International Journal of Stem cell Research and Therapy
Adult Brain Neurogenesis, Neural Stem Cells and Neurogenic Niches
Lina Maria Delgado-Garcia* and Rogerio Martins Amorim
Department of Veterinary Clinics, São Paulo State University, UNESP, Botucatu, Brazil
*Corresponding author: Lina Maria Delgado Garcia, Department of Veterinary Clinics, College of Veterinary Medicine and Animal Science, São Paulo State University, UNESP, Prof. Doutor Walter Mauricio Correa street, Botucatu, SP, Brazil, E-mail: lina@fmvz.unesp.br
Int J Stem Cell Res Ther, IJSCRT-3-039, (Volume 3, Issue 2), Mini Review; ISSN: 2469-570X
Received: February 02, 2016 | Accepted: July 24, 2016 | Published: July 28, 2016
Citation: Delgado-Garcia LM, Amorim RM (2016) Adult Brain Neurogenesis, Neural Stem Cells and Neurogenic Niches. Int J Stem Cell Res Ther 3:039. 10.23937/2469-570X/1410039
Copyright: © 2016 Delgado-Garcia LM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
During the late 60's, the experiments with brain lesions in rats, suggested the possibility of neurogenesis in the adult brain. Three decades later, the conceptual link between adult neurogenesis and stem cells was proposed. We now know that the adult mammalian brain may have the ability to regenerate through adult neural stem cells (NSCs). Here are briefly reviewed classical and current theories in adult neurogenesis, NSCs and neurogenic niches. Experiments in developmental biology have shown different neurogenic regions according to the capacity of neurogenesis and its presence in different species. Advances in the methodology such as optogenetics and gene reprogramming, allowing the manipulation of different conditions, will guide the discovery of new signaling pathways and interactions, to better understand some aspects recently discussed such as the variation of cell cycle length and the influence of disease conditions, cell heterogeneity and DNA changes. New discoveries in adult neurogenesis, NSCs and neurogenic niches and the developing of new technological tools should guide the NSCs-based therapy, such as gene reprogramming and the insertion of transposable elements (TEs) controlling flanking genes, causing a functional impact for different conditions.
Keywords
Cell cycle, Neuroblasts, Neurogenesis, Signaling pathways, Subgranular zone, Subventricular zone
Introduction
The cortex of lizards is a useful model to study and understand adult neurogenesis and regeneration mechanisms. Comparative studies in different species showed that lizards had an increase of the brain size with age, due to the proliferation of zinc-positive synapses and dendritic growth in the telencephalon [1,2]. In another set of experiments, induced neuronal degeneration triggered reactive neurogenesis and complete functional regeneration programs. In these experiments, the chemical lesion of the medial cortex produced the proliferation of the cells of the ependymal region and the subsequent migration and differentiation of neuroblasts up to the cortex for the complete regeneration of the zone. The neuroblasts progressively replaced dead neurons and new medial cortex was indistinguishable from a normal one [1,3,4].
Nevertheless, to accomplish the same feat on mammals, seemed an intricate issue. Progress in this matter had its starting point during the late 60's, when the experiments with brain lesions in rats, suggested the possibility of neurogenesis in the adult brain [5]. Three decades later, the conceptual link between adult neurogenesis and stem cells was proposed [6]. We now know that the adult mammalian brain may have the ability to regenerate through adult neural stem cells (NSCs). Here are briefly reviewed classical and current theories in adult neurogenesis, NSCs and neurogenic niches. Finally, are described NSCs-based therapies for spinal cord injury (SCI) and amyotrophic lateral sclerosis (ALS) disease.
Discovering Adult Brain Neurogenesis
During the past 19th century to early 20th, neurogenesis was restricted to the early stages of embryonic development. The central evidence was the lack of neurons with mitotic activity after birth in the mammalian brain [7-9]. Later Altman proposed that the neuroblasts might arise from non-differentiated precursor cells, thus, neural precursor cells generate newborn cells that differentiate and add new neurons to the existing population in the adult mammalian brain. This was proposed after the experiments in rats reviewed between 1962 and 1977. In autoradiographic studies, he showed that the chemical lesion in the brain of the rat resulted in the proliferation of newborn cells population of glia and some neuroblasts that were labeled with (H3) thymidine, a cellular marker of proliferation [5,10-14].
After this publication, many experiments tried to find these results but reproducibility of the (H3) thymidine experiments was not possible and the hypothesis of newborn neurons in the adult mammalian brain was controversial. First experiments with (H3) thymidine in non-human primates described a slow glial cell proliferation but failed to confirm adult neurogenesis [15,16]. Absence of adult neurogenesis in humans was supported by the idea that the brain of primates may be uniquely specialized in lacking the capacity for neuronal production in the adult stage. Prolonged period of interaction with the environment would require a stable set of neurons to retain acquired experiences in their synaptic connectivity [16].
Experiments carried out for the study of the brain events that control learning in canaries found opposite conclusions. Nottebohm used canaries as a model because of theirs ability of altering theirs songs; the process that he described was analogous to the acquisition of language in humans [17-19]. Descriptive studies of the brain of canaries showed a correlation between the complexity of the song and the size of specific forebrain regions controlling the songs, arguing that learned information required space for neurogenesis. Final acceptance of adult neurogenesis in mammals came after the evidence in late 90`s of neuroblasts in the human brain. The experiments used postmortem tissue from the hippocampus of cancer patients who received bromodeoxyuridine (BrdU) marker, a thymidine analog that is incorporated into the DNA of dividing cells and can be detected immunohistochemically in the progeny. Tissue of all patients showed that the SGZ of the hippocampus contained BrdU positive nuclei cells with the typical morphology of granule cell neurons. The study concluded that human hippocampus retains the ability to generate new neurons throughout life [20].
Now it seems clear that neurogenesis in the adult brain implies the coordinate control of proliferation, migration and differentiation of NSCs in the neurogenic niches for the production of neurons and play an essential physiological role in memory and learning tasks, and neurodegenerative diseases, in conjunction with the beneficial effects of exercise and environmental enrichment in the neurogenic niches.
Through studies in animal models of neurodegenerative diseases such as Alzheimer, impaired neurogenesis and age-related cognitive decline on hippocampus were found. Behavioral experiments showed that aging was associated with structural alterations and functional deficits on hippocampus [21]. Also, physical activity may induce adult hippocampal neurogenesis in aged mice [22,23] and the exposure to enriched environment induced plasticity of neuronal cells and neurogenesis [21,23-25].
Finally, new perspectives in normal health conditions and disease and the limits of adult neurogenesis in mammal species are now evaluated in the context of the comparative studies. Among rodents, non-human primates and carnivores it is observed an overall exponential decline in the quantity and function of neurogenesis and neurogenic niches; in species with longer life expectancies such as non-human primates and carnivores, neurogenesis and neurogenic niches were less abundant [2,26]. Thus, three groups of neurogenic regions with distinctive neurogenesis functions are proposed. Those regions that are constant between the different species and that display complete neurogenesis such as the subventricular zone (SVZ) and the subgranular zone(SGZ); those regions relatively constant in different species and with different degrees of neurogenesis depending on the species such as the striatum and cortex; and those regions with the presence of neurogenesis depending on the species such as the cerebellum in rodents. Differences between regions and animal species correspond to different degrees and types of plasticity adapted to different tissue environments and functional demands [27,28].
Adult Neural Stem Cells (NSC's) Activity
Neurogenesis in the adult brain implies the coordinate control of proliferation, migration and differentiation of NSCs in the neurogenic niches for the production of neurons. NSCs activity and multipotency are controlled by the combination of a wide variety of intrinsic and extrinsic mechanisms in the neurogenic niche, such as cell cycle regulators and signaling pathways.
Cell cycle inhibitors (CKIs) are proteins that inhibit cyclin-dependent kinases, regulating the G1 stage of the cell cycle [29]. Experiments with the selected deletion of CKIs proteins in normal and disease conditions showed that CKIp21 and CKIp27 proteins are expressed in rapid neural progenitor cells whereas CKIp57 proteins in radial glial cells and NSCs [30,31]. Thus, deletion of CKIp57 proteins allowed cell cycle progression and neurogenesis, not only in young adult mice but also in aged mice [31]. In vivo experiments with the selective labeling of NSCs in mice embryos also showed that, these CKIp57 NSCs were slowly dividing populations of neural progenitor cells in the embryonic ganglionic eminence, that later became adult quiescent NSCs [32]. The studies conclude that adult quiescent NSCs slowed their cell cycle, and were set aside as a reserve pool early in brain development, when other NPCs were dividing rapidly to build the brain [31-33].
A wide variety of signaling pathways regulate NSCs activity during embryonic development and later their maintenance in the adult neurogenic niches [34-37]. NOTCH signaling pathway induces maintenance and differentiation of NSCs. Upon activation of NOTCH by its receptor DELTA, the NOTCH intracellular domain (NICD) is transferred to the nucleus where it forms a complex with the DNA binding protein RBPJK and the coactivator Mastermind-like (MAML) [38,39]. During embryonic development, inactivation of RBPJK leads to a decrease in cell proliferation because dividing NSCs prematurely differentiate into neuroblasts. In contrast, in neurogenic niches, inactivation of RBPJK leads to an increase in cell proliferation because slowly dividing or adult quiescent NSCs become rapidly transit-amplifying progenitor cells [38]. Thus, the NICD-RBPJK-MAML complex induces the expression of bHLH transcriptional repressors, such as HES1, HES5 and HES3. These HES factors bind to specific DNA elements repressing the expression of target proneural genes such as ASCL1/MASH1 and NEUROGENIN2, which activate the transmembrane protein NOTCH in neighboring cells inhibiting neuronal differentiation [38-41]. Inhibition of neighboring cells is called lateral inhibition, a cell interaction process that prevents simultaneous differentiation, thereby achieving its prolonged maintenance [38].
Recently it was discovered that the expression of BHLH transcriptional repressors have an oscillating activity in BHLH factors. These results were obtained by optogenetic approach experiments, which induced gene expression by blue light illumination control [41]. Sustained light illumination induced ASCL1 expression that promoted neuronal fate determination and inhibits its cell-cycle progression, whereas oscillatory light illumination induced ASCL1 oscillatory expression that maintains proliferating neural progenitor cells. Thus, the multipotent state correlates with oscillatory expression of several fate determination factors, whereas the differentiated state correlates with sustained expression of a single factor [38,39,41]. Taken together, these results indicate that the BHLH transcriptional repressor HES regulates the expression of the proneural factor ASCL1 in an oscillatory versus sustained activity, which in turn regulates the proliferation of NSCs and the subsequent processes of neuronal fate determination. Additionally, because of its influence in proliferation and fate determination and differentiation, ASCL1 is also a central component of direct reprogramming of mouse and human fibroblasts to induced neural stem cells, a promising approach for human disease modeling and regenerative medicine [39].
WNT signaling increase neurogenesis, by contrast, its blockage reduces neurogenesis in vitro and abolishes almost completely in vivo [42]. The molecular mechanism of canonical WNT signaling pathway causes accumulation of β-catenin in the cytoplasm and its translocation into the nucleus to activate transcription factors to the T-Cell Factor/Lymphoid Enhancer Factor (TCF/LEF) family. In addition, WNT pathway may be involved in the activation of NEUROD1 transcriptional factor. This protein is a member of BHLH transcriptional factors, which activate the NEUROD gene; NEUROD1 has overlapping DNA-binding site for SOX2 and TCF/LEF, thus, the activation of this gene implies activation of the canonical WNT pathway and removal of SOX2 repression. This signaling pathway was demonstrated in experiments in mice where WNT signaling expression, together with the removal of SOX2, triggered the expression of NEUROD1 [43]. In addition, WNT signaling directly induce proneural genes such as NEUROG2 and PROX1 in intermediate progenitors [37,44].
Sonic hedgehog (SHH) and Bone Morphogenetic Proteins (BMPs) signaling act antagonistically through the modulation of EGF [45]. NSCs increase proliferation in response to higher than normal endogenous SHH levels. BMPs are part of the Transforming growth TGF families of proteins, during embryonic development, if ectodermal cells are exposed to BMPs, they assume an epidermal fate [36]. In the neurogenic niches, loss of BMPs signaling leads to an over proliferation of NSCs that ultimately depletes the population [37,46]. Activation of EGFR by EGF leads to the phosphorylation of SMAD1 in the linker region, silencing BMP signaling, whereas the EGFR activates SHH [46]. On the whole, activation of NOTCH and WNT signaling leads to maintenance of the neural stem cell population and neurogenesis, whereas inactivation of NOTCH induces rapid neuronal differentiation and depletes the NSCs population. In contrast, BMPs and SHH may act in opposite manners, BMP signaling inhibits neuronal differentiation and promotes gliogenesis, whereas endogenous SHH signaling enhances neurogenesis and the number of NSCs.
In addition to the mechanisms described above, epigenetic mechanisms such as DNA methylation, histone modifications and non-conding RNAs also influence NSCs activity [47]. Lately there is a discussion on epigenetic mechanisms related to the heterogeneity and the individual neuronal diversification in the brain, called transposable elements (TEs) [48]. There are two major classes of TEs, distinguished by their mechanism of transposition, DNA transposons, which move via cut-and-paste mechanism and are generally thought to be extinct in higher eukaryotes and RNA transposons or retrotransposons, which move via a copy-and-paste mechanism using RNA. Barbara McClintock, who discovered the TEs, and with these studies she won the Nobel Prize in Physiology and Medicine (1983) described two main functions, as insertional mutagens and as controlling elements that regulate the expression of nearby genes [49,50]. Retrotransposition-mediated events are not only confined to germ cells, but can produce somatic alterations in the brain and cancer; furthermore, it is not known whether transposon expression is a cause or a consequence of aging or of neurodegenerative disease [49]. Thus, almost half of mammalian genomes are comprised of retrotransposons, namely, Long-Interspersed Elements (LINE1s), which are the only known active and autonomous elements in the genome with the ability to mobilize and change locations in the genome [50]. In vitro experiments with adult rat NSCs showed that LINE-1 retrotransposed in neuronal precursors derived from the hippocampus. The resulting retrotransposition event altered the expression of neuronal genes, which, in turn, may influence neuronal cell fate [48].
Neurogenic Niche Microenvironment
As mentioned, neurogenic niches such as the SVZ and the SGZ are brain regions with complete neurogenesis throughout life, possible because of presence of NSCs and an appropriate cellular microenvironment to stimulate self-renewal and multipotency (Figure 1) [27,28,51].
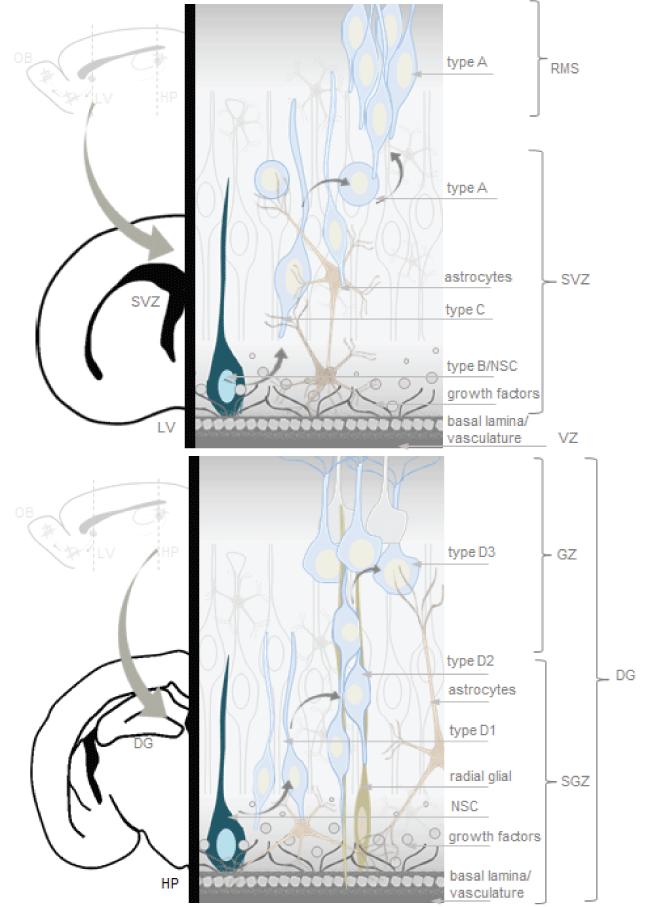
.
Figure 1: Neurogenic niches in the adult mammalian brain. The microenvironment of neural stem cell (NSCs) include a basal lamina, vasculature structures and support cells. The subventricular zone (SVZ) is the layer along the lateral ventricle. In the subventricular zone (SVZ) type B cells (NSCs-astrocytes) divide to give rise to type C cells (rapidly dividing transit amplifying immature progenitors), which in turn divide, forming a cluster that generates type A cells (neuroblasts). The SGZ is a germinal layer between the dentate gyrus and the hilus in the hippocampus. NSCs generate D1 cells (progenitors), which divide to generate D2 cells. D2 cells reach the granule layer becoming D3 cells (neuroblasts), which mature into new granule neurons. Artwork by Lina Maria Delgado-Garcia inspired in neural stem cell niches models from the cited literature of the current review.
View Figure 1
The SVZ is the layer along the lateral ventricle. There are three main cell types in the SVZ neurogenic niche: neuroblasts (Type A cells), NSC-SVZ astrocytes (Type B cells) and rapidly dividing transit amplifying immature progenitors (Type C cells). Type B and C cells are precursor cells, of those, Type B cells are the primary precursors or NSCs that divide to give rise to Type C cells or rapidly dividing transit amplifying cells or proliferating progenitors, which in turn generate migratory Type A cells or neuroblasts [52-57]. Neuroblasts of the SVZ join the network in the rostral migratory stream (RMS) to reach the olfactory bulb, where differentiate into granular and glomerular neurons [52-57]. In the olfactory bulb, primary sensory neurons project to glomeruli where they synapse onto mitral and tufted cells, which in turn relay information to the olfactory cortex, periglomerular neurons provide lateral inhibition between individual glomeruli, whereas granule cells provide lateral inhibition between mitral and tufted cells [47].
The SGZ is a germinal layer between the dentate gyrus and the hilus in the hippocampus. NSCs generate D1 cells, which divide to generate D2 cells or neuroblasts. D2 cells reach the granule layer becoming D3 cells, which mature into new granule neurons [57-59]. Granule neurons synapses onto hilar basket interneurons, each of which inhibit mature dentate granule cells. Granule cells also synapse onto hilar mossy cells, which also innervate many mature dentate granule cells on the contralateral dentate gyrus [47].
NSCs cellular microenvironment includes, a basal lamina for cell anchoring, extracellular matrix, which modulates the adhesiveness and activity of signaling molecules, vasculature structures and a single layer of ependymal cells to support and capture proteins secreted by the choroid plexus into the cerebrospinal fluid [52-59]. Tenascin-C, collagen-1, heparin sulfate proteoglycans, chondroitin sulfate proteoglycans and integrins are part of basal lamina and extracellular matrix [35]. Signaling molecules modulate gene expression acting at different stages of the stem cell lineage. Different in vivo and in vitro experiments, primarily involving extirpation or transplantation of different portions of immature and mature nervous system recognized that neurogenesis depends on the signals of growth factors such as EGF, FGF, IGF1, TGF, VEGF and LIF to regulate cell cycle and differentiation [51-62]. EGF can support in vitro proliferation of immature glial and neuronal progenitors from the adult mouse SVZ [60]. FGF-2 is effective to support the survival and proliferation of embryonic and adult rat SGZ progenitors in vitro and has been identified in vivo [51]. In vivo and in vitro experiments showed LIF stimulates neuronal differentiation from explants of the spinal cord primordium cells and dissociated culture [61]. Thus, SVZ and SGZ neuroblasts are integrated into preexisting neural circuits for remodeling and play an important role in higher brain functions such as cognitive activities.
Conclusions and Prospects for Brain Repair in Spinal Cord Injury and Amyotrophic Lateral Sclerosis
Neurogenesis in the adult brain, NSCs and neurogenic niches are issues discussed in several articles under different points of view. Since the discovery of adult neurogenesis and NSCs in mammals, experiments in developmental biology have showed different neurogenic regions according to the capacity of neurogenesis and its presence in different species. Now, it seems important to identify the population in these regions and the characteristics of its microenvironment. Other important points are the aspects of the intrinsic and extrinsic regulation of NSCs activity and the function of neurogenic niches in normal health conditions and disease. Advances in methodologies such as optogenetics and gene reprogramming, allow the manipulation of different conditions and will guide the discovery of new signaling pathways and interactions, to better understand some aspects recently discussed such as the variation of cell cycle length and the influence of disease conditions, cell heterogeneity and DNA changes.
New discoveries in adult neurogenesis, NSCs and neurogenic niches and the developing of new technological tools should guide NSCs-based therapy, such as gene reprogramming and the insertion of transposable elements (TEs) controlling flanking genes, causing a functionally impact for different conditions.
Spinal cord injury (SCI) and amyotrophic lateral sclerosis (ALS) are two devastating motor and sensory central nervous system conditions with poor regenerative potential. SCI results in motor and sensory deficits secondary to disrupted neuronal circuits been theirs remyelination a challenging topic. NSCs-based therapies for SCI aims to stimulate residents NSCs or generate neuroblasts and also give neuroprotection, acting by reducing endogenous toxic substances, inhibiting inflammatory reactions and/or releasing growth factors and neurotrophic substances [63,64]. Clinical trials in large animal models such as dogs showed that NSCs transplantation is feasibly and has therapeutic potential, producing functional improvement by long-term neurotrophic and neuroprotective effects in the neurogenic niche [65]. Clone 01 of human fetal NSCs derived from fetal spinal cord differentiated toward motor neuronal, astroglial and oligodendroglial phenotypes in the injured spinal cord and promoted functional recovery in the rat. In early and later stages, significant improvement in motor and sensory tests were mediated by the increased expression of neurotrophic factors, reduction of neurite growth-inhibitory substances, enhancement of the remyelination of the axons and the production of extracellular matrix permissive to regeneration [63].
ALS is a devastating neurodegenerative disorder in which the loss of upper and lower motor neurons produces progressive weakness and eventually death [66-68]. ALS is an inherited neurodegenerative disease mainly identified by SOD1 gene mutation, which produces progressive loss of motor function. In SOD1 transgenic models, application of NSCs grafts at multiple levels of spinal cord showed improvement of motor function, due to these results it was recently established the first phase I human-clinical trial. Results of the phase I showed that cervical transplantation of NSCs in the lumbar spinal cords of 12 ALS patients is feasible and well-tolerated, additional results showed that some cells differentiated into neurons, while others maintained their stem cells phenotype [68,69]. The fact that NSC produce new elements that can integrate within some regions of the mature brain, replacing lost neurons in pre-existent neural circuits, appears extremely fascinating in the perspective of regenerative therapeutic approaches. For the near future manipulation of NSCs after SCI and ALS for replacing lost elements seem very hard to be realized, neuroblasts themselves may not be useful to directly repair the brain, but the use of the intrinsic and extrinsic factors of theirs microenvironment to give neuroprotection seems a more handley approach.
Competing Interests
The authors declare that they have no competing interests.
Authors' Contributions
LMDG and RMA prepared manuscript. All authors read and approved the final manuscript.
References
-
Lopez-Garcia C, Molowny A, Nacher J, Ponsoda X, Sancho-Bielsa F, et al. (2002) The lizard cerebral cortex as a model to study neuronal regeneration. An Acad Bras Cienc 74: 85-104.
-
Grandel H, Brand M (2013) Comparative aspects of adult neural stem cell activity in vertebrates. Dev Genes Evol 223: 131-147.
-
Font E, García-Verdugo JM, Alcántara S, López-García C (1991) Neuron regeneration reverses 3-acetylpyridine-induced cell loss in the cerebral cortex of adult lizards. Brain Res 551: 230-235.
-
Molowny A, Nacher J, Lopez-Garcia C (1995) Reactive neurogenesis during regeneration of the lesioned medial cerebral cortex of lizards. Neuroscience 68: 823-36.
-
Altman J (1962) Are new neurons formed in the brains of adult mammals? Science 135: 1127-1128.
-
Gage FH, Ray J, Fisher LJ (1995) Isolation, characterization, and use of stem cells from the CNS. Annu Rev Neurosci 18: 159-192.
-
Ramon y Cajal S (1894) La fine structure des centresnerveux. Prox Roy Soc Lond B 55: 444-468.
-
Ramon y Cajal S (1897) Leyes de la morfología y dinamismo de las células nerviosas Rev Trim Micrografica 2: 1-12.
-
Ramon y Cajal S (1899) Textura del Sistema Nervioso del Hombre y de los Vertebrados. Moya; Madrid.
-
Altman J, Das GD (1965) Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol 124: 319-335.
-
Altman J, Das GD (1966) Autoradiographic and histological studies of postnatal neurogenesis. I. A longitudinal investigation of the kinetics, migration and transformation of cells ncorporatingtritiated thymidine in neonate rats, with special reference to postnatal neurogenesis in some brain regions. J Comp Neurol 126: 337-389.
-
Altman J (1969) Autoradiographic and histological studies of postnatal neurogenesis IV Cell proliferation and migration in the anterior forebrain, with special reference to persisting neurogenesis in the olfactory bulb. J Comp Neurol 137: 433-457.
-
Altman J (1969) Autoradiographic and histological studies of postnatal neurogenesis. 3. Dating the time of production and onset of differentiation of cerebellar microneurons in rats. J Comp Neurol 136: 269-293.
-
Kaplan MS, Hinds JW (1977) Neurogenesis in the adult rat: electron microscopic analysis of light radioautographs. Science 197: 1092-1094.
-
Rakic P (1985) DNA synthesis and cell division in the adult primate brain. Ann N Y Acad Sci 457: 193-211.
-
Rakic P (1985) Limits of neurogenesis in primates. Science 227: 1054-1056.
-
Burd GD, Nottebohm F (1985) Ultrastructural characterization of synaptic terminals formed on newly generated neurons in a song control nucleus of the adult canary forebrain. J Comp Neurol 240: 143-152.
-
Nottebohm F (1985) Neuronal replacement in adulthood. Ann N Y Acad Sci 457: 143-161.
-
Paton JA, O'Loughlin BE, Nottebohm F (1985) Cells Born in Adult Canary Forebrain are Local Interneurons. J Neurosci 5: 3088-3093.
-
Eriksson PS, Perfilieva E, Björk-Eriksson T, Alborn AM, Nordborg C, et al. (1998) Neurogenesis in the adult human hippocampus. Nat Med 4: 1313-1317.
-
Driscoll I, Howard SR, Stone JC, Monfils MH, Tomanek B, et al. (2006) The aging hippocampus: a multi-level analysis in the rat. Neuroscience 139: 1173-1185.
-
Kronenberg G, Bick-Sander A, Bunk E, Wolf C, Ehninger D, et al. (2006) Physical exercise prevents age-related decline in precursor cell activity in the mouse dentate gyrus. Neurobiol Aging 27: 1505-1513.
-
Lazarov O, Mattson MP, Peterson DA, Pimplikar SW, van Praag H (2010) When neurogenesis encounters aging and disease. Trends Neurosci 33: 569-579.
-
Kuhn HG, Dickinson-Anson H, Gage FH (1996) Neurogenesis in the dentate gyrus of the adult rat: age-related decrease of neuronal progenitor proliferation. J Neurosci 16: 2027-2033.
-
Kempermann G, Kuhn HG, Gage FH (1998) Experience-induced neurogenesis in the senescent dentate gyrus. J Neurosci 18: 3206-3212.
-
Amrein I, Isler K, Lipp HP (2011) Comparing adult hippocampal neurogenesis in mammalian species and orders: influence of chronological age and life history stage. Eur J Neurosci 34: 978-987.
-
Bonfanti L, Peretto P (2011) Adult neurogenesis in mammals-a theme with many variations. Eur J Neurosci 34: 930-950.
-
Bonfanti L (2016) Adult Neurogenesis 50 Years Later: Limits and Opportunities in Mammals. Front Neurosci 10: 44.
-
Mu Y, Lee SW, Gage FH (2010) Signaling in adult neurogenesis. Curr Opin Neurobiol 20: 416-423.
-
Kippin TE, Martens DJ, van der Kooy D (2005) p21 loss compromises the relative quiescence of forebrain stem cell proliferation leading to exhaustion of their proliferation capacity. Genes Dev 19: 756-767.
-
Furutachi S, Matsumoto A, Nakayama KI, Gotoh Y (2013) p57 controls adult neural stem cell quiescence and modulates the pace of lifelong neurogenesis. EMBO J 32: 970-981.
-
Fuentealba LC, Rompani SB, Parraguez JI, Obernier K, Romero R, et al. (2015) Embryonic Origin of Postnatal Neural Stem Cells. Cell 161: 1644-1655.
-
Furutachi S, Miya H, Watanabe T, Kawai H, Yamasaki N, et al. (2015) Slowly dividing neural progenitors are an embryonic origin of adult neural stem cells. Nat Neurosci 18: 657-665.
-
Bonaguidi MA, McGuire T, Hu M, Kan L, Samanta J, et al. (2005) LIF and BMP signaling generate separate and discrete types of GFAP-expressing cells. Development 132: 5503-5514.
-
Bonaguidi MA, Peng CY, McGuire T, Falciglia G, Gobeske KT, et al. (2008) Noggin expands neural stem cells in the adult hippocampus. J Neurosci 28: 9194-9204.
-
Sun Y, Hu J, Zhou L, Pollard SM, Smith A (2011) Interplay between FGF2 and BMP controls the self-renewal, dormancy and differentiation of rat neural stem cells. J Cell Sci 124: 1867-1877.
-
Urbán N, Guillemot F (2014) Neurogenesis in the embryonic and adult brain: same regulators, different roles. Front Cell Neurosci 8: 396.
-
Imayoshi I, Sakamoto M, Yamaguchi M, Mori K, Kageyama R (2010) Essential roles of Notch signaling in maintenance of neural stem cells in developing and adult brains. J Neurosci 30: 3489-3498.
-
Imayoshi I, Shimojo H, Sakamoto M, Ohtsuka T, Kageyama R (2013) Genetic visualization of notch signaling in mammalian neurogenesis. Cell Mol Life Sci 70: 2045-2057.
-
Shimojo H, Ohtsuka T, Kageyama R (2011) Dynamic expression of notch signaling genes in neural stem/progenitor cells. Front Neurosci 5: 78.
-
Kageyama R, Shimojo H, Imayoshi I (2015) Dynamic expression and roles of Hes factors in neural development. Cell Tissue Res 359: 125-133.
-
Varela-Nallar L, Inestrosa NC (2013) Wnt signaling in the regulation of adult hippocampal neurogenesis. Front Cell Neurosci 7: 100.
-
Kuwabara T, Hsieh J, Muotri A, Yeo G, Warashina M, et al. (2009) Wnt-mediated activation of NeuroD1 and retro-elements during adult neurogenesis. Nat Neurosci 12: 1097-1105.
-
Duncan RN, Panahi S, Piotrowski T, Dorsky RI (2015) Identification of Wnt Genes Expressed in Neural Progenitor Zones during Zebrafish Brain Development. PLoS One 10: e0145810.
-
Palma V, Lim DA, Dahmane N, Sánchez P, Brionne TC, et al. (2005) Sonic hedgehog controls stem cell behavior in the postnatal and adult brain. Development 132: 335-344.
-
Palma V, Ruiz i Altaba A (2004) Hedgehog-GLI signaling regulates the behavior of cells with stem cell properties in the developing neocortex. Development 131: 337-345.
-
Ming GL, Song H (2011) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70: 687-702.
-
Muotri AR, Chu VT, Marchetto MC, Deng W, Moran JV, et al. (2005) Somatic mosaicism in neuronal precursor cells mediated by L1 retrotransposition. Nature 435: 903-910.
-
Reilly MT, Faulkner GJ, Dubnau J, Ponomarev I, Gage FH (2013) The role of transposable elements in health and diseases of the central nervous system. J Neurosci 33: 17577-17586.
-
Levin HL, Moran JV (2011) Dynamic interactions between transposable elements and their hosts. Nat Rev Genet 12: 615-627.
-
Gage FH, Coates PW, Palmer TD, Kuhn HG, Fisher LJ, et al. (1995) Survival and differentiation of adult neuronal progenitor cells transplanted to the adult brain. Proc Natl Acad Sci U S A 92: 11879-11883.
-
Doetsch F, Garcia-Verdugo JM, Alvarez-Buylla A (1997) Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J Neurosci 17: 5046-5061.
-
Doetsch F, Caillé I, Lim DA, García-Verdugo JM, Alvarez-Buylla A (1999) Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97: 703-716.
-
Lim DA, Alvarez-Buylla A (1999) Interaction between astrocytes and adult subventricular zone precursors stimulates neurogenesis. Proc Natl Acad Sci U S A 96: 7526-7531.
-
García-Verdugo JM, Doetsch F, Wichterle H, Lim DA, Alvarez-Buylla A (1998) Architecture and cell types of the adult subventricular zone: in search of the stem cells. J Neurobiol 36: 234-248.
-
Alvarez-Buylla A, Garcia-Verdugo JM (2002) Neurogenesis in adult subventricular zone. J Neurosci 22: 629-634.
-
Doetsch F (2003) A niche for adult neural stem cells. Curr Opin Genet Dev 13: 543-550.
-
Seri B, García-Verdugo JM, McEwen BS, Alvarez-Buylla A (2001) Astrocytes give rise to new neurons in the adult mammalian hippocampus. J Neurosci 21: 7153-7160.
-
Seri B, García-Verdugo JM, Collado-Morente L, McEwen BS, Alvarez-Buylla A (2004) Cell types, lineage, and architecture of the germinal zone in the adult dentate gyrus. J Comp Neurol 478: 359-378.
-
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255: 1707-1710.
-
Richards LJ, Kilpatrick TJ, Bartlett PF, Murphy M (1992) Leukemia inhibitory factor promotes the neuronal development of spinal cord precursors from the neural tube. J Neurosci Res 33: 476-484.
-
Palmer TD, Ray J, Gage FH (1995) FGF-2-responsive neuronal progenitors reside in proliferative and quiescent regions of the adult rodent brain. Mol Cell Neurosci 6: 474-486.
-
Amemori T, Romanyuk N, Jendelova P, Herynek V, Turnovcova K, et al. (2013) Human conditionally immortalized neural stem cells improve locomotor function after spinal cord injury in the rat. Stem Cell Res Ther 4: 68.
-
Emgård M, Piao J, Aineskog H, Liu J, Calzarossa C, et al. (2014) Neuroprotective effects of human spinal cord-derived neural precursor cells after transplantation to the injured spinal cord. Exp Neurol 253: 138-145
-
Kim BG, Kang YM, Phi JH, Kim YH, Hwang DH, et al. (2010) Implantation of polymer scaffolds seeded with neural stem cells in a canine spinal cord injury model. Cytotherapy 12: 841-845.
-
Chen KS, Sakowski SA, Feldman EL (2016) Intraspinal stem cell transplantation for amyotrophic lateral sclerosis. Ann Neurol 79: 342-353.
-
Goutman SA, Chen KS, Feldman EL (2015) Recent Advances and the Future of Stem Cell Therapies in Amyotrophic Lateral Sclerosis. Neurotherapeutics 12: 428-448.
-
Tadesse T, Gearing M, Senitzer D, Saxe D, Brat DJ, et al. (2014) Analysis of graft survival in a trial of stem cell transplant in ALS. Ann Clin Transl Neurol 1: 900-908.
-
Glass JD, Boulis NM, Johe K, Rutkove SB, Federici T, et al. (2012) Lumbar intraspinal injection of neural stem cells in patients with amyotrophic lateral sclerosis: results of a phase I trial in 12 patients. Stem Cells 30: 1144-1151.





