International Archives of Nursing and Health Care
Patient Reported Outcomes in a New Home-Based Rehabilitation Programme for Prostate Cancer Patients
Brigitta R Villumsen1*, Martin Grønbech Jørgensen2, Jan Frystyk3, Britta Hørdam4 and Michael Borre5
1Urological department, Regional Hospital Holstebro and Aarhus University, Denmark
2Department of Geriatrics, Aalborg University Hospital, Denmark
3Medical Research Laboratory, Aarhus University & Department of Endocrinology and Internal Medicine, Aarhus University Hospital, Denmark
4Department of Political Science, University of Copenhagen, Denmark
5Department of Urology, Aarhus University Hospital, Denmark
*Corresponding author: Brigitta R Villumsen, Urological department, Regional Hospital Holstebro, Denmark, E-mail: brigvill@rm.dk
Int Arch Nurs Health Care, IANHC-1-022, (Volume 1, Issue 1), Research Article; ISSN: 2469-5823
Received: October 22, 2015 | Accepted: December 21, 2015 | Published: December 26, 2015
Citation: Villumsen BR, Hørdam B (2015) Patient Reported Outcomes in a New Home-Based Rehabilitation Programme for Prostate Cancer Patients. Int Arch Nurs Health Care 1:022. 10.23937/2469-5823/1510022
Copyright: © 2015 Villumsen BR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The most optimal exercise plan for men with prostate cancer (PC) receiving androgen deprivation therapy needs to be identified. We plan to investigate a 12-week home-based health programme (exergaming) on physical function, fatigue and metabolic parameters in this group. In addition, our study will explore the satisfaction and experience with the health game programme. To the best of our knowledge this is the first RCT study to investigate the effect of a home based health game programme on PC patients. No statistical analysis have been made thus far because inclusion is ongoing, however baseline observations show that most of the participants fulfil the criteria for being diagnosed with metabolic syndrome. Finally this study will assess the safety and efficacy of a novel exergaming modality for PC patients and the results are expected to influence recommendations on exercise given to this patient group.
Keywords
Prostate cancer, Androgen deprivation therapy, Home-based exercise, Interactive video gaming, Nursing intervention
Introduction
Men with prostate cancer receiving androgen deprivation therapy complain of fatigue and develop type 2 diabetes and bone fractures. These known adverse effects of the androgen deprivation therapy can be prevented or treated with exercise. Studies of exercise programmes for cancer patients form a growing area of research and involve both independent home-based and supervised exercise programmes. Exercise in those living with and beyond cancer should incorporate both supervised and independent exercise requirements [1]. To the best of our knowledge no studies have investigated home-based exercise for men with prostate cancer using an interactive video gaming console. This study will therefore be able to provide information about a novel exercise modality for this patient group.
To establish a solid basis for this study, a literature review was conducted in the PubMed, Cochrane, Cinahl and PsychInfo databases to establish the magnitude of the problem regarding the adverse effects of androgen deprivation therapy and to what extent exercise can improve these conditions.
Prostate cancer is treated with androgen deprivation therapy combined with curative radiation therapy or androgen deprivation therapy alone if metastatic disease is present at the time of diagnosis [2]. The treatment reduces the production of testosterone but has adverse effects such as hypercholesterolemia, obesity, type 2 diabetes, osteoporosis, loss of muscle mass and muscle strength, decreased quality of life, increased risk of cardio-vascular disease and increased fatigue [3-6]. Some of these adverse effects are part of the metabolic syndrome, and newer studies show that the metabolic syndrome occurs in more than 50 per cent of men receiving long-term androgen deprivation therapy [7]. A significant correlation is seen between the duration of treatment and the degree of insulin resistance, level of fasting glucose and increase in waist circumference [8].
Resistance and aerobic exercises increase muscle mass, muscle strength, cardio-vascular fitness and walking distance; decrease levels of insulin-like growth factor-I (IGF-1); improve quality of life, physical function, suppleness and haemoglobin percentage; and strengthen the immune system [9,10]. Studies also show that less nausea and fatigue, fewer disease symptoms, less fat mass, a lower pulse rate and systolic blood pressure is seen in patients who exercise [11,12]. Therefore resistance and aerobic exercises are successful in preventing the adverse effects of androgen deprivation therapy, but this type of exercise is usually not performed in the home setting. Furthermore gently physical movements such as qigong/tai chi has shown a positive effect on quality of life, fatigue, immune function and cortisol level in cancer patients [13].
There is a need to find the most optimal and individual exercise plan for men with prostate cancer receiving androgen deprivation therapy, and studies investigating home-based exercise are called for to examine the potential of this exercise modality. Furthermore, patients prefer to exercise at home alone, with relatives or friends, and therefore more studies must be conducted in this area. A flexible and spontaneous exercise pace is apparently preferred [14].
Interactive video games can be used to promote home-based exercise. The Nintendo Wii system has been shown to have a positive effect on mood, fatigue and physical function in cancer patients [15,16]. In an older population, an exercise intervention with Nintendo Wii has shown an 18 per cent increase in muscle strength, and the participants were entertained by this type of exercise [17]. The Xbox 360 Kinect interactive video game system gives the person playing more room for free movement and might therefore offer more possibilities than Nintendo Wii. Xbox 360 Kinect can be used for high intensity home-based exercise [18], and has shown positive results on motor function and activities of daily living in a study of chronic stroke patients undergoing rehabilitation [19]. To the best of our knowledge no studies have been published on the use of the Xbox 360 Kinect in the rehabilitation of cancer patients.
Men receiving androgen deprivation therapy need information about the adverse effects of androgen deprivation therapy, the treatment and prevention of it, and how important exercise is. Nurses are in a unique position to support and help patients making health promoting lifestyle changes [4], and physiotherapists can give practical instructions regarding a safe and effective exercise programme [20].
Aim of the study
To determine the efficacy of a 12-week home-based exercise programme on physical function, body composition, quality of life, fatigue and metabolic parameters in patients with prostate cancer receiving androgen deprivation therapy. In addition, we will investigate the satisfaction and experience with the Xbox 360 Kinect interactive videogame system as an exercise tool.
We hypothesise that patients in the intervention group will increase their walking distances, muscle power in the buttocks and thighs, insulin sensitivity and levels of adiponectin. Furthermore, we hypothesise that the intervention group will gain muscle mass and lose fat mass, experience a higher quality of life, less fatigue and decreased levels of cholesterol, fasting glucose and IGF-1.
Methods/design
The study is a pretest-posttest blinded study and has been approved by the Regional ethical committee Central Denmark Region and the Danish Data Protection Agency. Informed written consent is obtained from all participants prior to participation.
The participants will be randomly allocated to either a 12-week home-based exercise programme using the Xbox 360 Kinect system (n = 23) or usual care (n = 23).
Study population
The study population will consist of forty-six men with histologically confirmed advanced or locally advanced prostate cancer receiving androgen deprivation therapy at the urological departments in Central Denmark Region of Denmark. The expected mean age of the study participants is around 68 years of age. Most of the participants are expected to have received curative intended radiation combined with androgen deprivation therapy and the smaller part of participants receive androgen deprivation therapy due to bone- or lymph node metastases. All participants are expected to fulfil some of the criteria for having metabolic syndrome at baseline.
Inclusion criteria
• Male > 18 years old, diagnosed with prostate cancer receiving androgen deprivation therapy for at least 3 months
• Performance status 0-1
• Signed informed consent
• Being able to read and understand Danish.
Exclusion criteria
• Intense aerobic or strength exercise > 2 times pr week for 3 months or more before inclusion in the study
• Pacemaker
• Haemoglobin level ≤ 6.0 mmol/l
• Usage of opioid
• Any mental or physical condition that makes the patient ill-suited for participation
• Hydration-influencing conditions
• Hip- or knee replacement
• Long-term usage of steroids or low-energy fracture due to usage of steroids
• Ongoing participation in other intervention studies
• Curative intended radiation therapy within 3 months from date of inclusion.
Randomisation
After completion of all baseline assessments participants are randomised 1:1 to the intervention or control group. Sealed envelopes are used for this purpose. None of the study team members know which envelopes contain the control group slip and which contains the intervention group slip.
Study assessments
All tests and procedures will be done at baseline and after 12 weeks. Blood samples will be repeated at a follow-up visit 24-26 weeks after baseline.
Primary study end point
The primary study end point is change physical function as determined by walked distance during the six-minute walk test according to standard procedures.
Secondary endpoints
Secondary endpoints include muscle strength, body composition, patient-reported outcomes, metabolic function, blood pressure and waist circumference.
Individual interviews.
Assessment methods
Assessment of functional exercise capacity will be done by the international validated six-minute walk test. The test will be performed according to standard procedure by blinded physiotherapists.
A leg extensor power rig (University of Nottingham Medical School, Nottingham, England) is used to assess muscle power. Participants are instructed to perform a number of submaximal movements as a warm up before the maximal movement. The test will be performed by blinded physioterapists.
The Quad Scan 4000 (Bodystat, British Isles, England) will be used to derive information about body composition by calculating lean body mass and fat mass. The participants will follow instructions from the manufacturer before the test.
A number of questionnaires will be answered to investigate quality of life and fatigue:
The EORTC QLQ-C 30 questionnaire is an international validated instrument for measuring general cancer quality of life. Most of the questions have four response levels (not at all, a little, quite a bit and, very much). Two questions rely on a 7-point scale. Responses are transformed to produce scores on function scales (physical, role, emotional, cognitive and social functioning), and nine symptom scales. One scale represents global quality of life.
The Functional Assessment of Cancer Therapy-Prostate (FACT-P) questionnaire is an international validated instrument to measure quality of life in men with prostate cancer. This 27-item scale measures physical, social/family, emotional, and functional well-being on a 4-point Likert scale (not at all, a little bit, somewhat, quite a bit, and very much). Higher score represents better quality of life.
Fatigue will be assessed using the international validated questionnaire Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F). This 13-item scale measures the level of fatigue in individuals during their usual daily activities. The level of fatigue is measured on a 4-point Likert scale (not at all, a little bit, somewhat, quite a bit, and very much). This scale is commonly used to assess fatigue in cancer patients including cancer patients receiving exercise interventions.
The validated Exercise regulations questionnaire (BREQ-2) is used to identify reasons for people to engage in physical activity or refrain from physical activity. This 19-item scale measures external and intrinsic factors in regulation of exercise behavior.
The international validated Godin Leisure-Time exercise questionnaire is used to report the participants' activity level.
A number of blood samples will be collected to investigate the metabolic status: cholesterol, triglyceride, glucose, insulin sensitivity using the Homeostasis Model Assessment (HOMA), IGF-1 activity by cell based IGF-1 kinase receptor activation (KIRA) analysis, supplemented by assessment of immunological determined IGF-1 level and IGF-binding protein levels. Also adiponectin levels will be determined by immunological analysis.
Furthermore data will be collected on blood pressure, pulse, waist circumference, height, weight, body mass index, smoking habits and alcohol intake.
All tests and procedures will be done at baseline and after 12 weeks. Blood samples will be repeated at a follow-up visit 24-26 weeks after baseline.
To investigate the satisfaction and experience with the Xbox 360 Kinect interactive videogame system individual interviews will be conducted according to an interview guide developed from a literature review. Interviews will be analysed using the constant comparative method to identify categories related to satisfaction and motivation. The interview will be conducted immediately after the 12-week intervention period.
All test procedures and data collection will be conducted at Regional Hospital Holstebro, Denmark. Randomisation will not happen until all baseline procedures have been conducted. Each participant will be given a study-id number to ensure blinding. Only the study coordinator has the identification log with matching study-id numbers.
Treatment arms
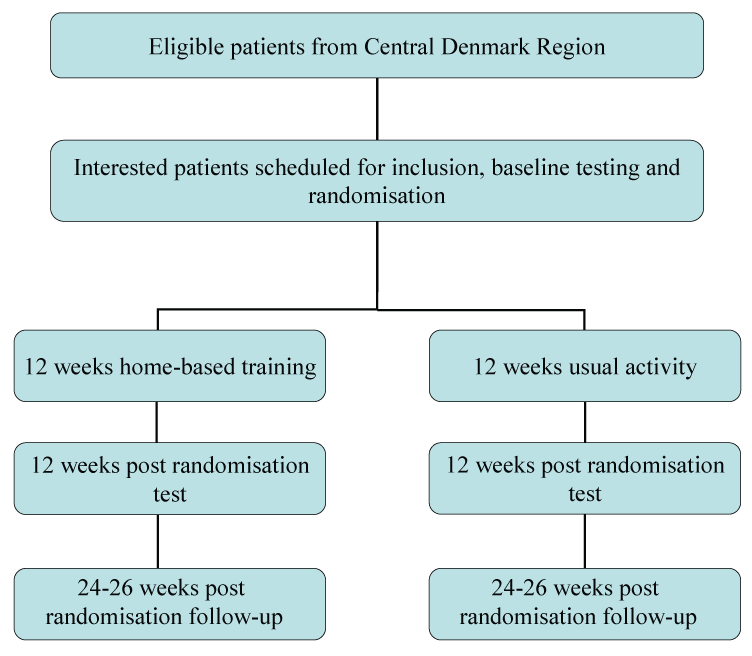
Patients from Central Denmark Region are invited to participate in the study (Figure 1).
Intervention group
Twelve weeks of home-based exercise three times a week in sessions of approximately 60 min using the Xbox 360 Kinect interactive videogame system.
Three video games have been selected for the exercise programme: Adventures, Sports and Your Shape Fitness Evolved. Adventures give the opportunity for entertainment concurrent with exercise. Sports offer a variety of games and give the opportunity to compete against a computer component or other components but also to compete together with other persons in teams. Your Shape Fitness Evolved gives the participant opportunity to do regular fitness training with a personal coach on a television screen.
To gauge progress during the 12 weeks, the intervention group will also use 0.5 kg dumbbells in weeks 1 to 4, 1.0 kg in weeks 5 to 8, and 2.0 kg in weeks 9 to 12.
The intervention group will be divided into smaller groups of 4 to 5 participants who will receive one group lesson as well as two individual lessons by physiotherapists as an introduction to the interactive videogame system and exercise programme.
During the study period the intervention group will keep a training diary to document the training.
Control group
Participants in the control group will be encouraged to maintain their usual activity level during the study. However, for ethical reasons, participants in the control group will only be excluded if they start intense aerobic or strength training > 3 times per week.
During the study period the control group will keep a diary to document activity level.
Statistical considerations
Sample size calculations
To detect a 100 meter increase in the six-min walk test in the intervention group assuming a standard deviation (SD) of 84.5 meter, 23 men are needed in each group when a dropout rate of 30% after randomisation is taken into consideration to reach the significant level and a power of 90%.
Data analysis
Statistical analysis will be performed using the STATA 13 software package. A biostatistician will support the research group in analysing data.
The primary statistical analysis will be a comparison between the two randomisation groups. Treatment duration and activity level at baseline will be adjusted for due to presumed influence of the results.
All data will be described and compared by means of descriptive statistics. The statistical methods will depend on whether data are normal or non-normal distributed and whether data are paired or unpaired. When comparing more than two groups the ANOVA or Bonferroni methods will be used to adjust for multiple comparisons.
Normal distributed data will be described using mean, standard deviation (SD) and 95% confidence interval (CI) while non-normal distributed data will be described using median, 25 and 75 percentiles and range.
All data will be tested at a significance level of 0.05. Correlation analysis is used to detect causality. A regression analysis will be conducted using treatment duration of < / > 12 months as independent variable.
Discussion
The goal of this project is to investigate whether an interactive videogame console provides a useful exercise tool that can help men with prostate cancer overcome their treatment-related side effects.
As this project seems to be the first to investigate this question, the findings will aid in the interdisciplinary rehabilitation of prostate cancer patients because both positive and negative results will be useful.
To the best of our knowledge no other studies have investigated the satisfaction and experience with the Xbox 360 Kinect interactive videogame system as exercise tool in prostate cancer patients receiving androgen deprivation therapy. In this study we hope to make use of the feed-back in every game in the form of a reward, an opportunity for earning points or seeing progress directly on the screen.
The choice of the Xbox 360 Kinect interactive video game system as exercise intervention tool is based on a number of considerations. Firstly, in the clinical setting we see prostate cancer patients who do not like going to a fitness centre, so alternatives are called for. Secondly, some patients are not motivated to do outdoor exercise or do not have enough surplus of mental resources to find an exercise modality that suits them and their life situation. Thirdly, some patients live in remote areas where it may be difficult to find a suitable exercise modality for them. Furthermore, the generation of prostate cancer patients receiving androgen deprivation therapy has grandchildren who grow up with interactive video gaming and who may be a motivating factor for the patients to exercise using interactive video gaming.
During the intervention period, all participants in this study will complete training diaries to compare test results with compliance to the programme and for the participants to be aware of how much or how little they exercise. We are aware that some of the participants in the control group might start to exercise based on the knowledge they acquire due to their participation in the study. The information from the diary can help reveal to what extent this will happen. A research coordinator contacts the participants every fortnight (intervention group) or once a month (control group) to gather information on compliance, adverse events and changes in medication intake.
The follow-up measurements will provide information regarding the sustainability of the exercise programme. Since patients with prostate cancer have a relatively long life expectancy, we would like to investigate whether the intervention leads to long-term changes in metabolic parameters.
Significance of this study
This project is expected to provide knowledge about a novel exercise modality for prostate cancer patients that give them the possibility to exercise together with family and friends at home and at the same time overcome treatment-related side effects.
This project is also expected to provide an indication of the usefulness of interactive video gaming in prostate cancer patients.
The results are expected to be useful for recommendations in evidence-based guidelines because this is the first study to investigate the effect of home-based exercise in prostate cancer patients using interactive video gaming.
Thus the results from this study are expected to influence the information given to this patient group and thereby to have an impact on the future interdisciplinary rehabilitation of these men.
Due to the novelty and methodological strength of this research project, the investigator was awarded at the EAUN (European Association of Urological Nurses) Congress in 2014 and given an unrestricted grant from Ferring Pharmaceuticals http://eaustockholm2014.uroweb.org/en/eaun-meeting/eaun-awards
Conflict of interest
The authors declare no conflict of interests.
Ethical Statement
The study was approved by the Regional ethical committee Central Denmark Region, protocol no. 1-10-72-195-14 and the Danish Data Protection Agency, protocol no. 1-16-02-536-14. The study is registered at ClinicalTrials.gov, ID NCT01762241. Informed written consent is obtained from all participants prior to participation.
Acknowledgement
The authors would like to thank Elgiganten Holstebro, Gamestop Holstebro and Sportsmaster Holstebro for delivering exercise equipment on special offer. The Family Kjærsgaard, Sunds foundation and Family Hede Nielsen foundation, hospital managers and department managers at Regional Hospital Holstebro for economic support. The Lundbeck center for fast track hip and knee surgery for lending us the leg extensor power rig. Mölnlycke Health Care ApS for sponsor Visual Analog Scales. Members of Journal Club at Regional Hospital Vest for academic support, staff at Regional Hospital Holstebro and last but not least prostate cancer patients and their spouses for giving us the idea to this research project.
References
-
Bourke L, Homer KE, Thaha MA, Steed L, Rosario DJ, et al. (2013) Interventions for promoting habitual exercise in people living with and beyond cancer. See comment in PubMed Commons below Cochrane Database Syst Rev 9: CD010192.
-
Heidenreich a, Bolla M, Joniau S, Mason MD, Matveev V, et al. (2011) Guidelines on Prostate Cancer. 53: 31-45.
-
Tivesten Å, Pinthus JH, Clarke N, Duivenvoorden W, Nilsson J (2015) Cardiovascular risk with androgen deprivation therapy for prostate cancer: Potential mechanisms. Urol Oncol Semin Orig Investig . Elsevier 1-12.
-
Harrington JM, Schwenke DC, Epstein DR, Bailey DE (2014) Androgen-deprivation therapy and metabolic syndrome in men with prostate cancer. Oncol Nurs Forum 41: 21-29.
-
Cheung AS, Zajac JD, Grossmann M (2014) Muscle and bone effects of androgen deprivation therapy: current and emerging therapies. Endocr Relat Cancer 21: R371-94.
-
Sadetsky N, Greene K, Cooperberg MR, Hubbard A, Carroll PR, et al. (2011) Impact of androgen deprivation on physical well-being in patients with prostate cancer: analysis from the CaPSURE (Cancer of the Prostate Strategic Urologic Research Endeavor) registry. Cancer 117: 4406-4413.
-
Conteduca V, Di Lorenzo G, Bozza G, Ardito R, Aieta M (2013) Metabolic syndrome as a peculiar target for management of prostate cancer patients. Clin Genitourin Cancer 11: 211-220.
-
Basaria S, Muller DC, Carducci MA, Egan J, Dobs AS (2007) Relation between duration of androgen deprivation therapy and degree of insulin resistance in men with prostate cancer. Arch Intern Med 167: 612-613.
-
McClellan R (2013) Exercise programs for patients with cancer improve physical functioning and quality of life. J Physiother 59: 57.
-
Gilbert SE, Tew GA, Fairhurst C, Bourke L, Saxton JM, et al. (2016) Effects of a lifestyle intervention on endothelial function in men on long-term androgen deprivation therapy for prostate cancer. Br J Cancer Jan 14. doi: 10.1038/bjc.2015.479.
-
Hasenoehrl T, Keilani M, Sedghi Komanadj T, Mickel M, Margreiter M, et al. (2015) The effects of resistance exercise on physical performance and health-related quality of life in prostate cancer patients: a systematic review. Support Care Cancer 23: 2479-2497.
-
Keogh JWL, MacLeod RD (2012) Body composition, physical fitness, functional performance, quality of life, and fatigue benefits of exercise for prostate cancer patients: a systematic review. J Pain Symptom Manage 43: 96-110.
-
Zeng Y, Luo T, Xie H, Huang M, Cheng AS (2014) Health benefits of qigong or tai chi for cancer patients: a systematic review and meta-analyses. Complement Ther Med 22: 173-186.
-
Harrington JM, Schwenke DC, Epstein DR (2013) Exercise preferences among men with prostate cancer receiving androgen-deprivation therapy. Oncol Nurs Forum 40: E358-67.
-
Jahn P, Lakowa N, Landenberger M, Vordermark D, Stoll O (2012) InterACTIV: an exploratory study of the use of a game console to promote physical activation of hospitalized adult patients with cancer. Oncol Nurs Forum 39: E84-90.
-
Hoffman AJ, Brintnall RA, Brown JK, Eye A von, Jones LW, et al. (2012) Too sick not to exercise: using a 6-week, home-based exercise intervention for cancer-related fatigue self-management for postsurgical non-small cell lung cancer patients. Cancer Nurs 36: 175-188.
-
Jorgensen MG, Laessoe U, Hendriksen C, Nielsen OBF, Aagaard P (2013) Efficacy of Nintendo Wii training on mechanical leg muscle function and postural balance in community-dwelling older adults: a randomized controlled trial. J Gerontol A Biol Sci Med Sci 68: 845-852.
-
Holmes H, Wood J, Jenkins S, Winship P, Lunt D, et al. (2013) Xbox KinectTM represents high intensity exercise for adults with cystic fibrosis. J Cyst Fibros 12: 604-608.
-
Lee G (2013) Effects of training using video games on the muscle strength, muscle tone, and activities of daily living of chronic stroke patients. See comment in PubMed Commons below J Phys Ther Sci 25: 595-597.
-
Murphy R, Wassersug R, Dechman G (2013) The role of exercise in managing the adverse effects of androgen deprivation therapy in men with prostate cancer. Maney Publishing Suite 1C 16: 269-277.