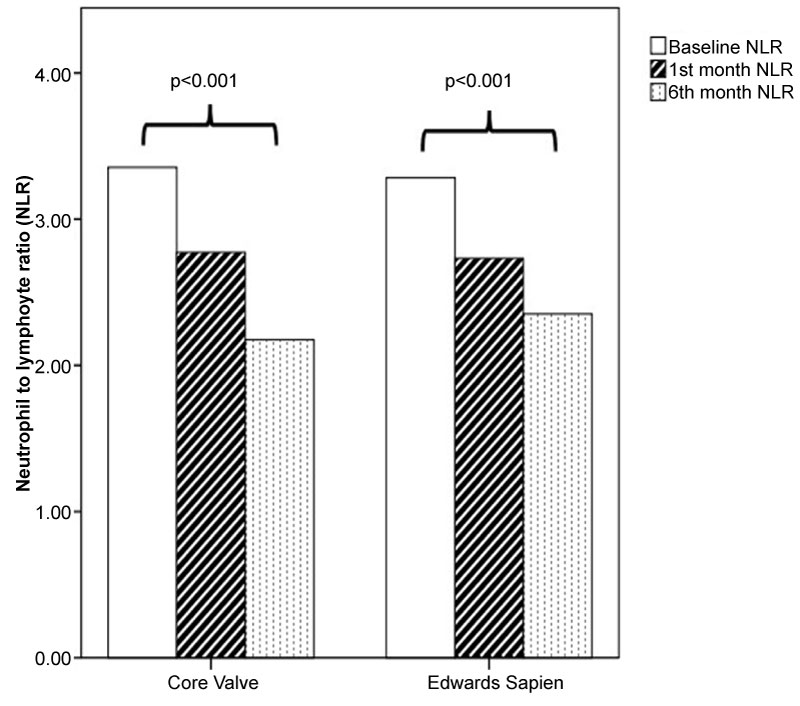 Figure 1: Neutrophil-lymphocyte ratio before and after transcatheter aortic valve implantation according to valve type.
View Figure 1
Figure 1: Neutrophil-lymphocyte ratio before and after transcatheter aortic valve implantation according to valve type.
View Figure 1
Table 3: Comparison of NLR parameters in the study groups. View Table 3
Results of repeated measures ANOVA were shown in Table 4. According to the significance values from pairwise comparisons, there was no significant difference in NLR and mean gradient, but there was a significant difference in LVMI between Time 1 (baseline), Time 2 (1 month), and Time 3 (6 months) (p < 0.019). In addition, The Tests of Within-Subjects Contrasts showed that there was a significant linear trend for LVMI (Figure 2).
 Figure 2: The profile plot shows decrease on left ventricular mass index (LVMI) for both groups on 1st month and on time point 6th month. The distance between each groups shows the mean significance.
View Figure 2
Figure 2: The profile plot shows decrease on left ventricular mass index (LVMI) for both groups on 1st month and on time point 6th month. The distance between each groups shows the mean significance.
View Figure 2
Table 4: Results of Repeated Measures ANOVA. View Table 4
The main finding of this study was that the NLR decreased significantly during follow-ups in patients with severe aortic stenosis undergoing TAVI. The AVA increased significantly on echocardiography at 1 and 6 months, with significant decreases in the maximal and mean gradients regardless of the implanted valve type. In addition, there was a significant decrease in LVMI values during the follow-up. This is the first study to demonstrate the effect of the TAVI procedure on the NLR according to the implanted valve type.
CAVD is a slow, progressive disorder that ranges from mild valve thickening without obstruction of blood flow, termed aortic sclerosis, to severe calcification with impaired leaflet motion, termed aortic stenosis [15]. The important role of inflammation in cardiovascular disorders has been well established, and WBCs and subtypes of WBCs play an important role in modulating the inflammatory response during the atherosclerotic process [5,16].
CAVD is considered an actively regulated and progressive inflammatory disease. CAVD is characterized by a cascade of cellular changes that initially cause fibrotic thickening, followed by focal subendothelial plaque-like lesions on the aortic side of the leaflets, at the cusp coaptation lines, and at the cusp attachment sites to the aortic wall that extend to the adjacent fibrosa [15]. Calcification, a property of aortic valve stenosis, is also a feature of the active inflammatory process. The predominant cell types are inflammatory cells, including monocytes, which infiltrate the endothelial layer and differentiate into macrophages, and, in turn, take up oxidized low-density lipoprotein cholesterol and become foam cells. T lymphocytes are also prominent, become activated, and release a variety of pro-inflammatory cytokines [2]. These cytokines promote cellular proliferation and remodeling of the extracellular matrix by acting on valvular fibroblasts. Moreover, pulsatile shear stress on the ventricular side and low and reciprocating shear stress on the aortic side of the cusps induces endothelial dysfunction/leakage with a buildup of lipids and minerals, which provoke inflammation. As stenosis progresses, the wall shear stress across the aortic valve dramatically increases [17].
The NLR has recently been a focus of attention, especially in cardiovascular disease. The NLR has been proposed for use in risk stratification of many cardiovascular diseases, including aortic stenosis [18,19] and heart failure [7]. A high NLR is associated with poor clinical outcomes in various cardiac diseases, including acute coronary syndrome, stable coronary disease, non-ST-segment elevation myocardial infarction (NSTEMI), and STEMI [20,21]. An increase in the NLR is related to the severity of calcific aortic stenosis and LV systolic dysfunction in patients with severe calcific aortic stenosis [9].
Cho, et al. [22] estimated that the major adverse cardiovascular event-free survival rate at 5 years for patients with severe calcific aortic stenosis undergoing SAVR or medical therapy is 84.6% for the low risk group (NLR ≤ 2). Habib, et al. [23] dichotomized 234 patients into two groups depending on their preoperative NLR level with a cutoff value of 3. They showed that a high NLR is associated with increased cardiac mortality in patients with aortic stenosis undergoing SAVR. In our study, the NLR value decreased from 2.7 to 2.13 at 6 months. Furthermore, there was no difference in the changes in NLR values according to the implanted valve type.
Condado, et al. [24] reported that an elevated NLR is correlated with occurrence of the composite early safety outcome at 30 days in patients who undergo balloon expandable TAVI. A high variation in the NLR from baseline to discharge is correlated with a higher 1-year survival and lower mortality. That study only included patients implanted with the Edwards SAPIEN valve. Our study enrolled patients who received one of two separate valves, but patients who had major cardiovascular events and mortality within 6 months were excluded from the study.
Gul, et al. [25] evaluated 33 patients (Edwards SAPIEN = 27, CoreValve = 6) who underwent TAVI in terms of hematologic parameters, including the NLR and the mean platelet volume. They demonstrated that the mean platelet volume progressively decreased, but there were no differences in the NLR values from before to 1 and 4 months after TAVI. In contrast, we detected a significant decrease in the NLR value after TAVI. One of the possible reasons for the different results was the smaller number of patients than in the present study. Another possibility is a shorter follow-up period than in our study.
Age, neutrophil and lymphocyte cell counts were higher in the Edwards SAPIEN group, whereas the Eurologistic score, chronic obstructive pulmonary disease, and systolic pulmonary arterial pressure were higher in the CoreValve group. These results are purely coincidental. Because our data were taken from 2 different centers. CoreValve prosthesis was applied in one center, while Edwards SAPIEN prosthesis was applied in the other center.
Increased LVMI (> 115 g/m2 and > 95 g/m2 for men and women respectively) indicates left ventricular hypertrophy (LVH). LVH and myocardial fibrosis are common compensating processes in the pressure overload mechanism of aortic stenosis [26]. LV overload with increased wall stress results in remodeling, which is predominantly governed by various inflammatory cascades. The pathophysiology of the remodeling process includes increased pro-inflammatory cytokine expression, which is accompanied by leukocyte infiltration and proteolytic myocardial destruction by neutrophil-originating enzymes [27,28]. Regression of myocardial hypertrophy due to a decrease in the ventricular afterload after SAVR and TAVI is a well-recognized phenomenon. In particular, the LV mass decreases primarily within the first 6 months after TAVI [29]. Similarly, LV mass and LVMI significantly regressed after the procedure during the follow-up in our study. The LVMI was higher in the Edwards SAPIEN group because of the data from 2 different centers and the retrospective study.
Several functional studies have indicated that epigenetic mechanisms play an important role in the initiation and progression of aortic stenosis as well as LVH. These mechanisms could contribute to endothelial dysfunction, disease-prone activation of monocytes and macrophages, and osteogenic transdifferentiation of aortic valve interstitial cells, leading to valvular inflammation, fibrosis, calcification, and pressure overload-induced maladaptive myocardial remodeling and LVH [30]. Villar, et al. reported that the regression of hypertrophy at 1-year after release of pressure overload is related to the preoperative myocardial expression of remodeling-related genes. MicroRNAs represent particularly useful non-invasive biomarkers that reflect disease progression and patient prognosis for recovery after SAVR [31].
Reduced exposure to turbulent flow at the aortic valve and regression of myocardial hypertrophy play an important role in the decrease of the NLR following the TAVI procedure. In this regard, the NLR has great potential to become a biomarker for future diagnostics and monitoring.
Some limitations of the present study should be mentioned. First, it was a retrospective, short follow-up study after TAVI, in which the selected population may not reflect the entire cohort. Second, the relationship between postoperative NLR values, major adverse cardiovascular events, and mortality was not investigated. Third, parameters with a possible role in the pathophysiology, such as the epigenetic markers, C-reactive protein (CRP), interleukins, mean platelet volume, platelet lymphocyte ratio, and high-sensitivity CRP, were not measured.
A decrease in the NLR can be a sign of both regression of valvular inflammation and myocardial hypertrophy in patients with degenerative aortic stenosis after TAVI. The NLR is calculated from a complete blood count, and is a cheap, easily obtainable, and widely available marker of inflammation that may be used during the follow-up of patients with TAVI. Further prospective studies, particularly those with epigenetic markers, are needed to support the present results.
This study received no grant from any funding agency in the public, commercial or not-for-profit sectors.
All authors have no conflicts of interest to declare.