International Journal of Diabetes and Clinical Research
An Oral Antioxidant Formulation Delaying and Potentially Reversing Canine Diabetic Cataract: A Placebo-controlled Double-masked Pilot Study
David Williams1* MA Vet MB PhD, Angus Fitchie1 MA VetMB MRCVS and Carmen Colitz2 DVM, PhD, DACVO
1Department of Veterinary Medicine, University of Cambridge, UK
2All Animal Eye Care, Inc, Jupiter, Florida, USA
*Corresponding author:
David Williams, MA VetMD PhD FRCVS, Department of Veterinary Medicine, University of Cambridge, Madingley Road, Cambridge, UK, E-mail: dlw33@cam.ac.uk
Int J Diabetes Clin Res, IJDCR-2-023, (Volume 2, Issue 1), Clinical Trial; ISSN: 2377-3634
Received: January 02, 2015 | Accepted: February 21, 2015 | Published: February 24, 2015
Citation: Williams D, Fitchie A, Colitz C (2015) An Oral Antioxidant Formulation Delaying and Potentially Reversing Canine Diabetic Cataract: A Placebo-controlled Double-masked Pilot Study. Int J Diabetes Clin Res 2:023. 10.23937/2377-3634/1410023
Copyright: © 2015 Williams D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: The majority of dogs with diabetes mellitus develop blinding mature cataracts through the action of the enzyme aldose reductase producing sorbitol with osmotic action drawing water into the lens thus causing opacification. Here we evaluate the use of OcuGLO� a formulation including the aldose reductase inhibitor alpha lipoic acid, grapeseed extract, carotenoids, omega-3-fatty acids, and coenzyme Q10 in the prevention of canine diabetic cataract in a prospective placebo-controlled double-masked study.
Materials and methods: Dogs with diabetes mellitus but as yet without the development of blinding diabetic cataracts were given either OcuGLO� or a placebo containing antioxidant vitamins. Dogs were examined monthly and their degree of lens opacification documented photographically using a Genesis D fundus camera at +10D. Time to progression of lens opacification was documented and compared between the OcuGLO� group and the placebo group, using Kaplan Meier survival curve statistics
Results: Mean time without change in lens opacification was 278 � 184 days with OcuGLO Rx� and 77 � 40 days in the placebo group this difference being statistically significant at p=0.0005. Twelve of 15 dogs taking the placebo developed significantly increased lens opacification while 5 of 15 dogs taking OcuGLO Rx� developed significant cataract. of these five dogs four animals did not receive daily OcuGLO Rx� as directed due to unrelated concurrent illness or owner non-compliance. The remaining dog progressed despite Ocu-GLO Rx� administration. In two dogs, diabetic cataract was reversed with regained vision on Ocu-GLO Rx�.
Discussion: This small preliminary study demonstrates that oral Ocu-GLO Rx� has beneficial effects in delaying cataract formation in dogs with diabetes mellitus. We look forward to further studies with larger case populations but note that the statistical significance reached between placebo and supplement-treated group, even with a small study population, demonstrates the efficacy of this commercially available dietary supplement.
Introduction
Cataract formation is a widely recognised complication of diabetes mellitus in the dog [1]. Lens opacification in diabetes is suggested to occur through three mechanisms. The first and most important in the dog is the accumulation of polyols, sugar alcohols and most particularly sorbitol, produced through the action of the enzyme aldose reductase (AR) and resulting in an osmotic stress and ingress of fluid into the lens, causing cataract [2]. While this development of lens opacity is generally considered to be rapid, changing a clear lens to a mature cataract within hours or days, and occurring bilaterally symmetrically in both eyes, these assumptions may not necessarily be correct, formed through the fact that most ophthalmologists will only see diabetic dogs when they have become blind. In fact, initial changes in lens opacity prior to the development of mature cataract may occur in diabetic dogs with equatorial vacuoles and cortical opacities occurring in a substantial number of cases, and not necessarily occurring bilaterally symmetrically. Similar changes are seen in dogs fed galactose [3], but the cataract formation may be more severe in this experimental model, since the osmoticon produced by AR from galactose, galacticol, is not further metabolised to fructose by an enzyme equivalent to sorbitol dehydrogenase. Inhibition of aldose reductase has been shown to prevent galactose and diabetic cataract in a number of experimental models [4-6], and in some cases reversal of diabetic cataract on use of sorbinil, an aldose reductase inhibitor, has been reported [7].
The other two mechanisms involve non-enzymatic glycation of lens proteins [8] and oxidative stress [9], which may itself occur through the competition between AR and glutathione reductase for NADPH leading to a depletion in reduced glutathione (GSH), a key intralenticular antioxidant. The part played by these mechanisms in the canine lens is at present unknown.
It has already been demonstrated that administration of the topical AR inhibitor Kinostat appears to inhibit the development of cataracts in diabetic dogs, but this product is not as yet commercially available and does not have these prophylactic effects in every treated dog [10]. Here we seek to show that a commercially available product, Ocu-GLO Rx�, which includes the AR inhibitor alpha lipoic acid, together with a set of antioxidant and free radical scavenging moieties already shown to have beneficial effects in reducing cataractogenesis, is effective in preventing formation of diabetic cataracts in dogs and even in reversing diabetic cataract in a small number of animals. Anecdotal evidence was already available suggesting that Ocu-GLO Rx� did prevent cataractogenesis in diabetic dogs but clearly a double-masked placebo-controlled study was required to confirm these findings in a controlled manner.
Alpha lipoic acid is a potent antioxidant, a potent metal chelator and scavenger of hydroxyl radicals, hypoclorous acid and singlet oxygen species. It also functions to regenerate oxidized glutathione, coenzyme Q10 and vitamins C and E to their reduced forms [11-16]. The compound�s reduced form, dihydrolipoic acid, acts as a scavenger of superoxide and a potent inhibitor of lipid peroxidation. Alpha lipoic acid has been shown to have beneficial effects in several models of diabetes, namely the streptozocin-induced diabetic cataract in rats [17]. It has also been shown to act as an AR inhibitor [18] although with a lower activity than previously investigated AR inhibitors such as sorbinil.
Grapeseed extract contains proanthrocyanins, oligomers and polymers of polyhydroxyfalavan-3-ols such as catechin and epicatechin, present as polyphenols in red wine and in grape seeds. These compounds have strong antioxidative and free radical scavenging activity and have anti-cataractogenic effects in diabetic rats [19], in rats with hereditary cataracts [20], and in rats with selenite-induced cataracts [21]. Coenzyme Q10, also known as ubiquinone, is a benzoquinone component of the mitochondrial respiratory chain, in addition to its antioxidant effects. It acts as a free radical scavenger and is widely recognised as a protectant against oxidative stress in a number of systems. Green tea extract contains epigallocatechin gallate (EGCG) a catechin with acts as a powerful antioxidant and has been shown in a mouse model to modify progression to the diabetic state [22,23] as well as to alleviate oxidative damage in the rat eye [24] and inhibit cataract formation in the rat streptozotocin-induced diabetes model [25,26].
Given the importance of oxidative stress in cataractogenesis, and the central role played by AR in the formation of diabetic cataracts, the combination of antioxidant moieties and AR inhibitor activity in Ocu-GLO Rx� can be understood as acting to promote its anti-cataractogenic activity. But is there firm evidence for such effects? Studies to determine the cataract-impeding effects of a number of molecules have produced less than definitive results in the human field, principally because it is difficult to confine a human subject to a specific dietary regime. Epidemiological studies have sought to correlate degree of cataract with dietary inputs in humans with limited success. Interventional studies are notoriously difficult to implement and to interpret, but interestingly in relation to Ocu-GLO Rx� being a mix of different compounds, one of the most successful interventional studies on dietary amelioration of age related cataractogenesis is the REACT (Roche European American Cataract Trial) protocol [27], which demonstrated a significant ameliorative effect on generation of age-related lens opacities in people. While investigations such as the VetCat study [28] failed to show an effect of vitamin E on impeding development of age-related cataract in people, it was dietary regimes requiring a mix of antioxidant products which yielded beneficial results.
In a veterinary field study, the topical AR inhibitor, Kinostat [5], has demonstrated anti-cataractogenic results. In a multicentre trial with 28 dogs receiving Kinostat and 12 receiving placebo, seven dogs (14 eyes) in the placebo group developed mature cataracts, 2 dogs (4 eyes) developed cortical opacities, and 1 dog (2 eyes) developed equatorial vacuoles, compared with 4 dogs (8 eyes) in the Kinostat group developing mature cataracts, 2 dogs (4 eyes) developing cortical opacities, and 1 dog (2 eyes) developing equatorial vacuoles. Here we demonstrate what might be considered even better results with a once daily oral formulation, which has demonstrated even more impressive results than with three times daily topical Kinostat.
Materials and Methods
This prospective placebo-controlled double-masked study compared time to onset and/or progression of cataract in diabetic dogs treated once daily with Ocu-GLO Rx� per os with onset and/or progression of cataracts in diabetic dogs given an oral placebo capsule once daily containing vitamin antioxidants but without alpha lipoic acid, grapeseed extract, carotenoids, or coenzyme Q10.
Dogs involved in the study were client-owned companion animals accessed from five first opinion veterinary clinics visited by DLW in an ambulatory ophthalmology referral service providing consultations at those clinics regularly. Clients gave full informed consent and agreed to give the relevant supplement daily per os and attend consultations monthly at which photographs of lenses would be taken after pharmacological mydriasis, as well as to inform DLW immediately of any change in their dog�s behaviour suggestive of deterioration in visual acuity.
All dogs were being treated with daily or twice daily injections of insulin and were stable and in good health in other respects, their endocrine disease being monitored by their referring veterinarians. Evaluation of the recent medical history in each dog showed that the diabetes was under control, and also that no concurrent disease was present at the time of admission to the study, which might complicate the study. Animals in which laboratory results or clinical findings indicated concurrent disease at the time of commencement of the trial were excluded from the study.
The dogs were given Ocu- GLO Rx� or the placebo per os in a capsule with the capsule size related to the weight of the dog to normalise the dose of antioxidants with regard to the weight of the animal.
The study was reviewed and accepted by the welfare and ethics committee of the Department of Veterinary Medicine, University of Cambridge. Given previous anecdotal evidence that Ocu-GLO Rx� would prevent cataract formation in diabetic dogs and the published successful results of the topical AR inhibitor in Kinostat to do the same [5], the use of a placebo group where lens opacification would be allowed to progress to a mature cataract then requiring surgery was considered unethical, especially as the animals involved would be client-owned dogs. It was thus decided that the end point for the study in each dog would be significant progression of lens opacity, as documented photographically rather than progression to mature cataract. If lens opacification had progressed in any dog in the study, the trial would be stopped for that animal and its treatment group would be unmasked. Were the dog to be on the placebo, it would then be transferred to the supplement arm of the study. In each animal on either arm of the study, time to progression of cataract, where such progression occurred, was used as the endpoint for use in the Kaplan-Meier survival plot statistics and time without cataract progression used where lens transparency did not change.
A power calculation showed that 12 dogs in each arm of the study would yield a valid result with 80% power given that in one year 75% of the animals would be expected to develop cataract and it was anticipated that Ocu-GLO Rx� Rx� would reduce this to 25%. To ensure a statistically valid result, 15 dogs in each arm of the study were chosen as the population size required.
Statistical analysis comparing the times to cataract progression in the two arms of the study was undertaken with the Kaplan-Meier estimator using SPSS software.
Blood samples were taken by the referring veterinarians during the study for blood glucose and fructosamine analysis but samples could not be taken for evaluation of serum alpha lipoic acid levels or levels of other antioxidant moieties, as the study was undertaken under the Veterinary Surgeons Act (1966) and not the Animal Scientific Procedures Act (1988). Therefore, in the UK blood samples could only be taken for the immediate benefit of the animals involved and not to answer a scientific research question.
Results
Signalment of dogs entered into the study is detailed in table 1 for dogs on the supplement and placebo arms of the study respectively. All dogs were diagnosed on the basis of the clinical signs of polydipsia and polyuria, weight loss in the face of polyphagia and laboratory findings of hyperglycaemia with glucosuria. All dogs were being treated with subcutaneous insulin injections. Duration of time since diagnosis of diabetes and fructosamine levels at the beginning of the study is given in table 1. The average length of time the animals had been on the study was 240 � 142 days with a range of between 618 and 48 days.
![]()
Table 1: Signalment of dogs on supplement and placebo arms of study
View Table 1
Six dogs had no signs of lens opacification at the commencement of the study while others had some lens changes such as nuclear sclerosis (16 dogs), equatorial vacuoles (7 dogs) or cortical spokes (19 dogs), some of which (nuclear sclerosis and cortical spokes, for instance) were consistent with age-related changes while others (particularly equatorial vacuoles) were consistent with the lens pathology seen in canine diabetic hyperglycaemia. Details of lens pathology are given in table 2 together with time on the study without change in lens opacification.
![]()
Table 2: Lens pathology at start of trial and time to significant deterioration in lens clarity in dogs on supplement and placebo arms of trial
View Table 2
Mean time without change in lens opacification at the time of writing was 278�184 days with Ocu-GLO Rx� and 77 � 40 days in the placebo group. Median duration without lens change was 261 and 84 days, respectively, this difference being statistically significant at p=0.0003.
Twelve of the 15 dogs (80%) taking the placebo developed significant lens changes while on the study protocol. Five of 15 dogs (33%) taking Ocu-GLO Rx� developed significant cataract, in three of these animals Ocu-GLO Rx� was not being given as directed due to pancreatitis leading to vomiting and thus presumed insufficient supplement intake, and in one dog inability of the elderly arthritic owner to ensure that the dog swallowed the capsule daily, again led to insufficient supplement intake. One animal, apparently taking the capsule daily and in normal health apart from its diabetic state, developed cataract in the face of treatment. Two dogs, Dachshund siblings, one of which presented with lenticular opacity at the beginning of the study and the other of which developed the opacity while on the placebo, both regained vision in their cataractous eye while on Ocu-GLO RxTM, as documented photographically in Figure 1. A Kaplan-Meier survival plot of the data for each arm of the study is shown in Figure 2.
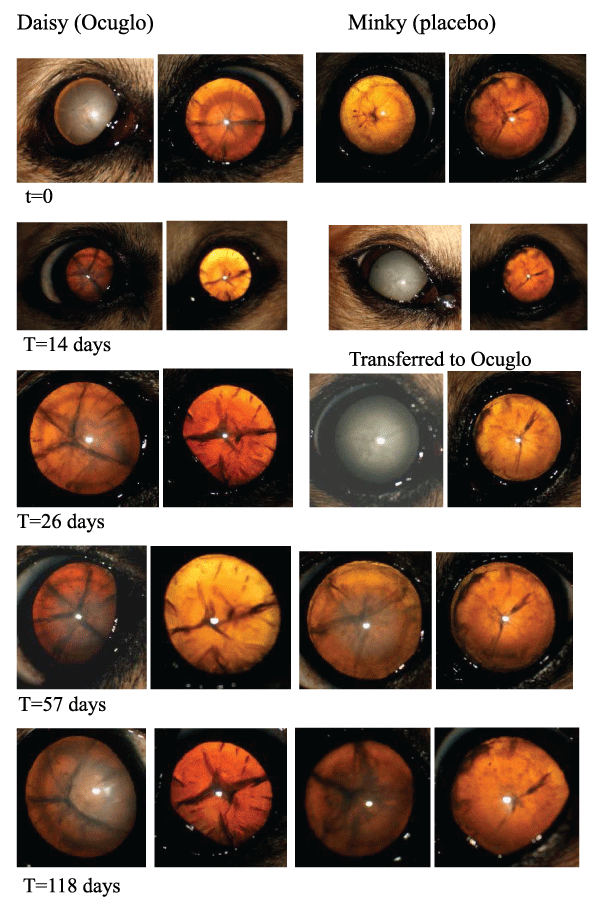
Figure 1: Lens changes in two diabetic dachshund siblings on OcuGLO (left) and placebo (right)
View Figure 1
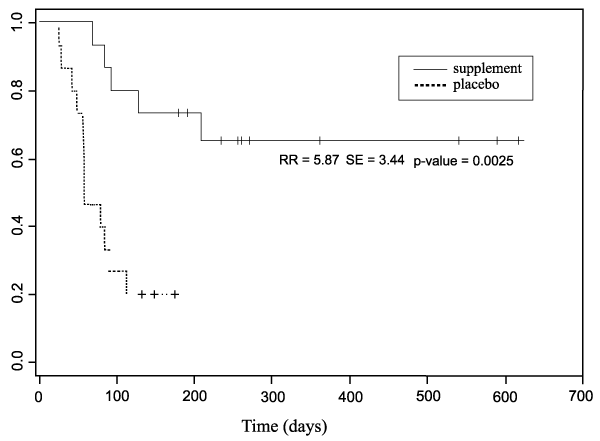
Figure 2: Kaplan Meier survival plot showing proportion of eyes with clear lenses over time in days for population of dogs given supplement (black line)
and those on placebo (dotted line).
View Figure 2
Discussion
Cataract is a well-recognised complication of diabetes mellitus in the dog, with 70% of cases developing sight-threatening lens opacification within 12 months of diagnosis [29]. As noted in the introduction, lens changes in the face of hyperglycaemia are thought to be caused by the action of the enzyme AR, which converts glucose in the lens to sorbitol. Sorbitol is normally produced in the lens to counteract the dehydrating activity of high extracellular glucose during normal physiological hyperglycaemia after eating [30] but in situations of persistent hyperglycaemia, as seen in diabetes for instance, the generation of excess intracellular sorbitol leads to osmotic stress and swelling of lens fibre cells. Recent work in lenses of diabetic rats has questioned whether AR activity is the key player in diabetic cataractogenesis in that species [31]. It is likely that oxidative stress also plays a part in cataract development in diabetes, as it does in the ageing changes seen in the non-diabetic lens.
Here we show that the formulation Ocu-GLO Rx� has a significant beneficial effect in reducing the development of sight-threatening mature cataract in diabetic dogs. It might be argued that this preliminary study involves a small number of dogs, yet the fact that a highly significant difference is seen with only fifteen dogs in each arm of the study demonstrates that the medication has a highly significant effect. We aim to further this study examining the beneficial effect of Ocu-GLO Rx� on a larger number of dogs as clinical cases in a multicentre study to address the small number of animals in the current study.
The formulation Ocu-GLO Rx� has been developed to include both the AR inhibitor and glutathione regenerator alpha lipoic acid, as well as a number of anti-oxidant moieties such as EGCG (containing quercetin), grapeseed extract, various carotenoids (lutein, zeaxanthin and lycopene), and coenzyme Q10 with the aim of reducing oxidative stress in the diabetic lens. It might be argued that such a mix of molecules with differing actions aimed at preventing cataractogenesis in these diabetic dogs does not provide sufficient rigour in allowing dissection of the different molecular actions important in impeding lens opacification. We acknowledge this potential deficit in the current study, which is aimed at assessing the value of Ocu-GLO Rx� as a clinical tool in preventing cataract formation in diabetic dogs rather than specifically as a scientific assessment of the molecular mechanisms of cataractogenesis in diabetic dogs. We may further this study by evaluating the effects of individual agents in this product, but note that the paucity of success in preventing cataract formation in human studies using individual agents has been overshadowed by the favourable outcomes in human trials employing a mixture of agents, such as the Roche European American Cataract Trial (REACT). We suggest that, in a similar fashion here, a mix of agents may be more efficacious than the sum of its parts.
The decision not to have progression to full blinding lens opacification as the end point might similarly be questioned. Using total blindness with a mature cataract as the endpoint might been seen as a more definitive endpoint, but as discussed above, it was decided that an endpoint of significant change in lens opacification should be used with dogs on placebo being transferred to the active treatment arm of the trial. This has the advantage of showing that Ocu-GLO Rx� appears to prevent further cataract formation in diabetic dogs where early changes such as equatorial vacuoles or cortical spokes have occurred.
Using client-owned animals might be seen as problematic in that owners may have supplemented their animals with other products or with Ocu-GLO Rx�, but since in the UK the product was not available at the time of the study and all owners agreed not to supplement their dogs other than with the trial formulations, this is unlikely to have confounded the study. The fact that a range of dog breeds was involved might also complicate the study if some breeds are more likely to develop denser lens opacities as is seen between human patients of different ethnicities, but we have no evidence currently that diabetic cataract in different dog breeds varies in a similar manner.
In conclusion this study provides preliminary data demonstrating that Ocu-GLO Rx� impedes cataractogenesis in diabetic dogs. Further studies to enlarge the case population will be necessary and are in the planning stages, but to have obtained a highly significant difference between dogs on the supplement and those on placebo even at this small sample size may be taken as a preliminary demonstration of the efficacy of this dietary supplement in diabetic cataract in the dog. The importance of aldose reductase and oxidative changes in the pathogenesis of other diabetic complications in human patients [32] suggests that use of an aldose reductase inhibitor and antioxidant cocktail such as this product may be valuable in human patients quite as much as in dogs.
References
-
Basher AW, Roberts SM (1995) Ocular manifestations of diabetes mellitus: diabetic cataracts in dogs. Vet Clin North Am Small Anim Pract 25: 661-676.
-
Varma SD (1980) Aldose reductase and the etiology of diabetic cataracts. Curr Top Eye Res 3: 91-155.
-
Sato S, Takahashi Y, Wyman M, Kador PF (1991) Progression of sugar cataract in the dog. Invest Ophthalmol Vis Sci 32: 1925-1931.
-
Kador PF, Lee JW, Fujisawa S, Blessing K, Lou MF (2000) Relative importance of aldose reductase versus nonenzymatic glycosylation on sugar cataract formation in diabetic rats. J Ocul Pharmacol Ther 16: 149-160.
-
Griffin BW, McNatt LG, Chandler ML, York BM (1987) Effects of two new aldose reductase inhibitors, AL-1567 and AL-1576, in diabetic rats. Metabolism 36: 486-490.
-
Banditelli S, Boldrini E, Vilardo PG, Cecconi I, Cappiello M, et al. (1999) A new approach against sugar cataract through aldose reductase inhibitors. Exp Eye Res 69: 533-538.
-
Beyer-Mears A, Cruz E (1985) Reversal of diabetic cataract by sorbinil, an aldose reductase inhibitor. Diabetes 34: 15-21.
-
Hashim Z, Zarina S (2011) Advanced glycation end products in diabetic and non-diabetic human subjects suffering from cataract. Age (Dordr) 33: 377-384.
-
Williams DL (2008) Oxidative stress and the eye. Vet Clin North Am Small Anim Pract 38: 179-192, vii.
-
Kador PF, Webb TR, Bras D, Ketring K, Wyman M (2010) Topical KINOSTAT™ ameliorates the clinical development and progression of cataracts in dogs with diabetes mellitus. Vet Ophthalmol 13: 363-368.
-
Zicker SC, Hagen TM, Joisher N, Golder C, Joshi DK, et al. (2002) Safety of long-term feeding of dl-alpha-lipoic acid and its effect on reduced glutathione:oxidized glutathione ratios in beagles. Vet Ther 3: 167-176.
-
Busse E, Zimmer G, Schopohl B, Kornhuber B (1992) Influence of alpha-lipoic acid on intracellular glutathione in vitro and in vivo. Arzneimittelforschung 42: 829-831.
-
Singh U, Jialal I (2008) Alpha-lipoic acid supplementation and diabetes. Nutr Rev 66: 646-657.
-
Kozlov AV, Gille L, Staniek K, Nohl H (1999) Dihydrolipoic acid maintains ubiquinone in the antioxidant active form by two-electron reduction of ubiquinone and one-electron reduction of ubisemiquinone. Arch Biochem Biophys 363: 148-154.
-
Suh JH, Shenvi SV, Dixon BM, Liu H, Jaiswal AK, et al. (2004) Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc Natl Acad Sci U S A 101: 3381-3386.
-
Bast A, Haenen GR (2003) Lipoic acid: a multifunctional antioxidant. Biofactors 17: 207-213.
-
Kojima M, Sun L, Hata I, Sakamoto Y, Sasaki H, et al. (2007) Efficacy of alpha-lipoic acid against diabetic cataract in rat. Jpn J Ophthalmol 51: 10-13.
-
Brownlee JM, Carlson E, Milne AC, Pape E, Harrison DH (2006) Structural and thermodynamic studies of simple aldose reductase-inhibitor complexes. Bioorg Chem 34: 424-444.
-
Yamakoshi J, Saito M, Kataoka S, Tokutake S (2002) Procyanidin-rich extract from grape seeds prevents cataract formation in hereditary cataractous (ICR/f) rats. J Agric Food Chem 50: 4983-4988.
-
Sasaki T, Yamakoshi J, Saito M, Kasai K, Matsudo T, et al. (1998) Antioxidative activities of 4-hydroxy-3(2H)-furanones and their anti-cataract effect on spontaneous cataract rat (ICR/f). Biosci Biotechnol Biochem 62: 1865-1869
-
Zhang X, Hu Y (2012) Inhibitory effects of grape seed proanthocyanidin extract on selenite-induced cataract formation and possible mechanism. J Huazhong Univ Sci Technolog Med Sci 32: 613-619.
-
Orts�ter H, Grankvist N, Wolfram S, Kuehn N, Sj�holm A (2012) Diet supplementation with green tea extract epigallocatechin gallate prevents progression to glucose intolerance in db/db mice. Nutr Metab (Lond) 9: 11.
-
Wolfram S, Raederstorff D, Preller M, Wang Y, Teixeira SR, et al. (2006) Epigallocatechin gallate supplementation alleviates diabetes in rodents. J Nutr 136: 2512-2518.
-
Chu KO, Chan KP, Wang CC, Chu CY, Li WY, et al. (2010) Green tea catechins and their oxidative protection in the rat eye. J Agric Food Chem 58: 1523-1534.
-
Vinson JA, Zhang J (2005) Black and green teas equally inhibit diabetic cataracts in a streptozotocin-induced rat model of diabetes. J Agric Food Chem 53: 3710-3713.
-
Babu PV, Sabitha KE, Srinivasan P, Shyamaladevi CS (2007) Green tea attenuates diabetes induced Maillard-type fluorescence and collagen cross-linking in the heart of streptozotocin diabetic rats. Pharmacol Res 55: 433-440.
-
Chylack LT Jr, Brown NP, Bron A, Hurst M, K�pcke W, et al. (2002) The Roche European American Cataract Trial (REACT): a randomized clinical trial to investigate the efficacy of an oral antioxidant micronutrient mixture to slow progression of age-related cataract. Ophthalmic Epidemiol 9: 49-80.
-
McNeil JJ, Robman L, Tikellis G, Sinclair MI, McCarty CA, et al. (2004) Vitamin E supplementation and cataract: randomized controlled trial. Ophthalmology 111: 75-84.
-
Beam S, Correa MT, Davidson MG (1999) A retrospective-cohort study on the development of cataracts in dogs with diabetes mellitus: 200 cases. Vet Ophthalmol 2: 169-172.
-
Chylack LT Jr, Tung W, Harding R (1986) Sorbitol production in the lens: a means of counteracting glucose-derived osmotic stress. Ophthalmic Res 18: 313-320.
-
Zhang P, Xing K, Randazzo J, Blessing K, Lou MF, et al. (2012) Osmotic stress, not aldose reductase activity, directly induces growth factors and MAPK signalling changes during sugar cataract formation. Exp Eye Res 101: 36-43.
-
Ramana KV (2011) ALDOSE REDUCTASE: New Insights for an Old Enzyme. Biomol Concepts 2: 103-114.





