We report a case of a 36-years-old female with known Fibrosing Mediastinitis (FM) suspected from prior Histoplamosis infection who presented for medical induction of first pregnancy. The case centers on the outpatient and peripartum management of pregnant patients in the setting of FM with consideration given to method of delivery and anesthetic options during delivery.
Fibrosing mediastinitis, Pregnancy, Pulmonary arterial hypertension, Functional pneumonectomy
Fibrosing Mediastinitis (FM) is a rare disorder of the lungs that can be idiopathic, associated with various granulomatous disorders, and most commonly is related to an inflammatory response to prior Histoplasma infection [1-3]. FM can affect any age, with a range of patients from 13 to 65 years of age reported in the literature but has strong predilection for young females [4]. There has been no reported ethnic predilection [5]. FM is characterized by invasive, calcified fibrosis centered at locations of lymph nodes, often resulting in occlusion of major vessels and airways. Patients present with symptoms such as fatigue, dyspnea, hemoptysis, and pleuritic chest pain. The disease is unpredictable in its course and can result in either spontaneous remission or chronic exacerbations. Various treatment modalities have been tried including systemic anti-fungal, corticosteroids, immunomodulators, and surgery with no current standardized therapy.
The following is a case of a known FM patient, a young African-American female, who became pregnant with a focus on the decisions and treatment options for delivery.
36-years-old female with past medical history of fibrosing mediastinitis (FM) presented for medical induction of first pregnancy. Patient was followed closely outpatient by Pulmonology and Maternal Fetal Medicine (MFM). Pulmonology first saw the patient at 19 weeks gestation. At the time, she was having no symptoms of dyspnea or functional intolerance. During this visit, previous studies were reviewed. Transthoracic echo three years prior showed normal right ventricular size/function. CT Chest three years prior showed auto-amputation of right-sided pulmonary vasculature with right lower lobe and right middle lobe airway collapse. Pulmonary function testing prior showed reduced Diffusion Capacity (DLCO) (52%) and no obstructive/restrictive disease (Figure 1 and Figure 2).
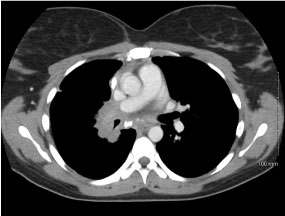 Figure 1: CT chest with IV contrast mediastinal window, axial view. View Figure 1
Figure 1: CT chest with IV contrast mediastinal window, axial view. View Figure 1
 Figure 2: CT chest with IV contrast lung window, axial view. View Figure 2
Figure 2: CT chest with IV contrast lung window, axial view. View Figure 2
At her initial pulmonary outpatient visits, she began serial Transthoracic Echo (TTE) every 4-5 weeks to monitor for pulmonary hypertension (PH) in the setting of increased intravascular volume with pregnancy and known loss of right sided pulmonary vasculature. Repeat CT Chest was deferred given stable pulmonary symptoms and risk of radiation to fetus. Patient continued to follow with Pulmonology and MFM throughout pregnancy with stable right ventricular size and function on TTE.
At follow-up at 37 weeks gestation, she was noted to have moderate respiratory distress with upper respiratory tract infection symptoms. Given the relative severity of her condition and early term gestation, the decision was made between MFM and Pulmonology to admit the patient for respiratory optimization prior to delivery. She was started on Azithromycin for presumed bronchitis with improved respiratory symptoms at rest but still remained more short of breath than baseline when supine. Sputum culture and Respiratory Viral Panel were negative with respect to causative agent. A multidisciplinary discussion between Pulmonology, Anesthesia, and MFM was held to discuss optimal method of delivery (vaginal vs. C-section), induction of labor vs spontaneous delivery, and safety of various anesthetic modalities. The patient, herself, expressed a desire for vaginal delivery.
For our patient, it was felt that vaginal delivery would allow for less respiratory compromise as she had significant orthopnea when lying flat, making surgical positioning difficult. There was also concern that C-section could worsen her respiratory status post-operatively due to potential for increased pain as compared to vaginal delivery. It was decided to delay any induction of labor until patient's respiratory status was back to baseline and acute infection had fully resolved. Should C-section be required urgently, plan was for slow dosed epidural opioids for pain control to minimize respiratory effects of opioids. General anesthesia was considered and rejected as it could cause increased cardio-respiratory depression. It could also cause increased positive pressure which leads to a decreased preload to the right ventricle and increased systemic afterload leading to increased pulmonary vascular resistance and right heart strain. The patient was discharged with a five-day course of Azithromycin and re-admitted at 38 weeks gestation for induction of labor.
Oxytocin was started on admission with anticipated vaginal delivery. Epidural was placed about 12 hours after induction started. Six hours after epidural placement, fetal tracings showed early decelerations. Oxytocin was stopped and patient was prepped for urgent C-Section. She was propped up on pillows during surgery and given O2 via face-mask. She was given slow epidural and local anesthesia for pain control. A healthy baby boy was delivered with Apgar scores of 9-9.
Patient did experience significant pain post-operatively. She was given small doses of oral Oxycodone and intravenous Morphine with good pain control and no resulting respiratory depression. Patient also experienced transient blood pressure elevations post-operatively which corrected with low dose intravenous Hydralazine. This was initially thought to be secondary to pain, but there was also concern for gestational hypertension vs. pre-eclampsia. Resulting lab work showed no evidence of proteinuria. Patient was monitored closely with her asymptomatic mild hypertension but ultimately the rest of the patient's hospital course was benign. She experienced no immediate post-operative cardio-respiratory complications. She was discharged in stable condition on post-partum #3 with a small dose of Nifedipine ER for systemic hypertension. This was discontinued at follow-up as patient's blood pressure had returned to normal levels.
The current plan was for patient to return to Pulmonology clinic for consideration of further imaging to re-evaluate status of FM.
Few studies exist to guide management in pregnant women with FM. The decisions in this case were guided by statements for pregnancy in the setting of pulmonary arterial hypertension (PAH) and prior case reports of pregnancy in the setting of pneumonectomy. Our patient was followed closely during pregnancy for PH and right heart dilation or dysfunction because of known mortality risk of right heart failure (RHF) in pregnancy [6-9].
There is a known link between FM and the development of PH [10]. The exact incidence of PH secondary to FM is unknown, but some studies have shown up to half of FM patients also have PH [11]. Management of PH secondary to FM is difficult as this subtype of PH is typically caused by mechanical compression of lobar or segmental arteries [11]. PH secondary to FM is classified as WHO group 5. No clear clinical improvement has been demonstrated with PAH therapy and steroids are effective only on a case by case basis for improving FM [12]. There are few reports of using pulmonary angioplasty or surgical bypass to resolve pulmonary arterial compression. However, these procedures, despite having low complication rates, are associated with limited durability and often required repeated nonsurgical interventions to maintain patency [5]. PH complicating FM is associated with a poor prognosis (70% alive after 3 years with PH and > 95% alive after 3 years without PH) [12].
The most significant effect of pregnancy on the cardiovascular system is an increase in plasma volume with steady increase during pregnancy and a peak increase of 30-50% during the third trimester [9]. The heart itself is stretched and dilated in all cardiac chambers causing increased resting heart rate by 15-20 beats/minute [13]. Cardiac output (CO) increases significantly (up to 40% above baseline) reaching peak around 20-28 weeks [13]. This is likely secondary to the Frank-Starling response from increased volume and possibly from direct inotropic effect of sex hormones [6]. The systemic vascular resistance (SVR) and pulmonary vascular resistance (PVR) decrease as a consequence of hormone related vasodilation [11]. Thus, with increased CO, and decreased SVR, the mean arterial pressure (MAP) and mean pulmonary arterial pressure (mPAP) stay roughly unchanged in patients without pulmonary vascular disease [6]. Oxygen consumption increases by 20% during pregnancy [13].
The management of pulmonary arterial hypertension (PAH) in pregnancy is still an evolving field of evidence. The current recommendations are to avoid pregnancy with use of contraceptive agents and to strongly consider termination of pregnancy should it occur [6-9]. There exists a high mortality rate in PAH secondary to right heart failure both during and after delivery, with the highest risk occurring during the peripartum period (between weeks 20-24) and the immediate postpartum period [6]. This is secondary to auto-transfusion of blood from the placenta and slow mobilization of third-space fluid which can cause dramatic right heart failure. Immediately following delivery there is a rise in cardiac output by 60-80% secondary to increased blood in circulation [13]. The normal physiologic response of the pulmonary vasculature to dilate with increased fluid is decreased or absent causing right ventricle strain and ultimately RHF [6-9].
Current recommendations, if patient proceeds with pregnancy, are planned, early Cesarean section as preferred mode of delivery with epidural or spinal-epidural anesthesia as opposed to general anesthesia [6-9]. It is recommended to monitor PAH patients during labor with central access, continuous O2, telemetry, and arterial lines [6-9]. Swan-Ganz catheters do not have a clear role in the management during labor.
There are few recent studies of post-pneumonectomy pregnant patients and their delivery course. One study of women from 1947-54 with pneumonectomy, secondary largely to TB and bronchiectasis, showed that these patients had a nearly normal obstetric history and normal life expectancy [14]. The study noted that women with prior pneumonectomies who were functionally able to complete activities of an "ordinary housewife" were fit for pregnancy. Another study in 1967 noted that pneumonectomy did not "prove troublesome" during pregnancy or the delivery period [15]. The study followed 19 women with prior pneumonectomies (primarily for TB) who, over the course of 22 years, had 34 uneventful pregnancies. Lung function was followed throughout pregnancy and noted that even women with < 1L of vital capacity were able to carry fetuses to term without complications.
With respect to FM, management for pregnancy in the gestational and peripartum period will largely be an individual decision due to lack of guiding evidence. Frequent screening TTE to assess for PH and RHF in the gestational period would seem integral as the discovery of either would change management significantly regarding delivery method, anesthesia method, and volume management. CT Chest could be used to detect functional pneumonectomies. However, as most historical studies show no associated morbidity or mortality in pregnant women with actual pneumonectomies, it would be reasonable to defer CT imaging in the gestational period.
Methods of delivery for pregnant FM patients also would be an individual decision. If no evidence of PH is found during the gestational period, it would be reasonable to attempt vaginal delivery with early, slow epidural anesthesia to limit respiratory complications. A multi-disciplinary approach with Pulmonary, Critical Care Medicine, Maternal Fetal Medicine, and Anesthesia is recommended.
FM is a rare disease with no standardized therapy. Pulmonary hypertension, secondary to occlusion of pulmonary vasculature, is a known complication. There is clear morbidity and mortality in PH patients who become pregnant, but few case reports exist that discuss FM in the setting of pregnancy. The patient in this case was managed effectively with close outpatient follow-up and frequent screening TTE. Her delivery plan was for vaginal delivery with epidural anesthesia; however, her course was complicated by fetal decelerations. An effective contingency delivery plan was put in place and she underwent C-section with epidural placement. She suffered no peri-operative or immediate post-operative complications, and she delivered a healthy child.