We report a rare case of double Intraductal Papillary Mucinous Neoplasm with multiple foci of adenocarcinoma originating from duodenal and gastric heterotopic pancreatic tissue, associated with gastric duplication and intestinal malrotation. Only few cases of Intraductal Papillary Mucinous Neoplasm arising within ectopic pancreas have been reported so far, and, to the best of our knowledge, no one with two coexisting lesions, both with foci of malignant degeneration, and associated to rare congenital anomalies.
Therefore, we want to report imaging features in Computed Tomography and Magnetic Resonance of this peculiar case.
IMPN, CT, MRI, Gastric duplication, Intestinal malrotation
Pancreatic ectopia is defined as pancreatic tissue that lacks anatomical and vascular connection to the normal pancreas, found in 0.5% to 13% of autopsies, most frequently in duodenum, stomach and jejunum. Although the presence of heterotopic pancreas is usually asymptomatic and often discovered incidentally, pathological changes that occur within the native pancreas may also occur rarely in the ectopic gland, including pancreatitis, benign and malignant lesions [1].
Intraductal Papillary Mucinous Neoplasm (IPMN) is a clinicopathological entity originating from the mucinous epithelium of the pancreatic ductal system, characterized by intraductal papillary proliferations and excessive mucin production, resulting in progressive cystic dilation of the pancreatic ducts [2]. IPMNs account for approximately of 20% of cystic pancreatic lesions and are classified in three types, on the basis of the site of the tumor involvement: Main duct IPMN, branch duct IPMN, and combined type. Most lesions are asymptomatic, discovered incidentally at cross-sectional imaging, but they can also be symptomatic, presenting with abdominal pain, weight loss, pancreatitis, pancreatic insufficiency [2].
Pathologically IPMNs can be divided into three groups: Benign without dysplasia, borderline neoplasms and carcinoma (invasive or non-invasive) [3]. Carcinoma is found in about 70% of IPMNs involving the main duct and in about 25% of IPMNs involving only side-branches [3].
At imaging, they are identifiable as ductal dilatation secondary to mucin production and retention, with involvement of the main pancreatic duct, the branch duct or both and can be evaluated with different imaging modalities [4]; however to evaluate patients who are suspected to have pancreatic malignancy (eg, raise in CA 19-9 values), Computed Tomography (CT) continues to be the primary technique [4].
Side-branch IPMNS can have two different appearances: A macrocystic pattern, with a unilocular or multilocular cyst with few septa, or a microcystic pattern, in which multiple thin septa separate numerous fluid-filled spaces [2]; a connection to the pancreatic duct is often present.
IPMNs can also arise within heterotopic pancreatic parenchyma. In Literature we found only few cases of IPMNs arising from ectopic pancreas: Located in the proximal hepatic duct [5] duodenum [6] jejunum [7] stomach [8], but no one focused on imaging aspects of the disease. Therefore our aim is to describe a rare case of a female patient who came to our attention with vague abdominal symptoms and rising tumor markers whose imaging investigations and surgery demonstrated a rare case of double IMPN arising from ectopic pancreatic parenchyma, associated with congenital gastroenteric anatomic anomalies.
A previously healthy 56-year-old Chinese female was referred to our clinic for non-specific recurrent abdominal pain, centered over epigastrium and right subcostal region.
The physical examination was unremarkable.
Tumor marker values were 2.5 ng/mL (normal range < 5.0 ng/mL) for Carcinoembryonic Antigen (CEA) and 95 ng/mL (normal range < 37 ng/mL) for CA 19.9. The other laboratory parameters were within the normal range.
Due to high values of CA 19.9, a thoracic and abdominopelvic enhanced Computed Tomography (CT) was executed (Figure 1, Figure 2, Figure 3 and Figure 4). The examination showed various pathologic findings: First, an oval paraduodenal lesion with a cluster of cystic-like structures (Figure 1), then a second lesion with analogous characteristics not dissociable from the gastric fundus (Figure 2), and the presence of a gastric duplication (Figure 3), finally an anomalous disposition of the entire small bowel and colon and an inversion of the superior mesenteric artery and vein (Figure 4). No pancreatic anomalies were present, the caliber of the Wirsung and secondary pancreatic ducts were normal. Chest CT showed was negative for pulmonary metastases.
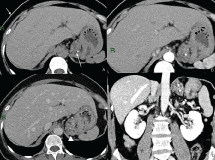 Figure 1: Contrast enhanced CT: Lesion of the gastric fundus Non-enhanced CT (A), arterious phase (B), venous phase (C) and coronal reconstruction (D) showing multiple small, clustered cystic foci combined in a lesion not dissociable from the gastric fundus (arrowhead in C). Two calcifications are well visible (arrow in A). View Figure 1
Figure 1: Contrast enhanced CT: Lesion of the gastric fundus Non-enhanced CT (A), arterious phase (B), venous phase (C) and coronal reconstruction (D) showing multiple small, clustered cystic foci combined in a lesion not dissociable from the gastric fundus (arrowhead in C). Two calcifications are well visible (arrow in A). View Figure 1
 Figure 2: Contrast enhanced CT: Periduodenal lesion Non-enhanced CT (A), arterious phase (B), venous phase (C) and coronal reconstruction (D) Venous phase demonstrating clustered small cysts conjoined in periduodenal region (arrows in C and D). View Figure 2
Figure 2: Contrast enhanced CT: Periduodenal lesion Non-enhanced CT (A), arterious phase (B), venous phase (C) and coronal reconstruction (D) Venous phase demonstrating clustered small cysts conjoined in periduodenal region (arrows in C and D). View Figure 2
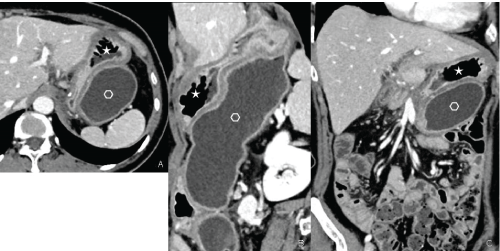 Figure 3: Contrast enhanced CT: Gastric duplication Axial plane (A), sagittal (B) and coronal (C) reconstructions showing the presence of two distinct cavities: Normal gastric cavity (star); duplication cyst (empty hexagon) with tubular morphology. View Figure 3
Figure 3: Contrast enhanced CT: Gastric duplication Axial plane (A), sagittal (B) and coronal (C) reconstructions showing the presence of two distinct cavities: Normal gastric cavity (star); duplication cyst (empty hexagon) with tubular morphology. View Figure 3
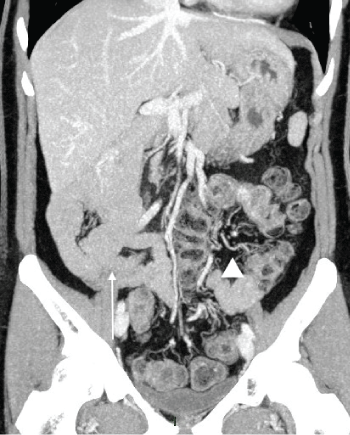 Figure 4: Contrast enhanced CT: Intestinal malrotation Maximum Intensity Projection coronal reconstruction showing anomalous disposition of the entire small bowel and colon, with small bowel located on the right side of the abdomen (arrow), while colon is on the left side (arrowhead). View Figure 4
Figure 4: Contrast enhanced CT: Intestinal malrotation Maximum Intensity Projection coronal reconstruction showing anomalous disposition of the entire small bowel and colon, with small bowel located on the right side of the abdomen (arrow), while colon is on the left side (arrowhead). View Figure 4
Contrast Enhanced abdominopelvic Magnetic Resonance (MR) confirmed CT findings (Figure 5 and Figure 6).
 Figure 5: Contrast enhanced MRI: Lesion of the gastric fundus T2 fat saturated axial image (A) and T1 fat saturated post-contrast image (B) showing a paraaortic lesion with multicystic appearance (arrows). View Figure 5
Figure 5: Contrast enhanced MRI: Lesion of the gastric fundus T2 fat saturated axial image (A) and T1 fat saturated post-contrast image (B) showing a paraaortic lesion with multicystic appearance (arrows). View Figure 5
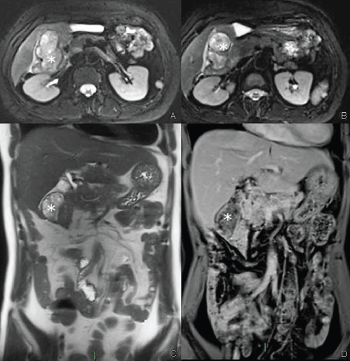 Figure 6: Contrast enhanced MRI: Paraduodenal lesion T2 fat saturated axial image (A,B), coronal HASTE image (C), post-contrast coronal T1 fat saturated (D) showing a well-defined lesion with multicystic appearance (asterisks) with thick rim-like enhancement well visible in the delayed phase (in D). View Figure 6
Figure 6: Contrast enhanced MRI: Paraduodenal lesion T2 fat saturated axial image (A,B), coronal HASTE image (C), post-contrast coronal T1 fat saturated (D) showing a well-defined lesion with multicystic appearance (asterisks) with thick rim-like enhancement well visible in the delayed phase (in D). View Figure 6
Due to imaging results at CT and MR, the patients did not underwent gastroscopy, but an Endoscopic Ultrasound (EUS) that endorsed the presence of a para-duodenal lesion infiltrating the wall of the duodenal second portion and of a second lesion involving the gastric fundus; EUS-guided FNA performed on the lesion of the gastric fundus demonstrated cytological atypia, however the cellularity was not adequate to make a definitive diagnosis.
The patient underwent laparotomy with duodenocephalopancreatectomy and resection of the gastric fundus lesion; the presence of a mesenterium commune, with small intestine located at the subhepathic region and colon in the midline and in the left flank, was confirmed.
Pathology of the specimen demonstrated ectopic pancreatic tissue with cystic dilatations of the pancreatic ducts, within the ducts, columnar mucin-containing epithelium with papillary proliferations was seen; the presence of multiple foci of adenocarcinoma was found in both lesions. The duodenal lesion was full-thickness infiltrating the visceral wall.
These histological findings were consistent with foci of adenocarcinoma arising from IPMN in ectopic pancreatic tissue.
Abdominal CT is the widest used imaging technique in the diagnosis of IPMN, especially since the introduction of multidetector scanner that enables 2D and 3D reconstruction with a better localization of the lesions.
Side branch IPMNs can arise from native and ectopic pancreatic parenchyma and can have a macrocystic or a microcystic appearance: In our case both lesions developed in ectopic pancreatic parenchyma and manifested as cyst-like structures in cluster with dilated wall ducts most easily visualized in portal venous phase, with a microcystic pattern [2].
These imaging aspects can consisted both with a branch duct type IPMN with microcystic pattern, and with a serous cystadenoma. Serous cystadenoma represents approximately 20% of cystic lesions of the pancreas, occurring in females in about 75% of cases [9], the typical imaging appearance is the microcystic form consisting of a cluster of several small cysts, each less than 1 cm in diameter, often in a honeycomb configuration, separated by fibrous septa [5]; sunburst calcifications of the central scar may be present in about 30% of cases [10].
For differentiating IPMNs from other cystic neoplasms, EUS-guided FNA with cyst fluid analysis can be useful [11], but unfortunately in our case it was not diagnostic.
The indication for surgical treatment of IPMNs is based on symptoms and malignant potential of the lesions: Consensus guidelines recommend surgical resection for all symptomatic lesions, for all side branch IPMNs greater than 3 cm in diameter, as well as for raise in CA 19.9 [12], and all these features were present; moreover the lesions in our case showed amorphous calcifications, reported to be associated with invasive carcinoma (sensitivity of 33%, specificity of 77%) [3].
Besides the IPMNs in ectopic pancreas, our patient presented gastric duplication and intestinal malrotation. Duplications of the gastrointestinal tract are rare congenital anomalies that can occur in any segment of the digestive tract, often in the ileum, rarely in the stomach and duodenum [13]; gastric duplication is the rarest of all gastrointestinal duplications, accounting for less than 4% of cases [14] and can be morphologically classified into two types: Cystic and tubular, with the cystic type more common [14]. They are mostly located along the greater curvature [13,14], and may communicate or not with the stomach, in most cases (85%), as in our patient, non-communicating [10]. The duplication cyst is lined by gastric mucosa, often with patches of ectopic intestinal epithelium, but also by jejunal, ileal, colon or pancreatic epithelium [12], sharing vascular system and muscular wall with the stomach.
In about 38% of cases, gastric duplication is associated with other congenital abnormalities, such as other duplications of the gastrointestinal tract, aberrant pancreas, as in our case, esophageal atresia, congenital heart disease, diaphragmatic hernia, or, as in our case, it may coexist with a malrotation of the bowel [14].
Intestinal malrotation is the result of abnormal embryological development: It is defined as any deviation of the normal rotation of the bowel around the superior mesenteric artery, with an incidence of 1/200-1/500 living birth, asymptomatic in most cases [15]. It is classically considered as a pediatric disease; but it can present in adulthood with an acute bowel obstruction, midgut or cecal volvulus, duodenal obstruction or internal herniation, but in many patients it remains asymptomatic, incidentally recognized during imaging examinations, as in our patient [16]. Various anatomic configurations can result from rotation anomalies of the bowel, with nonrotation as the most common variant. Most commonly, the small bowel remains on the right side of the abdomen, while the caecum is on the left, due to the absence of a ligament of Treitz [17], and this is the aspect we have seen in our patient.
So, in conclusion we present a very unusual case of double IMPNs with foci of adenocarcinoma arising from ectopic pancreatic tissue in periduodenal and perigastric site, coexisting with two congenital anomalies, gastric duplication and intestinal malrotation. The presence of these uncommon conditions all together makes this case very singular. In this case the nonspecific symptomatology may be misdiagnosed; therefore the CT played a major role in the identification of IPMN [18], and, due to the possibility of multiplanar and maximum intensity projection reconstructions, provided precise anatomical information for the surgical workup.
I certify that there is no actual or potential conflict of interest in relation to this article.