Extrapulmonary forms of tuberculosis (TB) are on the rise in sub-Saharan Africa and pose a major public health problem. The spine is the most frequent location of musculoskeletal tuberculosis. Involvement of the spine causes severe back pain and weakness in the lower extremities. We report 11 cases of TB spondylodiscitis, commonly referred to as Pott's disease, who presented to the internal medicine department at the Military Hospital of Gabon (HIA OBO).
This is a case series of eleven patients with Pott's disease, who presented to the Military Hospital of Gabon between March 2015 and October, 2016. The objective of this article is to highlight the clinical and physical findings of patients presenting with tuberculous spondylodiscitis and describe the medical treatments available in our hospital.
We reviewed the charts of eleven patients with spondylodiscitis. The age range was from 23 to 63 years old. 5 patients were male, 6 were female. Data extraction focused on clinical presentation, blood parameters, and diagnostic findings. The duration of the treatment varied from 9 to 12 months depending on severity of the disease.
Bone biopsy remains a critical diagnostic tool to optimize and confirm the diagnosis of Pott's disease and rule out other causes of spondylodiscitis.
Spondylodiscitis, Pott's disease, Tuberculosis, TB treatment
Tuberculosis (TB) is endemic in sub-Saharan Africa and Asia [1]. It is caused by the Mycobacterium tuberculosis and most commonly affects the lungs. However, in TB endemic countries, extrapulmonary presentations are reported frequently and include spinal tuberculosis, ganglionic tuberculosis, and urogenital involvement also known as Pott's disease. Diagnosis and treatment of Pott's disease are often delayed because initial signs and symptoms are nonspecific in nature. According to the literature, the average time between the onset of first symptoms and diagnosis ranges between two and six months [2].
Pott's disease is the most frequent osteo-articular tuberculosis in endemic areas. Because it is a rare manifestation, most case series were conducted over two years [3,4]. However, the incidence of spondylodiscitis infectious in the world has doubled and the new challenge is the outcome of patient after infection [5].
Physical symptoms were a good indicator for diagnosis in low- and middle-income countries where most of national laboratories are not yet equipped to perform culture and molecular biology. Magnetic resonance imaging (MRI) is a helpful diagnostic tool [6].
Gabon is one the TB endemic countries in Central Africa, where multidrug resistant TB (MDR-TB) was reported with a rate of over 4% [3]. The main challenge is the diagnosis of all tuberculosis forms using culture, imaging and drug susceptibility testing [6,7]. Pott's disease is mainly diagnosed based on symptoms as imaging is not available in all countries because of financial difficulties. Furthermore, differential diagnosis is difficult because physicians are not trained to perform bone biopsies. We present a case series of spondylodiscitis at the department of internal medicine in the "Hôptal d'Instruction des Armées" Omar Bongo Ondimba (HIA OBO) of Libreville, Gabon.
We describe eleven patients who developed tubercular spondylodiscitis which affected cervical, thoracic, and lumbar vertebrae, responded well to treatment, and resulted in partial or total recovery.
The objective of this article is to describe the symptomatic presentation, clinical diagnostics, and medical treatment of spondylodiscitis in our practice.
The Hôpital d'Instruction des Armées in Libreville, Gabon, is a university hospital structure (NATO level 4), comprised of nine departments and 170 beds. In 2015, we registered 50150 outpatients and 6792 inpatients, with a turnover of 58.39%. We take care of the military and their families (20%) and there is also a care of the civilian population (80%).
It is a retrospective, monocentric study of eleven patients, spread from March 2015 to October 2016 in the Internal Medicine Service of HIA OBO. The internal medicine department houses 32 hospital beds; taking care of post-emergency internal medicine, infectious and tropical diseases, cardiology, nephrology and pneumonology.
Each of the eleven patients came to our unit for acute or subacute dorsalgia and/or low back pain. The presumptive diagnosis of Pott's disease was retained on the epidemiological, clinical, biological inflammatory maker and morphological aspects.
The epidemiology aspect is based on the fact that Gabon is part of endemic area for tuberculosis. All our patients benefit from a complete clinical exam. We emphasized on neurology and rheumatology full exam, to highlight dorsalgia, low back pain or neurological deficit.
The biological inflammatory marker we use is the C-reactive protein (CRP) in the blood sample. We consider as an acute inflammation any CRP above 5 mg/L before anti-tuberculosis treatment and a negative CRP after chemotherapy (CRP < 5 mg/L).
The morphological exams we used for spondylodiscitis diagnostics were MRI (1.5 Tesla) and/or Computerized Tomography scan (CT scan). In our military hospital we used a 1.5 tesla for MRI and 64 barrettes for CT scan. All patients underwent medical examination every 3 months, a biological tolerance assessment throughout the duration of the treatment; a clinical and morphological evaluation at the end of the treatment.
A total of eleven patients have been retained for this study. The mean age of the patients was 48.5 years and ranged from 23 to 63 years of age. Six patients were female and five patients were male. All patients were originally from Gabon, without any recent tuberculous exposure. One patient had bifocal, vertebral and lymph node involvement.
The period between onset of symptoms and diagnostic confirmation was on average 6 months. Clinically, spinal pain was the main symptom in all cases. Neurological deficits such as dysesthesia and decreased motility was noted in 5 of 11 patients. We did not record any paraparesis (Table 1).
Table 1: CRP levels (mg/L), chronic diseases and symptoms. View Table 1
The existence of an acute inflammatory marker in association with clinical symptoms was present in almost all patients with chronic diseases; with an average C-reactive protein (CRP) level of 59.5 mg/L (maximum 252 mg/L) before treatment (Table 1 and Figure 1). None of our 11 patients developed immune restoration syndrome (IRIS).
 Figure 1: Clinical signs and C-reactive protein (mg/L) levels. View Figure 1
Figure 1: Clinical signs and C-reactive protein (mg/L) levels. View Figure 1
Spondylodiscitis and HIV-1 coinfection were found in 3 of the patients (all female), two of which had a CD4 count greater than 500/mm3. Five patients had cardiovascular risk factors such as hypertension, one patient had high blood pressure and type 2 diabetes, and two patients suffered from chronic alcoholism. One patient had post-HBV cirrhosis complicated by hepatocellular carcinoma and arterial hypertension (Table 1).
All patients received a chest x-ray. In addition, all 11 patients had MRI (1.5 Tesla) or CT (64 Barrettes) imaging performed at diagnosis.
Six of our eleven patients (54.5%) had low lumbar lesions, three had thoracic lesions (27.3%), and one patient had a cervical lesion (9.1%) and one a thoracolumbar (9.1%) lesion (Table 2).
Table 2: Analysis of bone lesions found on CT or MRI for each of the 11 patients. View Table 2
Regarding lumbar lesions, CT scan without injection in sagittal sections showing spondylodiscitis (Figure 2), with L2-L3 disc nip, mirror erosions performing anterior osteolysis of the vertebral body and osteocondensation of the vertebral body of L3; thickening of the prevertebral soft tissues (Figure 2A) and L2-L3 disc narrowing, discrete kyphosis, L2 body erosions, L3 upper vertebral plateau collapse, and L3 vertebral body osteocondensation (Figure 2B).
 Figure 2: CT scan of the lumbar spine without injection in sagittal sections showing spondylodiscitis.
Figure 2: CT scan of the lumbar spine without injection in sagittal sections showing spondylodiscitis.
A) L2-L3 disc nip, mirror erosions performing anterior osteolysis of the vertebral body and osteocondensation of the vertebral body of L3; thickening of the prevertebral soft tissues; B) L2-L3 disc narrowing, discrete kyphosis, L2 body erosions, L3 upper vertebral plateau collapse, and L3 vertebral body osteocondensation. View Figure 2
We also identified acute phase of spondylodiscitis with medullary MRI (T2 sequence) (Figure 3). The CT shows spondylodiscitis with abscess of the vertebral bodies D10 and D11, opacity of the lower half of the vertebral body of D11 which is partially destroyed, destruction of vertebra D12 and vertebral compression of L1 responsible for kyphosis deformation; paravertebral density with posterior epidural reaction and compression of the medullary cone (Figure 3A). The other CT shows a density of the D7-D8 disc and mirror erosions of the vertebral plates responsible for an enlargement of the disc (Figure 3B). Apart from bone involvement, the other tuberculosis sites were mainly pleural, pulmonary, splenic and ganglionic.
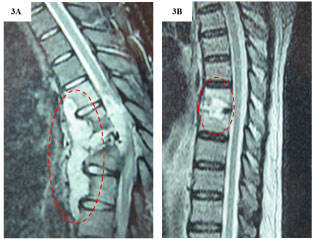 Figure 3: Medullary MRI (T2 sequence) acute phase of spondylodiscitis.
Figure 3: Medullary MRI (T2 sequence) acute phase of spondylodiscitis.
A) Spondylodiscitis with hyperintense abscess of the vertebral bodies of D10, D11, hyposignal of the lower half of the vertebral body of D11 which is partially destroyed, destruction of vertebra D12 and vertebral compression of L1 responsible for kyphosis deformation; paravertebral hypersignal collection with posterior epidural reaction and compression of the medullary cone; B) Hypersignal of the D7-D8 disc and mirror erosions of the vertebral plates responsible for an enlargement of the disc. View Figure 3
We used a non-surgical approach and treated, according to the standard protocol, with 4-drug chemotherapy for 3 months, then two drug therapy for an average duration of 9 to 12 months; and seven out of eleven patients received a 9-month antituberculosis treatment (Table 3). There was no increase in neurological lesions throughout the course of treatment. Most of the patients were followed as outpatients, only 3 patients required hospitalization due to analgesic treatments.
Table 3: Duration of tuberculosis treatment. View Table 3
One patient was able to benefit from a medical evacuated to Morocco and had a bone biopsy which confirmed the diagnosis of Pott's disease. He also received surgical treatment in addition to medical treatment.
At the end of treatment, all patients had improved. None of the patients died. Only one patient had residual cruralgia.
An average of 9 to 12 months depending on the severity of the clinical and morphological signs.
In our study, the diagnosis of infectious spondylodiscitis was made using clinical presentation, CRP level, and CT and MRI imaging.
As reported previously in the literature, the patients suffered from dorso-lumbalgia, low back pain with dysesthesia and/or lumbo-sciatalgia with moderate intensity [8-10].
The CRP averaged 59.5 mg/l with a maximum of 252 mg/l before treatment. After chemotherapy all our patients had a negative CRP. These results are consistent with others studies which looked at erythrocyte sedimentation rate and CRP before, during and after medical and surgical treatment of spinal tuberculosis. The authors found an average CRP of 44.3 mg/l before treatment in 67 patients [11,12]. In another study of 53 patients with lumbosacral tuberculosis, Jiang, et al. found a CRP average of 12.09 mg/dl [13]. This finding indicates that a high CRP level is an important disease activity parameter and a good biological marker for Pott's disease before and after treatment.
In 54.5% the tuberculous spondylodiscitis was found in the lumbar spine, in 27.3% in the thoracic spine, and in 1% in the cervical spine. Similar statistics are found in the literature. Lumbar sites account for approximately 50% of spondylodiscitis, thoracic spinal involvement for 40%, and cervical involvement occurs in only 1% of patients [14-16].
The spine scan allowed us to evaluate the bone abnormalities during the initial management and post-therapeutic follow-up. Kouamé, et al. took a similar diagnostic approach. Eighteen patients received a spine scan allowing initial diagnosis and biopsy under guided scan [4]. However, the literature proposes a systematic Magnetic Resonance Imaging (MRI) in the event of strong suspicion of infectious spondylodiscitis [17]. The advantage of MRI scanning lies in the fact that it makes it possible to identify radicular or medullary compressions [17]. It also makes it possible to identify acute lesions of spondylodiscitis as it shows focal infiltrate of perilesional edema, affecting one or two adjacent trays with the appearance of hyposignal T1, hypersignal T2 and enhancement after injection of gadolinium. Late spondylodiscitis lesions may result in bone loss of the underlying bone blade with vertebral infiltrates limited to the vertebral plateaus and appear as strips or affect the entire vertebral body [6,17].
However, depending on the technical set-up, some authors propose the use of PET scanners to distinguish between early and late infectious spondylodiscitis lesions [18]. However, although a helpful diagnostic tool, it is still inaccessible in our African region.
In our study of 11 patients, only one patient benefited from a bone biopsy with anatomo-pathological analysis confirming the diagnosis of Pott's disease.
Bone biopsy under guided scanning [4,17] allows for pathological and cytobacteriological analysis. For this reason, biopsy under guided scanning has also been recommended in other the studies [19].
Without the results of the bone biopsy, other causes of infectious spondylodiscitis need to be considered and pathogens such as pyogenic bacteria (Staphylococcus aureus, S. epidermidis, E. coli, Pseudomonas, S. sanguis and S. agalactiae) need to be ruled out [18].
A study carried out in Morocco by Zahoun, et al. with 30 specimens (biopsy and blood cultures) revealed gram positive cocci (75%), in particular Staphylococcus aureus (25%) in culture (BACTEC). The other pathogens were gram-negative bacilli [20]. In addition, Chang-Hua, et al. identified a fungal spondylodiscitis related to Candida albicans in a drug-addicted patient [21].
Currently, the use of MALDITOF for identification of pathogens allows for early diagnosis and therefore fast diagnosis and early treatment [22].
The main chronic diseases associated with spondylodiscitis tuberculosis were cardiovascular disease, high blood pressure ,and/or type 2 diabetes [23]. We also had two patients with chronic alcoholism.
Three of the eleven patients had HIV-1 had a CD4 count greater than 500/mm3. Clinical manifestations of spinal tuberculosis in seronegative patients were compared with those with human immunodeficiency virus (HIV). The authors concluded that clinical presentations of spinal tuberculosis are similar in HIV-positive and HIV-negative patients [24]. Two of our patients were considered immunocompetent [25].
All our patients were diagnosed with spondylodiscitis related to Pott's disease and received standard tuberculosis treatment for an average treatment duration of 9 months. Only one of the patients had a bone biopsy and surgery [2,11,26].
If other infectious agents are involved, pyrogenic spondylodiscitis should be treated with antibiotics for less than or equal to 6 weeks [27].
In case of infectious spondylodiscitis with negative biopsies, an empirical two-drug treatment with Fluoroquinolones and a Betalactam or Fosfomicin [28] might be worth considering.
In summary, in countries where TB is endemic, physicians should suspect infectious spondylodiscitis if the patient is presenting with disabling cervical, thoracic, or lumbar pain accompanied by fever and elevated inflammatory laboratory parameters. Patients who present with these symptoms and lab results should receive a scan or MRI of the spine which can identify early and active lesions of spondylodiscitis, reduce diagnostic delay, and allow for timely and appropriate pharmacological treatment which results in improved prognosis. However, these diagnostic tools cannot confirm a suspect case of spondylodiscitis and the growing number of extrapulmonary tuberculosis of the spine calls for additional diagnostics such as bone biopsies to confirm the diagnosis.
We were interested in this study because it had never been realized in our structure and gives us an overview of the type of lesions that we can observed in case of Pott's disease especially since the treatment is still accessible by the National tuberculosis program through the World Health Organization.
The lack of a certain diagnostic tools such as bone biopsy makes the differential diagnosis in our region more difficult.
We recommend further studies, using MRI imaging and additional diagnostic tools such as bone biopsies to rule out other causes of spondylodiscitis. It would be interesting to determine the actual prevalence of Pott's disease in Gabon compared to other countries.