We present a case of a 68-year-old man who underwent control colonoscopy 10 years after sigmoid resection due to a previous adenocarcinoma. An ascending colon polypectomy was performed by endoscopic mucosa resection and a sessile polyp was resected from the sigmoid colon. Two hours after colonoscopy, the patient complained of a swollen neck, mild dyspnoea without any abdominal pain. Subcutaneous emphysema was detected in his neck and upper thorax. The patient was hemodynamically stable. Plain x-rays showed pneumoperitoneum and pneumomediastinum, besides subcutaneous emphysema. The patient was transferred to the surgical department for further observation. Combined antibiotic therapy was continued along with fasting. Symptoms of peritonitis were not present during observation. Control x-rays showed the amount of air decreased. The patient was treated successfully with conservative therapy. On the 3rd hospital day hematochezia causing anaemia was detected. Control colonoscopy detected no active bleeding signs and no signs of perforation. The base of the sigmoid polypectomy was fixed and could not be resected with colonoscopy. Histology of sigmoid tumor revealed adenocarcinoma and the patient went through sigmoid resection one month later. During surgery a retroperitoneal inflammatory mass was detected next to the sigmoid colon. Certain parts of the colorectal system are secondarily retroperitoneal. Through weaknesses in the bowel wall air may escape into the retroperitoneum during colonoscopy and through anatomical spaces it is possible for the free air to reach the mediastinum and subcutaneous tissues developing emphysema even in the absence of perforation. Patients should be closely monitored if ectopic air appears after colonoscopy and could be treated conservatively with a favourable outcome.
Colonoscopy, Subcutaneous emphysema, Pneumomediastinum, Pneumoperitoneum, Extraperitoneal air
Normally gases appear in particular parts of the human body such as the respiratory tract, paranasal sinuses and gastrointestinal system. When air appears in other regions that could be spectacular such as subcutaneous emphysema, which means air trapped in tissues beneath the skin. But ectopic gas can lead to or can be caused by life-threatening conditions. For example, pneumothorax, which is air between the pleural layers. Gas can turn up in ectopic regions because of infections by gas-producing agents, spontaneous alveolar rupture due to pressure differences, as well as disruption of cutaneous and mucosal barriers [1]. Occasionally these may originate from medical procedures like injections, drainage catheters and endoscopy. Air can travel through anatomical connections of the body and can present symptoms at regions far from the leakage where air escaped. Consequently, even cervical subcutaneous emphysema can be a complication of colonoscopy. In this paper we report a case of subcutaneous emphysema, pneumomediastinum and pneumoperitoneum after colonoscopic polypectomies without any endoscopically observable perforation.
A 68-year-old man live with a prosthetic aortic valve and treated with a vitamin K antagonist was admitted to our department for control colonoscopy 10 years after sigmoid resection due to previous adenocarcinoma. Before the procedure, coumarin was replaced by low-molecular weight heparin. After complete colonic preparation the colon was viewed up to the ileocoecal junction. During colonoscopy a 3 cm long polyp was resected by Endoscopic Mucosa Resection (EMR) in the ascending colon (Figure 1). On the base of this area, a deeper mucosal hiatus was clipped. Furthermore, an approximately 6 cm long sessile polypus was detected in the sigmoid colon, which was extracted in pieces. No complications were detected through the examination. Two hours after colonoscopy the patient complained of a swollen neck, with pain in his neck, as well as mild shortness of breath without any abdominal pain. On physical examination crepitus was palpated in the neck and upper thorax of the patient, clearly signs of subcutaneous emphysema. Breathing sounds were normal. On physical examination the abdomen was palpable, soft, tenderness was not detected and normal bowel sounds were heard. The patient was hemodynamically stable, conscious, and afebrile. Chest and abdominal x-rays showed 26 mm wide left subdiaphragmatic air. Air was also detected in the mediastinum continuing to the aorta, as well as cervical subcutaneous emphysema (Figure 2). Initial laboratory parameters were in the normal range, C-reactive protein was 3.4 mg/l, and white blood cell count was 7020/μl. As perforation was suspected, 400 mg b.i.d. ciprofloxacin and 500 mg t.i.d. metronidazole combined intravenous antibiotic therapy was started immediately. The patient was transferred to the surgical department for further observation. Because symptoms of peritonitis were not present and inflammatory laboratory tests remained in a normal range, conservative therapy i.e. combined broad spectrum antibiotics and a fasting regimen was continued. On control x-rays, the amount of air decreased everywhere. On the 3rd hospital day, hematochezia causing anaemia was detected. Control colonoscopy detected no active bleeding signs and no signs of perforation. In the ascending colon, clear base of mucosectomy with clips were seen. A 3 cm large remnant polyp was seen in the sigmoid colon, the base of the previous sigmoid polypectomy was fixed and could not be resected with colonoscopy. After transfusion the patient was stable. Histology of the sigmoid tumor revealed adenoma with high grade dysplasia with focal carcinoma that was limited to the mucosal layer. Because of this finding, the patient went through open sigmoid resection one month later. During surgery a retroperitoneal inflammatory mass was detected next to the sigmoid colon. The excised specimen was histologically diagnosed as villous adenoma with high grade dysplasia. No residual tissue was identified at the resection margins. He was discharged fully recovered. Oncological follow-up of the patient will be continued.
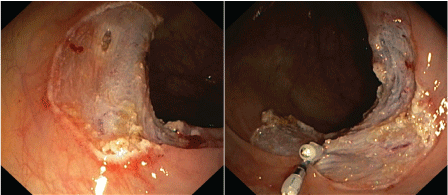 Figure 1: In ascending colon a 3 cm long flat polyp was resected by endoscopic mucosa resection with piecemeal technique. A deeper mucosal hiatus was closed with two haemoclips. View Figure 1
Figure 1: In ascending colon a 3 cm long flat polyp was resected by endoscopic mucosa resection with piecemeal technique. A deeper mucosal hiatus was closed with two haemoclips. View Figure 1
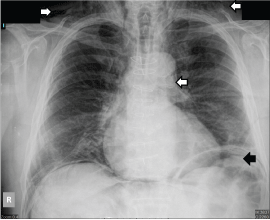 Figure 2: Chest x-ray showing subcutaneous emphysema, subdiaphragmatic air and pneumomediastinum (see arrows). View Figure 2
Figure 2: Chest x-ray showing subcutaneous emphysema, subdiaphragmatic air and pneumomediastinum (see arrows). View Figure 2
Gases ordinarily appear only in the respiratory and gastrointestinal systems of the human body. If aerous materials are situated in other parts of the body they are named after their localisation. Subcutaneous emphysema means that air is trapped in the lower layers of the skin. On the neck it can be seen after dental procedures, trauma of air-containing structures, foreign bodies, infections of the soft tissue leading to necrotising fasciitis of the head and neck, such as dental abscess [2]. When air appears in the mediastinum it is called pneumomediastinum. Trauma of large airways, when a pressure difference causes the rupture of alveoli or oesophageal perforation originating from medical procedures, Boerhave syndrome, foreign body or tumours could lead to pneumomediastinum [2]. Intraperitoneal air, pneumoperitoneum can be seen after laparoscopy or laparotomy, gynaecologic causes, peritonitis with gas-producing germs and any condition which can induce perforation of intraperitoneal bowels [3]. Retroperitoneal gas could originate from an emphysematous infection, organ perforation, or residual air after retroperitoneal surgery [2]. Moreover there could be seen pneumopericardium, pneumoscrotum, pneumothorax as well. One common point in these entities that they can emerge as a complication of colonoscopy. Mostly case reports can be found about this complication in the literature [4-7]. Most of them present patients with perforation. Perforation could be caused by different methods, pneumatic, mechanical and are associated with therapeutic procedures such as polypectomy, endoscopic mucosa resection or endoscopic submucosal dissection [8]. Pneumatic and mechanical perforation appear mainly at weaknesses of the bowel i.e. diverticula, inflammatory or neoplastic diseases, or recent bowel operation [8]. Moreover risk factors can be older age, previous stroke, diabetes mellitus, and cardiovascular diseases [9].
Air can escape from the bowel system without visible perforation as well. Herniation of the colonic mucosa can develop and the wall may become permeable to gases [7]. Perforation could manifest as intraperitoneal, extraperitoneal, and very rarely both. Intraperitoneal perforation could induce peritonitis with abdominal pain, tenderness, vomiting, fever, elevated white blood cell count, absence of bowel sounds, and pneumoperitoneum. Pneumoretroperitoneum, pneumomediastinum, pneumoscrotum, subcutaneous emphysema suggest extraperitoenal rupture. Gas can reach the retroperitoneum directly, via mesentery which is now considered to be an independent retroperitoneal organ or pneumatosis coli meaning intramural gas of the intestine [2]. Elevated intraabdominal pressure due to colonoscopy helps this escape. Along facial planes air can travel from the retroperitoneum to the mediastinum. Outer layer of the chest and abdominal cavity, i.e. endothoracic and transversal fasciae are continuous at the lumbocostal arches, the aortic and oesophageal hiatus, and behind the diaphragm [2]. Another route can be congenital or acquired fenestrations in the diaphragm which can drive air to the chest cavity [10]. Pneumomediastinum can be derived from the peritoneal cavity, too, and air may transit over the oesophageal hiatus. From the mediastinum, air can reach the neck because certain cervical spaces communicate directly with the mediastinum. Subcutaneous tissues have no blockade thorough the body and have low resistance to expansion; therefore emphysema here can affect adjacent areas such as the face or upper chest.
In our case, the location and size of ectopic gas was uncommon, the patient had no obvious perforation, and the accurate origin of the emphysema cannot be identified. From the ascending colon, which is a secondarily retroperitoneal organ, air could reach the retroperitoneum directly over the weakened mucosa at the spot of the implemented EMR. In the intraperitoneal sigmoid colon, polypectomy was carried out and the patient had a previous sigmoidectomy, which are also predisposing factors, both of which lessening the strength of the intestinal wall. Small perforation after EMR could not be excluded completely, however, on second total colonoscopy done a few days later; perforation was not detected at any part of the colon. During subsequent surgery no abnormality was detected at coecum, retroperitoneal inflammatory mass was seen next to the sigmoid colon. This gave rise to suspicion that here was a perforation or microperforation. However, no perforation was identified in excised sigmoid colon during histological analysis. This inflammation could originate from intraluminal site caused by endoscopic treatments. The source of peritoneal air remained also undetected in the presented case, both treated bowel segments are in connection with the intraperitoneal space.
Perforation secondary to colonoscopy can be managed non-surgically as well as operatively. Conservative treatment alone and closure with endoscopic clips could be chosen in patients in good general condition without any sign of peritonitis and if the perforation is recognized early [11]. This means 4 hours according to ESGE guideline on iatrogenic endoscopic perforation, provided the bowel is clean after proper preparation. Radiological examinations, principally a CT scan, are also recommended to prevent diagnostic delay. Therapeutic consideration also depends on the size of discontinuity and expertise of the endoscopist. Every patient, irrespectively of certified perforations or without obvious discontinuity should be treated with infusions, intravenous broad spectrum antibiotics, nothing by mouth regimen, pain medication on demand, and close multidisciplinary monitoring. In case of any suspicion for perforation, conservative therapy is recommended. If any sign of peritonitis is seen immediate surgical intervention should be done.
Colonoscopy is a widely used diagnostic and therapeutic medical procedure and is considered to be safe, however, complications may occur. The presented case suggests that colonoscopy can cause even distant complications, namely pneumomediastinum or subcutaneous emphysema in the neck and air can escape from the bowels without any noticeable perforation. Therapeutic colonoscopy increases its opportunity. In the management of iatrogenic perforation due to colonoscopy, endoscopic and conservative therapeutical methods are safe and effective, and may be chosen as a first line choice instead of surgery in many conditions.