Brunner's gland adenoma is a rare benign tumour of the duodenum. Less than 200 cases have been reported in the literature. Usually asymptomatic, these lesions rarely manifest as upper gastrointestinal haemorrhage or duodenal obstruction. We report a 40-year-old gentleman who presented with melaena and iron deficiency anaemia. Duodenoscopy revealed a large pedunculated tumour in the duodenal bulb. Due to the huge size, the patient underwent surgical polypectomy through laparotomy and duodenotomy. Histological examination confirmed it to be Brunner's gland adenoma. Endoscopic and radiological features are illustrated along with a literature review of this rare, yet important and easily overlooked cause of gastrointestinal haemorrhage.
Brunner's gland adenoma, Gastrointestinal haemorrhage, Brunneroma
Brunner's gland adenoma also known as Brunneroma, is a harmartoma of the brunner's glands-branched acinotubular glands situated in the submucosa of the first part of duodenum which neutralizes acid chyme by its alkaline secretion. They are usually asymptomatic, found incidentally at endoscopy. Occasionally, they cause gastrointestinal haemorrhage or duodenal obstruction necessitating surgical excision. Endoscopic polypectomy is the treatment of choice, though large size, as in our case may require open surgical polypectomy.
A forty-year-old male presented with 2-3 episodes of malaena of few days duration. There was no associated significant past medical history. Clinical examination was normal, apart from pallor. Routine blood investigations revealed Hb of 8.0 gm/dl and ferritin level of 2.0. Upper gastrointestinal endoscopy revealed a smooth polypoidal tumour arising in the duodenal bulb (Figure 1), biopsies of which was reported as normal. Small bowel meal revealed a smooth polypoidal filling defect in the first part of duodenum (Figure 2).
`
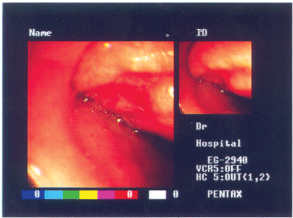 Figure 1: Upper gastrointestinal endoscopy revealing smooth polypoidal tumour arising in the duodenal bulb. View Figure 1
Figure 1: Upper gastrointestinal endoscopy revealing smooth polypoidal tumour arising in the duodenal bulb. View Figure 1
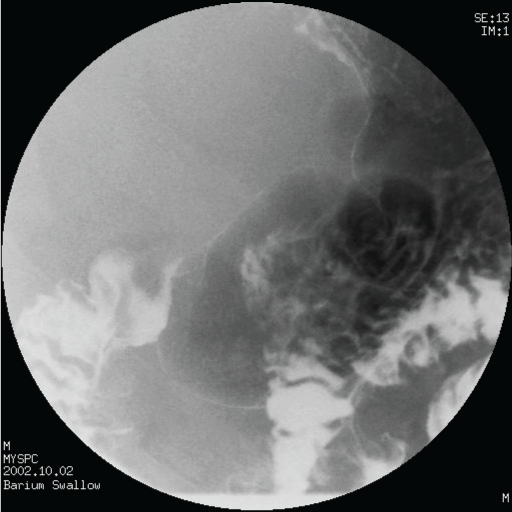 Figure 2: Small bowel meal showing a smooth polypoidal filling defect in the first part of duodenum. View Figure 2
Figure 2: Small bowel meal showing a smooth polypoidal filling defect in the first part of duodenum. View Figure 2
At laparotomy, a soft mobile swelling was palpable within the first part of duodenum. Duodenotomy revealed a 5 × 3 cm sized pedunculated polypoidal growth arising from the mucosa (Figure 3). It was excised and the patient made an uneventful recovery. At 1 year follow up, the patient was completely asymptomatic with no further episodes of malaena.
 Figure 3: 5 × 3 cm polypoidal growth at duodenotomy. View Figure 3
Figure 3: 5 × 3 cm polypoidal growth at duodenotomy. View Figure 3
The cut surface of the tumor had a grayish color, revealing multiple cystic spaces and histopathology examination revealed submucosa with numerous Brunner's glands in a lobular architecture surrounded by bundles of fibromuscular and connective tissue, confirming the diagnosis of Brunner's gland adenoma.
Brunner's gland adenomas (BGA) are rare benign tumours arising from the Brunner's glands of the duodenum and account for about 10.6% of benign duodenal tumors [1]. Since the first reported cases by Curveilhier in 1835 and Salvioli in 1876, less than 200 cases of BGA have been reported in the literature [2]. The first case of a Brunner's gland adenoma was described in 1835 by Cruveilhier [1,3] who presented a woman who died from intussusception secondary to obstruction.
Histologically, Brunner's glands are branched acinotubular glands located in the submucosa. They produce alkaline secretion which empty into the Crypts of Lieberkuhn in the duodenum and protect the duodenal mucosa from the damaging action of acid chyme [4]. They also secrete an enteric hormone - enterogastrone, which is an inhibitor of gastric acid secretion. Thus, it is thought to play an important role in duodenal resistance to ulcer formation.
The distribution of these glands is mostly proximal to the ampulla of Vater, with most of them being just distal to the pylorus. Brunner's gland adenomas are most commonly located in the duodenal bulb, with the second and third portions of the duodenum being much less affected. Usually small in size (1-2 cm), only a few cases more than 4 to 5 cm in size have been reported in the literature [5]. Lesions less than 1 cm represent hyperplasia, whereas those more than 1 cm are called adenoma.
The exact pathogenesis of BGA still remains unclear. Some have suggested that the Brunner's glands are stimulated to undergo hyperplasia by increased acid secretion [6]. Studies suggest that concurrent H. pylori infection is very common in patients with BGA, but it's role remains unclear [7]. The present view is that BGA represent hamartomas in the duodenum. They may be sessile or pedunculated. Benign, proliferative changes of the brunner's gland are very rare and account for about 10% of duodenal bulb neoplasias. They are occasionally associated with gastric hyperchlorhydria or chronic pancreatitis. Malignancy seems to occur only very rarely, with only 14 cases reported in the literature [8].
Patients with BGA usually present in the 4th to 6th decade of life. Most are asymptomatic or present with nonspecific, vague abdominal pain or discomfort, nausea, or bloating. These patients are diagnosed incidentally in the form of small polypoid lesions at barium examination or at endoscopy. Atypical presentations include jaundice, obstruction, and gastrointestinal bleeding. Anemia results from intermittent chronic blood loss secondary to ulceration from the mucosa overlying the mass. Uncommon manifestations include palpation of an abdominal mass, obstructive jaundice, biliary fistula, recurrent pancreatitis and intussusception [5,9].
The diagnosis is usually made by radiological studies followed by upper gastrointestinal endoscopy, which can also provide definitive treatment. Barium studies may reveal a duodenal filling defect, seen as a “vacuole sign” - smooth-walled, polypoid-filling defects, sessile or pedunculated, in the bulb or corresponding portion of the duodenum [2]. Endoscopy demonstrates the tumor in the duodenal lumen and facilitates biopsy from the lesion, which is usually negative owing to the submucosal location of the tumor. Endoscopic ultrasound helps to establish a preliminary diagnosis depending on the layer from which the lesion arises and also from the characteristic appearance of the lesion. It can also detect involvement of the surrounding structures [10]. Computed tomography can be used to delineate the extraluminal extent of large adenomas. Differential diagnosis includes leiomyoma, lipoma, carcinoid tumor, lymphoma, aberrant pancreatic tissue, prolapsed pyloric mucosa, and foreign body. Since bleeding associated with this condition is not of high volume, a contrast enhanced CT is usually not helpful in pinpointing the source of bleed and these lesions are best described as heterogeneous with variable echogenicity [11].
Endoscopic or surgical removal of the adenoma has been suggested in symptomatic patients to prevent development of complications. Local excision of the lesion is the main treatment and may be done by either endoscopic snare-cautery technique or excision of the mass via duodenotomy. As the majority of BGA are quite small, endoscopic polypectomy will confirm the diagnosis and cure the condition in most cases [12]. However, if the size and the anatomic location makes endoscopic removal more difficult or impossible, excision via duodenotomy must be performed. Diffuse involvement may require partial resection of the duodenum.
BGAs are rare benign tumours and usually asymptomatic. Symptoms are often due to obstruction or ulceration of the lesion within the duodenum. Total removal of the lesion leads to minimal morbidity and eliminates the risks of complications.
Nil.