International Journal of Neurology and Neurotherapy
Caspase Cleaved Tau in Alzheimer's Disease: A Therapeutic Target Realized
Troy T. Rohn*
Department of Biological Sciences, Science Building, Room 228, Boise State University, USA
*Corresponding author:
Troy T. Rohn, Professor, Department of Biological Sciences, Boise State University, 1910 University Drive, Boise, ID 83725, USA, Tel: 208-426-2396, Fax: 208-426-1040, E-mail: trohn@boisestate.edu
Int J Neurol Neurother, IJNN-2-014, (Volume 2, Issue 1), Hypothesis; ISSN: 2378-3001
Received: January 06, 2015 | Accepted: January 09, 2015 | Published: January 12, 2015
Citation: Rohn TT (2015) Caspase Cleaved Tau in Alzheimer's Disease: A Therapeutic Target Realized. Int J Neurol Neurother 2:014. 10.23937/2378-3001/2/1/1014
Copyright: © 2015 Rohn TT. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Tau; Caspase 3; neurofibrillary tangles; Alzheimer's disease; cleavage
Alzheimer's disease (AD) is a progressive neurodegenerative disorder characterized by an array of symptoms affecting memory and cognition. Some common symptoms of AD include memory loss that disrupts daily life, challenges in planning or solving problems, confusion with time or place, and changes in mood and personality [1]. Central dogma to the etiology of AD is the beta-amyloid cascade, which stipulates that beta-amyloid in oligomeric forms represents the earliest step in a cascade eventually leading to the formation of senile plaques and neurofibrillary tangles (NFTs) and neurodegeneration [2]. For many years the connection between plaques and tangles was unknown, however, in 2002 we reported that caspase activation and the cleavage of tau might link these two molecular entities in AD [3]. Our evidence was based on the synthesis and application of a caspase-cleavage site directed antibody to a known caspase-cleavage site within tau located at the amino-terminus (position 25). Figure 1 depicts the first experiment ever performed with affinity-purified tau caspase-cleavage antibody that revealed widespread labeling predominantly within NFTs, neuropil threads, and dystrophic neurites (Figure 1A) that was absent in age-matched control sections (Figure 1B)
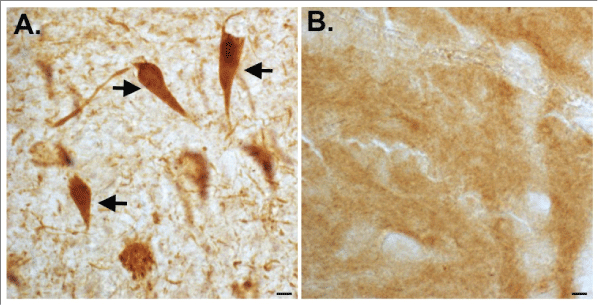
Figure 1: First known demonstration of the caspase-cleavage of tau in
the human AD brain. On October 21, 2001 we performed an immunohistochemical
experiment on hippocampal brain sections utilizing a purified
caspase-cleavage antibody to tau. The results indicated strong labeling in
NFTs of the AD brain (arrows, A), that were absent in age-matched control
sections (B). Scale bars represent 10µm.
View Figure 1
The model we proposed in a subsequent review article was the activation of apoptotic pathways by beta-amyloid leads to the cleavage of tau and promotes the formation of NFTs in the AD brain [4]. Shortly thereafter, two studies largely confirmed our hypothesis by demonstrating that the caspase cleavage of tau is an early event in NFT evolution, and links beta-amyloid to NFT formation in the AD brain [5,6]. Both studies relied heavily on data obtained using identical site-directed antibodies to the C-terminal caspase-cleavage site within tau located at amino acid residue D421 and supported a general role for caspase-3 as being the major executioner protease involved in cleaving tau at this C-terminal site. The conclusion from both studies was that the caspase-cleavage of tau was an early event in AD disease tangle pathology and that caspase-3 may serve as the link between beta-amyloid deposition and the formation of NFTs [7]. Subsequently, several studies have confirmed a key role for caspase activation and the cleavage of tau as a proximal event in promoting tangle pathology [8-14]. Of great value to the field, the antibody developed by Lester Binder's group at Northwestern University was made available commercially and this antibody, known as TauC3 has been instrumental in documenting the role of caspase-mediated truncation of tau in AD. By all accounts, this monoclonal antibody is an excellent antibody that shows no reactivity with full-length tau or other tau C-terminal truncations and is specific for NFTs, and caspase-cleaved tau within neuritic plaques and neuropil threads [12].
Based on the role of caspase-mediated cleavage of tau in promoting NFT formation in AD, blocking this cleavage event may provide a potential therapeutic strategy for the treatment of this disease. Recently Intellect Neurosciences, Inc. acquired the worldwide development and commercialization rights to TauC3 under an exclusive license agreement with Northwestern University. This past year, Intellect Neurosciences announced it obtained proof of concept in a preclinical Alzheimer's model for its TauC3 monoclonal antibody indicating its potential utility as a therapeutic agent. The study was carried out in collaboration with University of California, Irvine's Dr. Frank LaFerla, Director, Institute for Memory Impairments and Neurological Disorders as well as Dr. Kim Green. The results from this unpublished study showed that the TauC3 antibody "effectively engaged the target and reduced phosphorylated pathological forms of Tau indicating that the treatment with the peripherally administered antibody had an effect in the brain and is able to be disease modifying" http://www.prweb.com/releases/2014/1/prweb11489644.htm. Although these findings have not yet been peer-reviewed, if confirmed they provide exciting preclinical data that may be used to formulate human clinical trials in the near future.
The story of caspase-cleavage of tau in AD represents how studying the basic mechanisms underlying a disease can lead to a better understanding of the disease process as well as identifying new drug targets. As an investigator who has spent his entire academic career studying the role of caspases in neurodegenerative diseases, it is gratifying to see the realization of such research from the bench to the clinic.
Acknowledgement
This work was funded by National Institutes of Health Grant 1R15AG042781-01A1.
References
-
Alzheimer's Association (2013) 2013 Alzheimer's disease facts and figures. Alzheimers Dement 9: 208-245.
-
Hayden EY, Teplow DB2 (2013) Amyloid ß-protein oligomers and Alzheimer's disease. Alzheimers Res Ther 5: 60.
-
Rohn TT, Rissman RA, Davis MC, Kim YE, Cotman CW, et al. (2002) Caspase-9 activation and caspase cleavage of tau in the Alzheimer's disease brain. Neurobiol Dis 11: 341-354.
-
Rohn TT, Rissman RA, Head E, Cotman CW (2002) Caspase Activation in the Alzheimer's Disease Brain: Tortuous and Torturous. Drug News Perspect 15: 549-557.
-
Gamblin TC, Chen F, Zambrano A, Abraha A, Lagalwar S, et al. (2003) Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer's disease. Proc Natl Acad Sci U S A 100: 10032-10037.
-
Rissman RA, Poon WW, Blurton-Jones M, Oddo S, Torp R, et al. (2004) Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology. J Clin Invest 114: 121-130.
-
Rohn TT (2010) The role of caspases in Alzheimer's disease; potential novel therapeutic opportunities. Apoptosis 15: 1403-1409.
-
Guo H, Albrecht S, Bourdeau M, Petzke T, Bergeron C, et al. (2004) Active caspase-6 and caspase-6-cleaved tau in neuropil threads, neuritic plaques, and neurofibrillary tangles of Alzheimer's disease. Am J Pathol 165: 523-531.
-
Yin H, Kuret J (2006) C-terminal truncation modulates both nucleation and extension phases of tau fibrillization. FEBS Lett 580: 211-215.
-
Ding H, Matthews TA, Johnson GV (2006) Site-specific phosphorylation and caspase cleavage differentially impact tau-microtubule interactions and tau aggregation. J Biol Chem 281: 19107-19114.
-
Gastard MC, Troncoso JC, Koliatsos VE (2003) Caspase activation in the limbic cortex of subjects with early Alzheimer's disease. Ann Neurol 54: 393-398.
-
Guillozet-Bongaarts AL, Garcia-Sierra F, Reynolds MR, Horowitz PM, Fu Y, et al. (2005) Tau truncation during neurofibrillary tangle evolution in Alzheimer's disease. Neurobiol Aging 26: 1015-1022.
-
Spires-Jones TL, de Calignon A, Matsui T, Zehr C, Pitstick R, et al. (2008) In vivo imaging reveals dissociation between caspase activation and acute neuronal death in tangle-bearing neurons. J Neurosci 28: 862-867.
-
Spires-Jones TL, Stoothoff WH, de Calignon A, Jones PB, Hyman BT (2009) Tau pathophysiology in neurodegeneration: a tangled issue. Trends Neurosci 32: 150-159.





