International Journal of Sports and Exercise Medicine
Feasibility of Focused Cardiac Ultrasound in Pre-participation Screening
Miriam L. Fishman, Jennifer Shea, Brian G. Choi and Allen J. Solomon*
Division of Cardiology, The George Washington University Hospital, USA
*Corresponding author: Allen J. Solomon, MD, Division of Cardiology, The George Washington University Medical Faculty Associates, 2150 Pennsylvania Avenue, NW, Suite 4-420, Washington DC 20037, USA, Tel: 202-741-2323, Fax: 202-741-2324, E-mail:asolomon@mfa.gwu.edu
Int J Sports Exerc Med, IJSEM-1-011, (Volume 1, Issue 2), Original Research; ISSN: 2469-5718
Received: March 30, 2015 | Accepted: May 26, 2015 | Published: May 29, 2015
Citation: Fishman ML, Shea J, Choi BG, Solomon AJ (2015) Feasibility of Focused Cardiac Ultrasound in Pre-participation Screening. Int J Sports Exerc Med 1:011. 10.23937/2469-5718/1510011
Copyright: © 2015 Fishman ML, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Current American Heart Association (AHA) guidelines for pre-participation athletic screening recommend a 12-point history and exam to minimize the risk of sudden cardiac death. We tested the hypothesis that focused cardiac ultrasound (FCU) performed and simultaneously interpreted by a cardiologist using a handheld ultrasound device would be a feasible addition.
Methods: We performed pre-participation screening according to AHA recommendations on high school athletes in a multi-purpose room at their school. In addition to the standard 12-point assessment, a cardiologist simultaneously performed and interpreted a FCU on each athlete using a handheld ultrasound.
Results: The mean age of the athletes was 16.6 ± 3.4 years; 68% were male. No evidence of left ventricular hypertrophy, cardiomyopathy, bicuspid aortic valve, or aortopathy was identified. Coronary ostia could not be visualized. Echocardiography added 1.35 ± 0.51 minutes to the standard exam.
Conclusion: This feasibility study suggests that the addition of handheld echocardiography with real-time interpretation performed by a cardiologist to a standard AHA pre-participation screening adds less than two minutes of time to the assessment. While the study is not as comprehensive as an office based echocardiogram, it can provide valuable information which may be useful in ruling out some of the most common causes of sudden cardiac death in the young athlete or in selecting those who would benefit from further testing.
Keywords
Sports medicine, Echocardiography, Screening, Cardiology prevention
Introduction
While the incidence of sudden cardiac death (SCD) in athletes throughout the country is relatively small, ranging from 1:341,551 participants in high school sports in Minnesota to up to 1:3,100 male NCAA Division I Basketball players, the emotional impact of even one death is devastating not only for the family, but also for the town or university [1,2]. Although we have learned a great deal regarding the various causes of SCD, the data remain scarce for effective preventative measures. The American Heart Association (AHA) recommends pre-participation screening, which consists of a 12 point history and physical exam [3]. It is not necessary that the screening be performed by a physician and the screening has not been shown to impact mortality.
We sought to determine whether the addition of focused cardiac ultrasound (FCU) with real-time interpretation was a feasible addition to pre-participation screening in a multi-purpose room setting. Europe and the International Olympic Committee have mandated inclusion of screening electrocardiograms in pre-participation screening, based on the Italian experience in Veneto, Italy [4]. This study showed that inclusion of screening ECGs decreased the incidence of SCD in Italian athletes by 89% compared to non-screened non-athletes [5]. However, the results of the study are controversial and have not borne out in Israel or when compared to the United States experience [6,7]. A recent cost effectiveness projection of the Italian model to high school and college athletes in the United States suggests that a 20 year program would save 4,813 lives at a cost of 10.6-14.4 million dollars per life saved [8]. In addition to the financial limitations, widespread ECG usage as a screening tool is encumbered by difficulty with interpretation. Furthermore, normal or near-normal ECGs can be seen in 10% of patients with HCM (false negatives) [9].
The routine performance of an echocardiogram is not mandated, except in a relatively few forums. At this point, only 13% of professional sports teams incorporate echocardiography, although the number is higher for basketball [10]. At the NCAA Division I level in football, a group of athletes with a relatively high risk of SCD, only 23% of programs responding to a survey reported screening echocardiography [11]. Feasibility of full echocardiograms on a mass scale is limited by time, space and financial resources.
Development of handheld echocardiography enabling FCU could drastically change the role for screening echocardiograms. Good concordance of the measurements typically required for the diagnosis of hypertrophic cardiomyopathy, as well as assessment of valvular regurgitation have been observed between handheld and standard echocardiography [12,13]. The most recent guidelines on FCU put forth by the American Society of Echocardiography incorporate the use of eFCU - defined as FCU performed by an experienced echo cardiographer - as a reasonable means by which to diagnose hypertrophic cardiomyopathy in an athletic screening [14].
Method
Two cardiologists and one cardiology fellow performed a pre-participation screening in a multi-purpose room at a public high school. Vital signs were obtained by two volunteer registered nurses. The standard AHA 12 point pre-participation history and physical exams were performed. FCU images were obtained by a level II trained cardiologist with a General Electric Vscan handheld echocardiogram (GE Healthcare, Wauwatosa, WI). Vscan is a 1.4 � 7.3 � 2.8cm device weighing 390g with an 8.9cm diagonal, 240 � 320 pixel display (Figure 1). Vscan uses a phased-array transducer (1.7-3.8MHz) with a fixed sector angle of 75� and maximum depth of 25 cm. Color flow Doppler is available within a 30� maneuverable sector. Spectral Doppler or M-mode imaging is not available with the device. Images are stored on the device as JPEG still captures or MPEG-4 cine loops.
Subjects lay supine and parasternal long-axis views were obtained with and without color Doppler. Short axis views were obtained at the level of the aortic valve, mitral valve, papillary muscles and left ventricular apex. Apical windows were obtained in subjects with prior cardiac history or abnormal physical exam. Abnormal findings at point-of-care analysis or off-line analysis would lead to referral of the subject to their pediatrician for full evaluation.
Subsequent off-line analysis of the images was performed on a Vscan Gateway workstation version 1.0.0.12 (GE Healthcare). Measurements of the wall thickness of the interventricular septum and posterior wall of the left ventricle were measured, as well as the dimensions of the left ventricle. Ejection fraction was also calculated. The presence and severity of mitral and aortic regurgitation was assessed. The presence of a bicuspid aortic valve was ascertained and the diameter of the aortic root was measured. Coronary artery origins were not assessed. This protocol was approved by The George Washington University Institutional Review Board.
Statistical analysis
Results are expressed as the mean ± standard deviation or as absolute values or percentages, as appropriate.
Results
A total of 68 athletes underwent Vscan echocardiography. The mean age of the athletes was 16.6 ± 3.4 years and 68% were male. The majority of the high school athletes played soccer (41%), followed by cross country (21%), cheerleading (13%), volleyball (11%), tennis (6.5%), field hockey (4.3%) and golf (2.2%). Echocardiography added only 1.35 ± 0.51 minutes to the standard exam. No abnormalities were noted real-time by the scanning and interpreting cardiologist (Figure 2). Subsequent offline analysis showed that the mean thickness of the septum and posterior wall were 0.69 ± 0.15cm and 0.72 ± 0.13cm, respectively. The mean left ventricular internal dimension (in diastole) was 4.2 ± 0.48cm. No evidence of left ventricular hypertrophy, cardiomyopathy, bicuspid aortic valve, or aortopathy was identified on any of the echocardiograms. Coronary ostia could not be visualized.
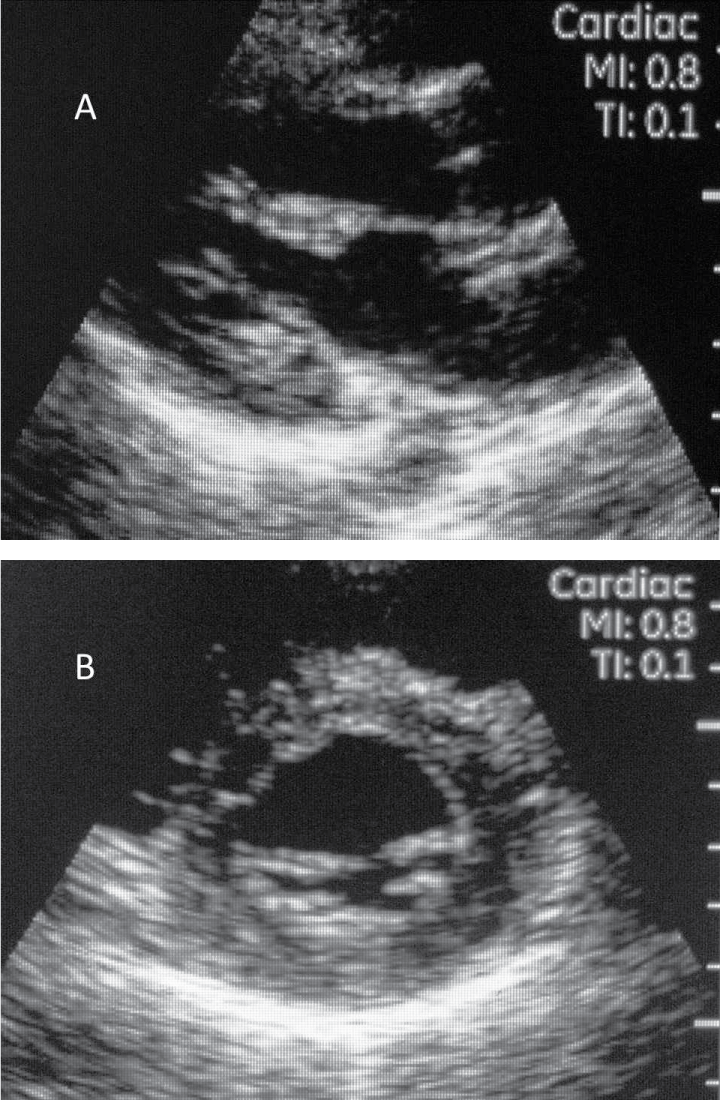
.
Figure 2A & 2B: Parasternal long axis view of the heart.
Short axis view of the heart at the level of the mitral valve.
View Figure 2A & 2B
Discussion
The standard AHA screening exam alone is extremely lacking in sensitivity. Two recent studies found that screening with history and physical alone is associated with a sensitivity of less than 50% which is generally considered unacceptable for a screening test [15,16]. The addition of an electrocardiogram to the standard exam does increase sensitivity (to about 90%), while decreasing specificity (from 76.4% and 94.1% to 69.5% and 82.7%, respectively). It is clear that while an electrocardiogram may strengthen the effectiveness of screening, mass screening requires bulky equipment and can result in unnecessary downstream testing in the setting of more frequent false positive screening results. When a standard screening and an electrocardiogram suggest an abnormality, the next diagnostic test is echocardiography. Incorporating the echocardiogram at the outset, especially in a high risk population, may be a cost effective solution.
There are limitations to the data acquired by echocardiography. First, while hypertrophic cardiomyopathy is the most common cause of sudden cardiac death in the young athlete, it is not the only cause and a normal echocardiogram may cause false reassurance. In addition, cardiovascular exam by FCU is not as complete as standard echocardiography, and should not be considered as a replacement. FCU does not have the capacity for spectral and tissue Doppler, which are crucial for hemodynamic assessment, evaluation of outflow tract obstruction and diastolic function. Diastolic function is one of the key distinguishing characteristics between a physiologically remodeled athlete's heart and hypertrophic cardiomyopathy. Especially without concurrent electrocardiographic testing, we would recommend full standard echocardiography for athletes who screen positive based on the 12 point AHA pre-participation screening or in those with any concerning findings on their FCU.
Cost effectiveness is already debated for screening electrocardiograms. In our case, the FCU was performed and simultaneously interpreted by an experienced cardiologist, volunteering with a group of cardiologists without additional costs beyond the fixed cost of the device. This fulfills the vision of the FCU serving as an extension of the physical exam. The inclusion of the FCU could reduce unnecessary downstream testing, as the echocardiogram is considered the first evaluation subsequent to an abnormal electrocardiogram in pre-participation screening. Currently eFCU is a non-reimbursable procedure requiring an expert to volunteer time and a device - a possibly insurmountable logistical boundary for widespread screening.
Our study demonstrates that it is feasible to incorporate FCU into pre-participation screening. It requires very little additional space and equipment, and can be done in an ordinary room, adding minimal time to the examination. Given that the echocardiogram is so frequently performed in downstream testing, immediate performance and interpretation of echocardiograms in a mass screening setting could add valuable information to the screening process. More research is still needed to assess the efficacy and cost effectiveness of FCU in the screening of the elite athlete at higher risk for SCD.
References
-
Maron BJ, Haas TS, Ahluwalia A, Rutten-Ramos SC (2013) Incidence of cardiovascular sudden deaths in Minnesota high school athletes. Heart Rhythm 10: 374-377.
-
Harmon KG, Asif IM, Klossner D, Drezner JA (2011) Incidence of sudden cardiac death in National Collegiate Athletic Association athletes. Circulation 123: 1594-1600.
-
Maron BJ, Thompson PD, Ackerman MJ, Balady G, Berger S, et al. (2007) AHA Scientific Statement: Recommendations and Considerations Related to Preparticipation Screening for Cardiovascular Abnormalities in Competitive Athletes: 2007 Update. A Scientific Statement from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism. Circ 115: 1643-1655.
-
Corrado D, Pelliccia A, Bjornstad HA, Vanhees L , Biffi A, et al. (2005) Cardiovascular pre-participation screening of young competitive athletes for prevention of sudden death: proposal for a common European protocol. Eur Heart J 26: 516-524.
-
Corrado D, Basso C, Pavei A, Michieli P, Schiavon M, et al. (2006) Trends in sudden cardiovascular death in young competitive athletes after implementation of a preparticipation screening program. JAMA 296: 1593-1601.
-
Steinvil A, Chundadze T, Zeltser D, Rogowski O, Halkin A, et al. (2011) Mandatory electrocardiographic screening of athletes to reduce their risk for sudden death proven fact or wishful thinking? J Am Coll Cardiol 57: 1291-1296.
-
Maron BJ, Haas TS, Doerer JJ, Thompson PD, Hodges JS (2009) Comparison of U.S. and Italian Experiences With Sudden Cardiac Deaths in Young Competitive Athletes and Implications for Preparticipation Screening Strategies. Am J Cardiol 104: 276-280.
-
Maron BJ, Doerer JJ, Haas TS, Tierney DM, Mueller FO (2009) Sudden deaths in young competitive athletes: analysis of 1866 deaths in the United States, 1980-2006. Circulation 119: 1085-1092.
-
Halkin A, Steinvil A, Rosso R, Adler A, Rozovski U, et al. (2012) Preventing sudden death of athletes with electrocardiographic screening: what is the absolute benefit and how much will it cost? J Am Coll Cardiol 60: 2271-2276.
-
Harris KM, Sponsel A, Hutter AM Jr, Maron BJ (2006) Brief communication: Cardiovascular screening practices of major North American professional sports teams. Ann Intern Med 145: 507-511.
-
Coris EE, Sahebzamani F, Curtis A, Jennings J, Walz SM, et al. (2013) Preparticipation cardiovascular screening among National Collegiate Athletic Association Division I Institutions. Br J Sports Med 47: 182-184.
-
Coletta C, De Marchis E, Lenoli M, Rosato S, Renzi M, et al. (2006) Reliability of cardiac dimensions and valvular regurgitation assessment by sonographers using hand-carried ultrasound devices. Eur J Echocardiogr 7: 275-283.
-
Prinz C, Voigt JU (2011) Diagnostic accuracy of a hand-held ultrasound scanner in routine patients referred for echocardiography. J Am Soc Echocardiogr 24: 111-116.
-
Spencer KT, Kimura BJ, Korcarz CE, Pellikka PA, Rahko PS, et al. (2013) Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr 26: 567-581.
-
Magalski A, McCoy M, Zabel M, Magee LM, Goeke J, et al. (2011) Cardiovascular screening with electrocardiography and echocardiography in collegiate athletes. Am J Med 124: 511-518.
-
Baggish AL, Hutter AM Jr, Wang F, Yared K, Weiner RB, et al. (2010) Cardiovascular screening in college athletes with and without electrocardiography: A cross-sectional study. Ann Intern Med 152: 269-275.