Introduction: Bariatric surgery is considered the primary treatment against severe obesity, as it is effective in sustained weight loss and reduces the prevalence of associated comorbidities. However, bariatric surgery is not free of complications observed at various postoperative stages, including fistulas.
Purpose: To describe the creation of an experimental model of a gastric fistula after the performance of Sleeve Gastrectomy (SG) in pigs.
Methods: Sleeve gastrectomy was performed in pigs, using tubes for postoperative fistula induction, following the ethical principles of animal experimentation.
Results: After an observation period of 21 days, it was possible to confirm the formation of the fistulous tract. None of the animals died.
Conclusion: The experimental model of post-Sleeve Gastrectomy fistula in pigs is efficient, effective, and reproducible and can be used in future studies to test new diagnostic methods and innovations in the therapeutic management of postoperative fistulas.
Bariatric surgery, Gastrectomy, Postoperative complications, Fistula, Models, Animal, Swine
Morbid obesity is a chronic disease considered by the World Health Organization (WHO) as a worldwide epidemic related to excess body fat, dietary profile, physical activity, and population genetics [1].
It is estimated that by 2025, 2.3 billion adults worldwide will be overweight, of which 700 million individuals will be obese, with a body mass index (BMI) above 30 and severe obesity from 35 [1-3].
Excess weight is associated with diabetes, hypertension, cardiac and pulmonary dysfunction, deep vein thrombosis, depression, predisposes patients to infections, incision-alhernias, and increases operative morbidity and mortality, related to complex surgical risks, intraoperative technical difficulties, and serious surgical complications [2-4].
Diet therapy is the first option in the fight against overweight associated with a change in life habits, such as adopting daily physical exercises for a balanced and healthy weight loss. However, studies have shown that, on average, 75%-95% of obese people fail to maintain weight loss diets due to the strong relationship between overweight and bing eating [5,6].
Thus, bariatric surgery is considered the primary treatment against severe obesity, as it is effective in sustained weight loss and reduces the prevalence of associated comorbidities [7].
Concurrent with the high prevalence of obesity, bariatric surgery has increased in number and different types of techniques. It is currently one of the most performed elective surgeries in the world [8,9].
The most common bariatric operations are laparoscopic sleeve gastrectomy (LSG), Roux-en-Y Gastric Bypass (RYGB), and Adjustable Gastric Band (AGB). The Biliopancreatic Diversion is also with Duodenal Switch (BPD-DS) and Intragastric Balloon (IB) [10-12].
In 2018, it was estimated that 63,969 and 252,000 bariatric procedures were performed in Brazil and the United States, respectively, with emphasis on laparoscopic sleeve gastrectomy, among the various techniques used [13,14].
Sleeve gastrectomy reduces gastric volume and appetite by lowering ghrelin levels and increasing GLP-1 production, contributing to weight loss. In this sense, laparoscopic sleeve gastrectomy (LSG) has become a standard procedure for the surgical treatment of patients with different degrees of obesity. Advantages include no bowel bypass, reduced risk of internal hernia obstruction, and sustained weight loss [15].
Despite the benefits above, bariatric surgery is not free of complications observed at various postoperative stages, depending on the procedure used. The mechanisms involved are associated with the surgical technique, the surgical team's experience, ischemia, use of medications, and the presence of comorbidities [16].
The management of these complications has evolved due to technological advances to gain the best results, where early diagnosis and treatment are important prognostic factors [6,17].
Regarding sleeve gastrectomy, there is a risk of staple line bleeding, strictures (the midor distal portion of the residual stomach), reflux, and gastric fistulas that increase morbidity and mortality, resulting in abdominal sepsis multiple organ failure, and death [1,7-9].
Gastric fistula is the most feared complication of GS due to its complex management and long clinical course. Up to 90% of post-SG fistulas occur at the esophago-gastric junction (EGJ) and rarely at the distal part of the staple line. The prevalence of fistulas is 2.2%-2.4%, according to systematic literature reviews [3,9].
In Roux-en-Y gastroplasty, fistulas are more frequent, with an incidence between 0.3% and 8.3%, especially when the stomach is not separated from the excluded gastric pouch [18].
Since bariatric surgery represents a challenge to the body due to the endocrine and metabolic response, each technique must be tested and perfected before hand to minimize any possible intercurrence [3,5-7].
Based on the above, the feasibility of the procedure and its results, complications, and forms of treatment, in particular postoperative fistulas, can be previously observed through the adoption of experimental models. In this way, the chosen animals must be physiologically like humans, guarantee technical equivalence and reproducibility [4,8].
The objective of the present study was to describe the creation of an experimental model of a gastric fistula after the performance of Sleeve Gastrectomy (SG) in pigs to provide an alternative for future studies involving different studies types of treatment in obese patients with this surgical complication.
The Ethics Committee approved the experimental protocol on the Use of Animals, Potiguar University (UnP), number 3251/2019, Brazil. Animals were handled following the guide for the care and use of laboratory animals, US national research council, 1996. The Institutional Committee on Ethics in the Use of Animals approved the research project under the protocol. Brazilian College of Animal Experimentation (Law11.794/2008-CONCEA-Brazilian college of animal experimentation). Three cross bred (Large white × Land race) male pigs, 25.4 ± 1.8 kg (73 ± five days of age), were purchased from a commercial farm and housed in the Animal Facility facilities of UnP one week before the surgery for acclimatization. The surgeries were performed in the experimentation sector of the animal house at UnP, where the animals were randomly separated and placed in individual pens lined with wood shavings. During the first week, the animals were weighed (Electronic Scale for Pigs/Pig/Filizola ® 2022), identified with an ear tag, dewormed (1% Ivermectin, 1 mL/35 kg/SC, Crack ® , Chile), injected with Vitamin A, D, E, 0.5 mL/animal IM, Drag ® Pharma), and vaccinated against erysipelas (2 mL/animal) and mycoplasma (2 mL/animal/SC, Intervet Veterinaria ® , Chile); for adaptation, remaining one week in the acclimatization period, under a standard controlled temperature of 20 °C ± two °C, humidity around 45% ± 15% and ambient lighting, with an inverted 12-hour light-dark cycle. In addition, the environment remained under noise below 60 dB, and the diet offered was standardized with commercial food from Purina ® and filtered water ad libitum. The animals were evaluated daily for weight and performance status. There was a random division, following the principles of the 3R's - Reduce, Refine and Replace - to guarantee the bioethical principles of the study. The three animals underwent the same operative procedure and were observed for 21 days, a period sufficient for the emergence and maturation of the spent cutaneous fistula, after removing the tubes.
The pigs were submitted to 12h of fasting. They were sedated during the preoperative period with an intramuscular association of 2 mg/kg of acepromazine (Acepran ® 1%) and 0.1 mg/kg of midazolam (Dormire ® ). After establishing intravenous fluid therapy, anesthesia was induced with 1 mg/kg of sodium thiopental (Thiopentax ® /Cristália) and 10 mg/mL propofol (Provive ® 1%). After endotracheal intubation, the anesthesia was maintained with Isoflurane (Isoforine ® ) and oxygen under mechanical ventilation. The animals were placed in a dorsal decubitus, and atrichotomy of the entire ventral abdominal region was performed by surgical antisepsis with 0.5% Chlorhexidine Gluconate (Vic Pharma ® ). The animals were placed on a surgical table with a heated mattress, and the surgery began (Figure 1).
 Figure 1: A) Animal in a dorsal decubitus and endotracheal intubation; B) Nasogastric tube and supra-umbilical median laparotomy; C) Nasogastric tube implanted to remove excess intra-gastric gas.
View Figure 1
Figure 1: A) Animal in a dorsal decubitus and endotracheal intubation; B) Nasogastric tube and supra-umbilical median laparotomy; C) Nasogastric tube implanted to remove excess intra-gastric gas.
View Figure 1
In each animal, the same surgical procedure was applied. A supra-umbilical median laparotomy was performed, opening the animal's abdominal cavity in planes (pig) and placing cavity retractors to expand the operative field. A nasogastric tube was implanted to remove excess intra-gastric gas. The stomach was isolated while the short vessels between the stomach and spleen were ligated and sectioned to allow sleeve gastrectomy without bleeding. Starting with the gastric antrum, covering the entire greater curvature of the stomach, sleeve gastrectomy was performed through stapling and sectioning of the greater curvature starting from the antrum and going to the angle of his, using 3-4 Selectable NTLC75 - linear cutter with 3d staples (ETHICON-Johnson & Johnson ® ). After this, the stomach was reduced to 30-40% of its initial volume. Then, a gastrostomy was performed at the Angle of His with the placement of a kehr 20Fr "T" drain next to a 22 Fr pezzer Probe, which was inserted into the stomach through a counter-opening in the pig's abdominal wall. The gastrostomy was internally fixed to the abdominal wall, parietal peritoneum, using 2.0 propylene thread (ETHICON - Johnson & Johnson ® ). The kehr drain and the pezzer probe were externally fixed to the abdominal wall with 1.0 polydioxanone thread (ETHICON - Johnson & Johnson ® ), followed by the creation of a subcutaneous tunnel for gastric secretion drainage to ensure that the devices did not would be removed by the animal itself and the postoperative period. At the end of the operation, the abdominal wall was closed in layers. To avoid hypothermia and consequently acidosis and postoperative hemorrhages, thermal mattresses were used on the operating table and heater and cage seals during the transport of the operated animal back to the vivarium. Postoperative pain control was performed through the administration of Meperidine (solution) 50 mg/2 mL-Intramuscularly (IM-DOLOSAL ® - Cristália), 50-100 mg/day, the dose was repeated every 3-4h. After anesthetic recovery (12 ± two h after surgery), the animals were kept in individual pens, lined with wood shavings, drinkers and feeders. The ambient temperature was maintained between 22 and 26 °C using heaters (Figure 2, Figure 3, Figure 4, Figure 5 and Figure 6).
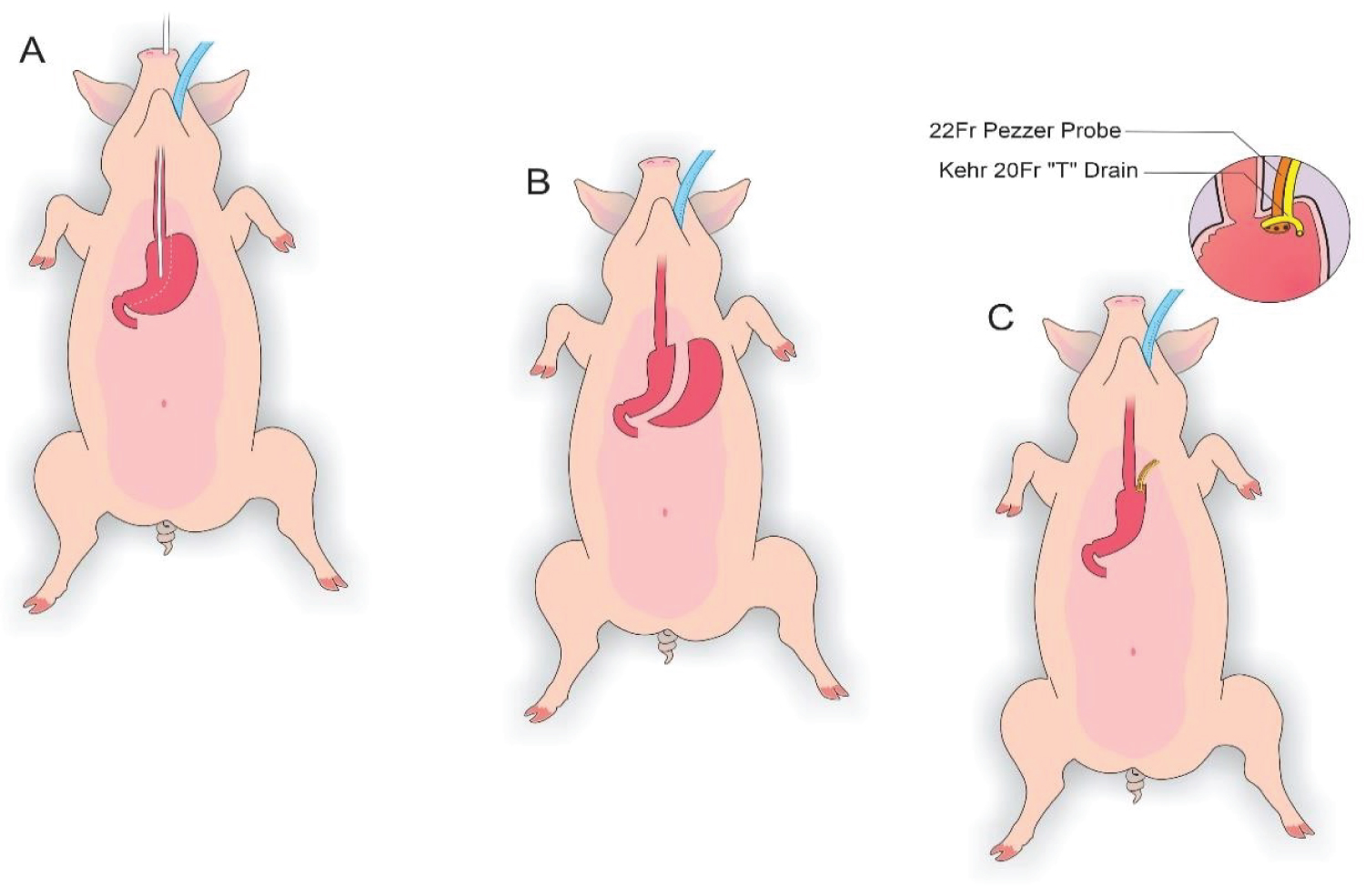 Figure 2: A) Delimitation of the gastrectomy; B) Gastrectomy performed; C) Placement of a Kehr 20Fr "T" Drain next to a 22Fr Pezzer Probe.
View Figure 2
Figure 2: A) Delimitation of the gastrectomy; B) Gastrectomy performed; C) Placement of a Kehr 20Fr "T" Drain next to a 22Fr Pezzer Probe.
View Figure 2
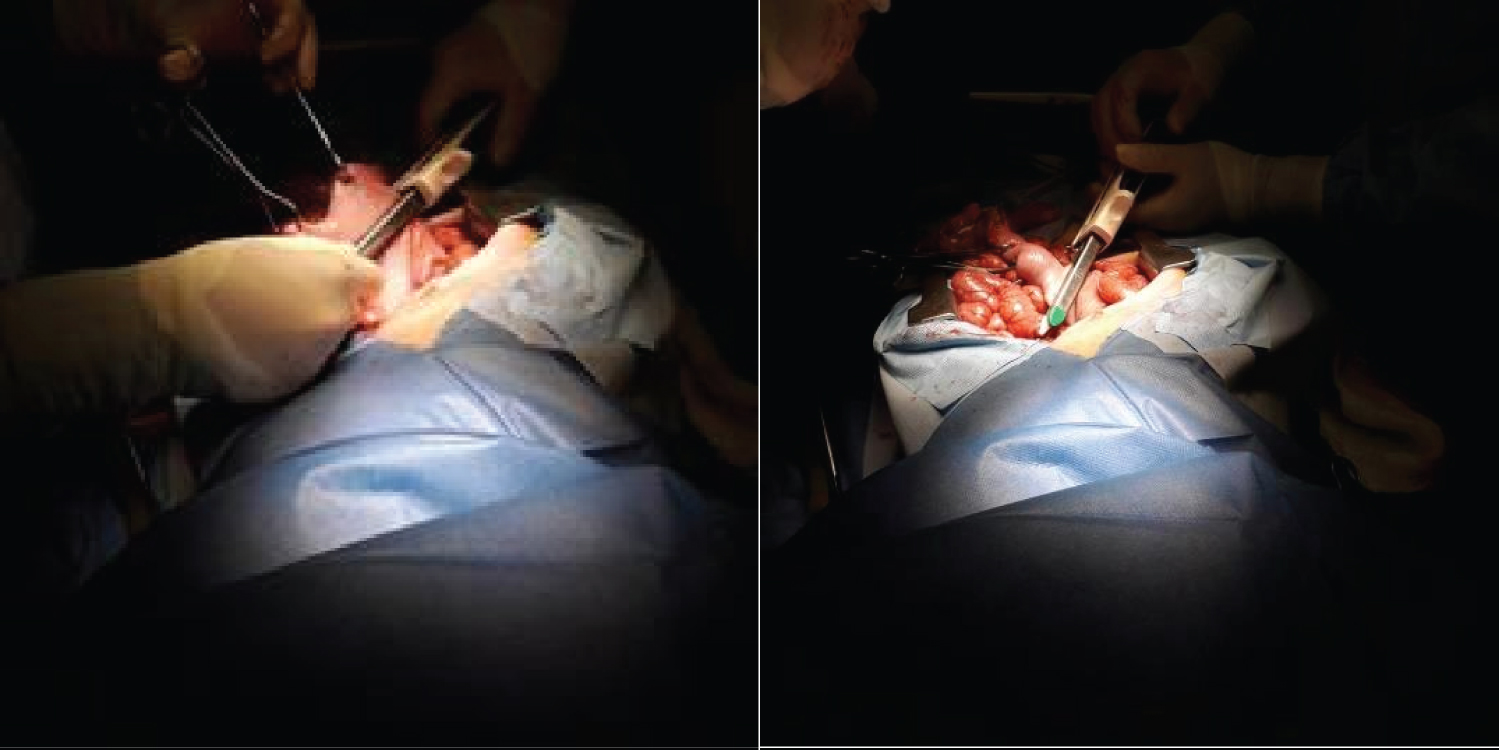 Figure 3: Sleeve Gastrectomy.
View Figure 3
Figure 3: Sleeve Gastrectomy.
View Figure 3
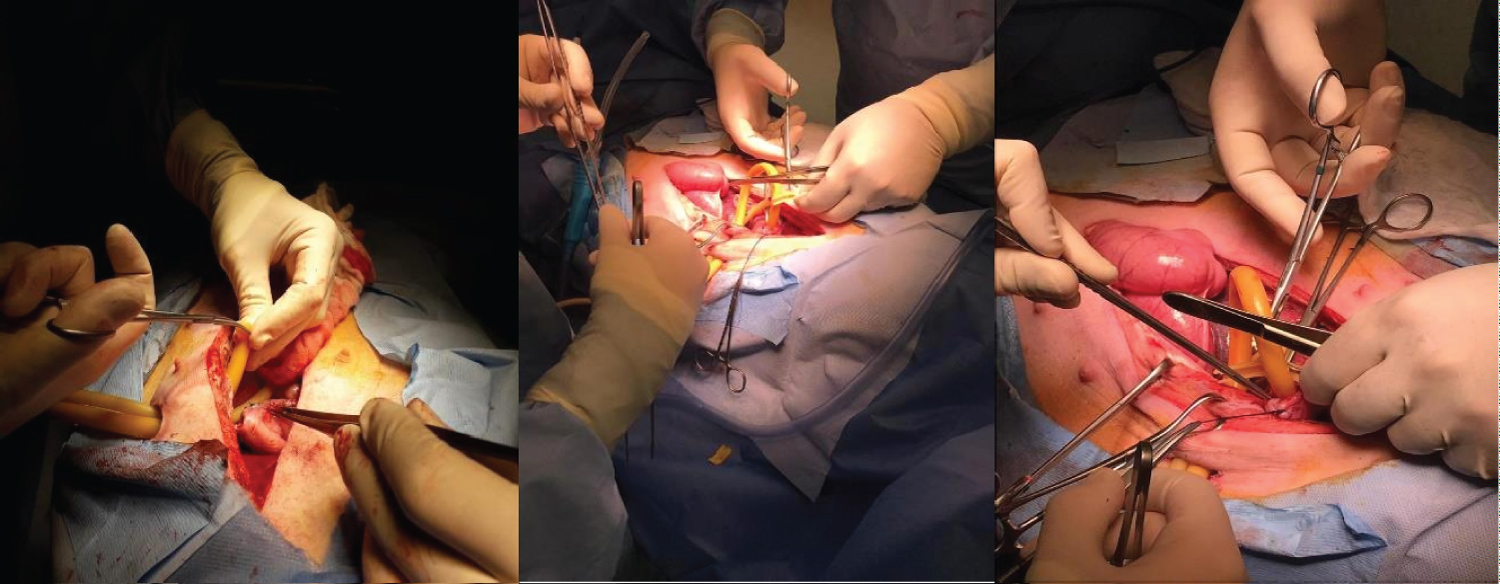 Figure 4: Kehr Drain and Pezzer Probe inserted into the Angle of His (stomach) through counter-opening in the abdominal wall after Sleeve Gastrectomy.
View Figure 4
Figure 4: Kehr Drain and Pezzer Probe inserted into the Angle of His (stomach) through counter-opening in the abdominal wall after Sleeve Gastrectomy.
View Figure 4
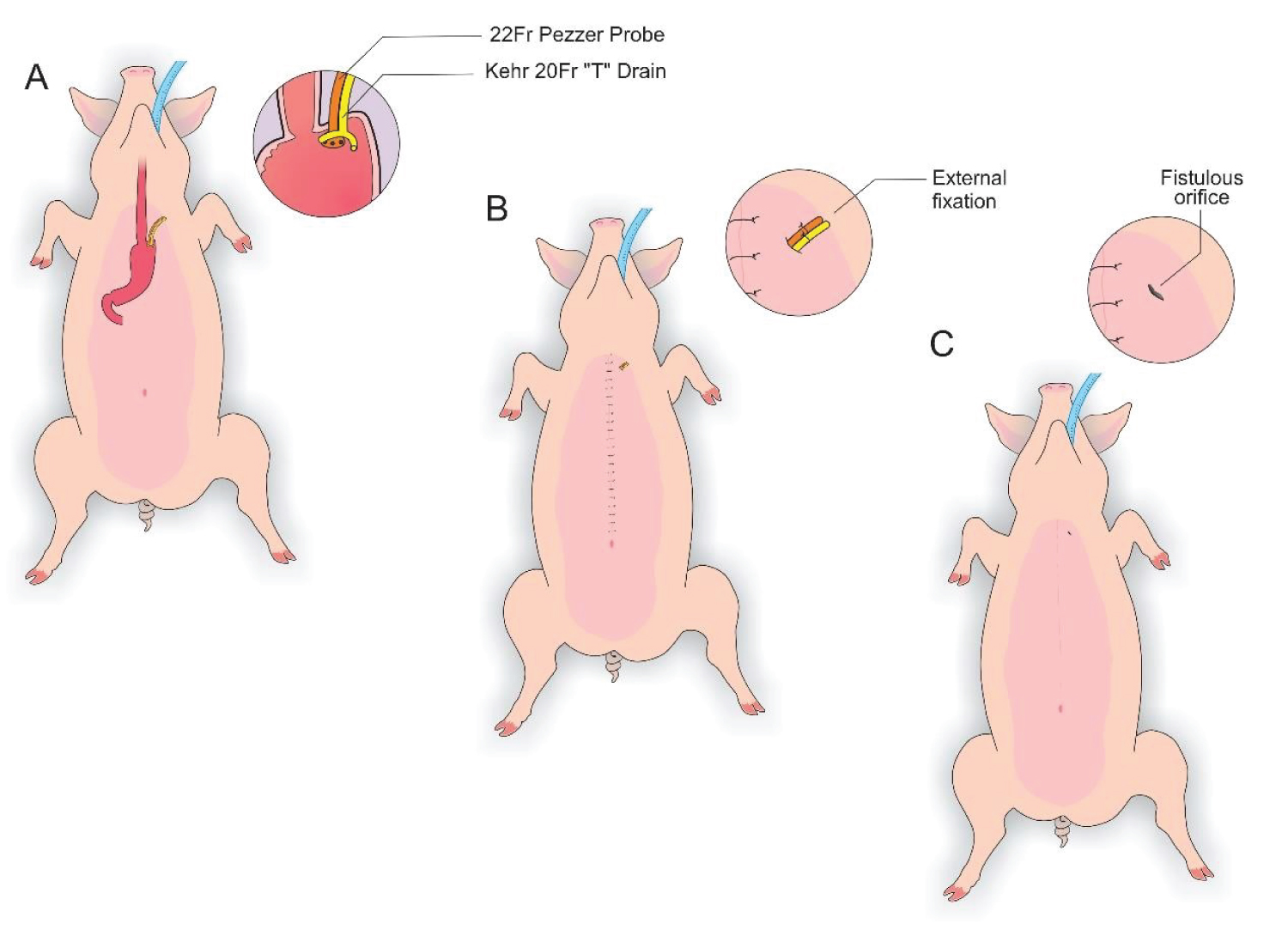 Figure 5: A) Visualization of a Kehr 20Fr "T" Drain next to a 22Fr Pezzer Probe; B) External fixation to the abdominal wall with 1.0 polydioxan; C) Visualization of the fistulous orifices.
View Figure 5
Figure 5: A) Visualization of a Kehr 20Fr "T" Drain next to a 22Fr Pezzer Probe; B) External fixation to the abdominal wall with 1.0 polydioxan; C) Visualization of the fistulous orifices.
View Figure 5
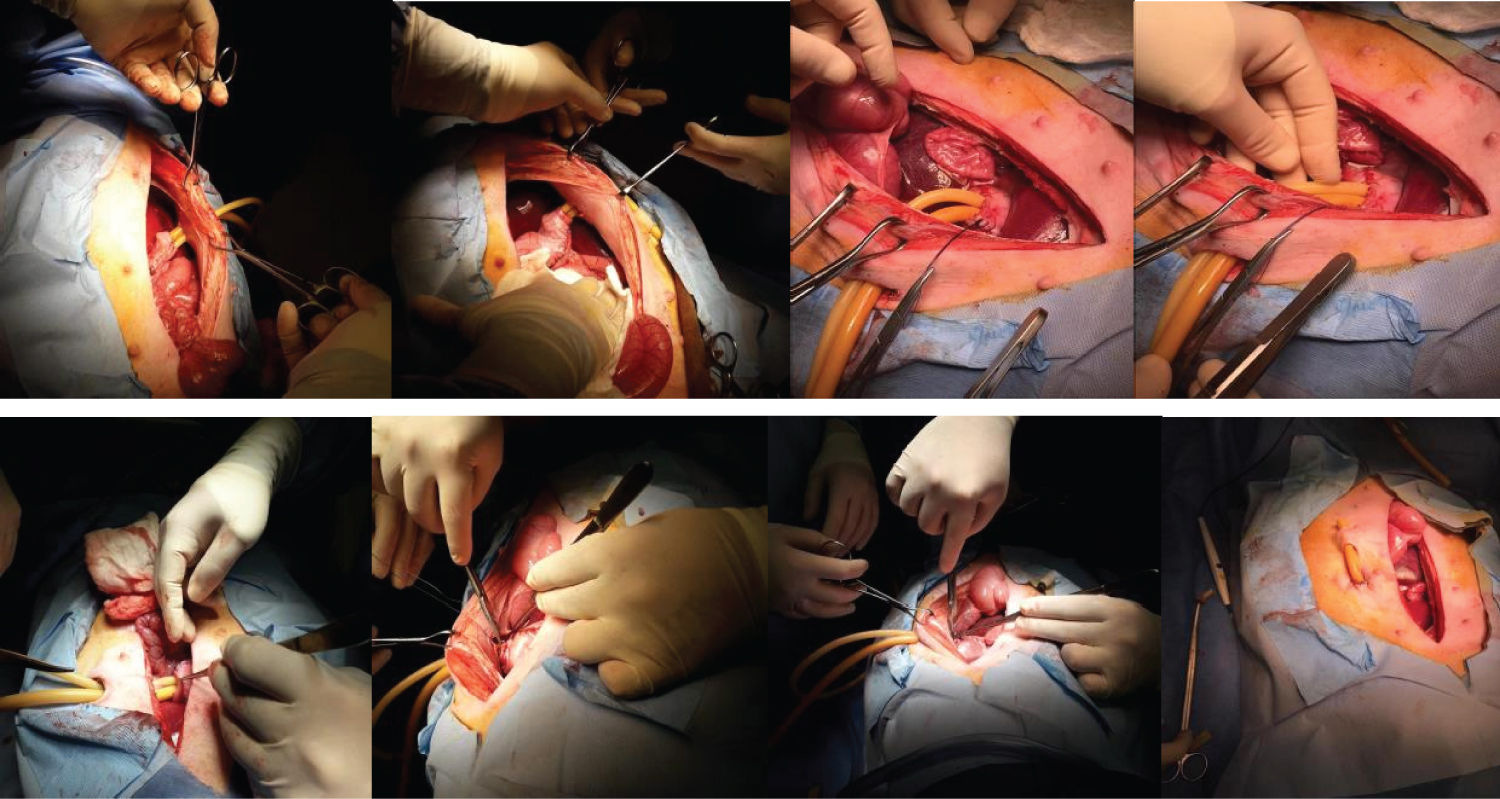 Figure 6: Fixation of the gastrostomy (Angle of His) to the abdominal wall.
View Figure 6
Figure 6: Fixation of the gastrostomy (Angle of His) to the abdominal wall.
View Figure 6
The total observation period of the study was 21 days. The animals received a soft liquid diet while observed for vomiting, pain, or lack of appetite in the first three postoperative days. Then, the regular diet from Purina ® and filtered water ad libitum was resumed for the remainder of the study. On this condition postoperative day, it was possible to observe the output of gastric secretion through the tubes placed in the stomach. None of the three animals died. A weight-loss varied in each animal, around 15-25% of the initial weight. On the 21 st postoperative day, the animals were again anesthetized. Then the radiological and endoscopic control of the fistulous path formed before and after the tubes were removed and the direct visualization of the fistulous orifices in the abdominal wall of the three animals. After verifying that there was actually a fistulous tract formed, with gastric secretion output by the various methods of analysis, the animals still anesthetized were killed without suffering after intracardiac transthoracic injection of an anesthetic overdose of Sodium Thiopental 5 mg/kg (Thiopentax ® -Cristália) using an epidural needle guided by a portable echocardiogram device (HM70 ® -Samsung) - (Figure 7, Figure 8, Figure 9 and Figure 10).
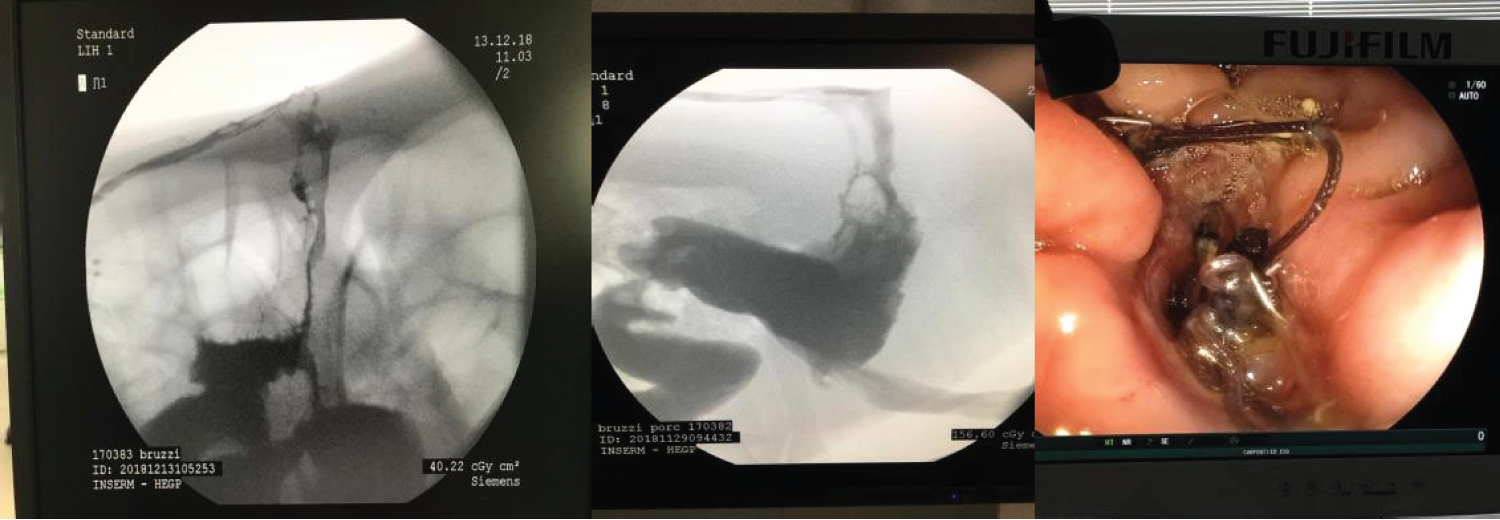 Figure 7: Radiological and endoscopic control of the fistulous tract on the 21st postoperative day.
View Figure 7
Figure 7: Radiological and endoscopic control of the fistulous tract on the 21st postoperative day.
View Figure 7
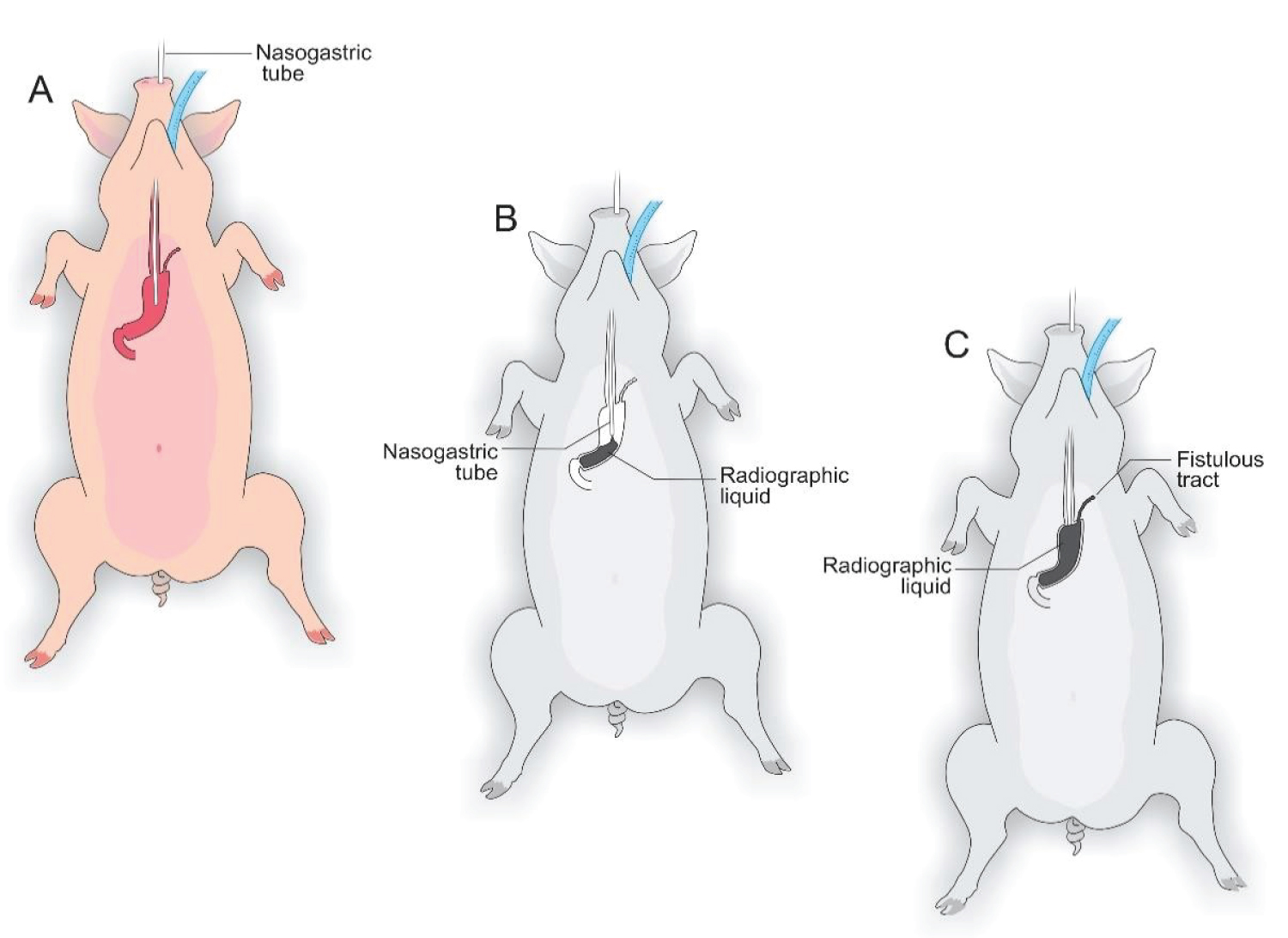 Figure 8: A) Nasogastric tube; B and C) Radiological and endoscopic control of the fistulous path formed after the tubes were removed.
View Figure 8
Figure 8: A) Nasogastric tube; B and C) Radiological and endoscopic control of the fistulous path formed after the tubes were removed.
View Figure 8
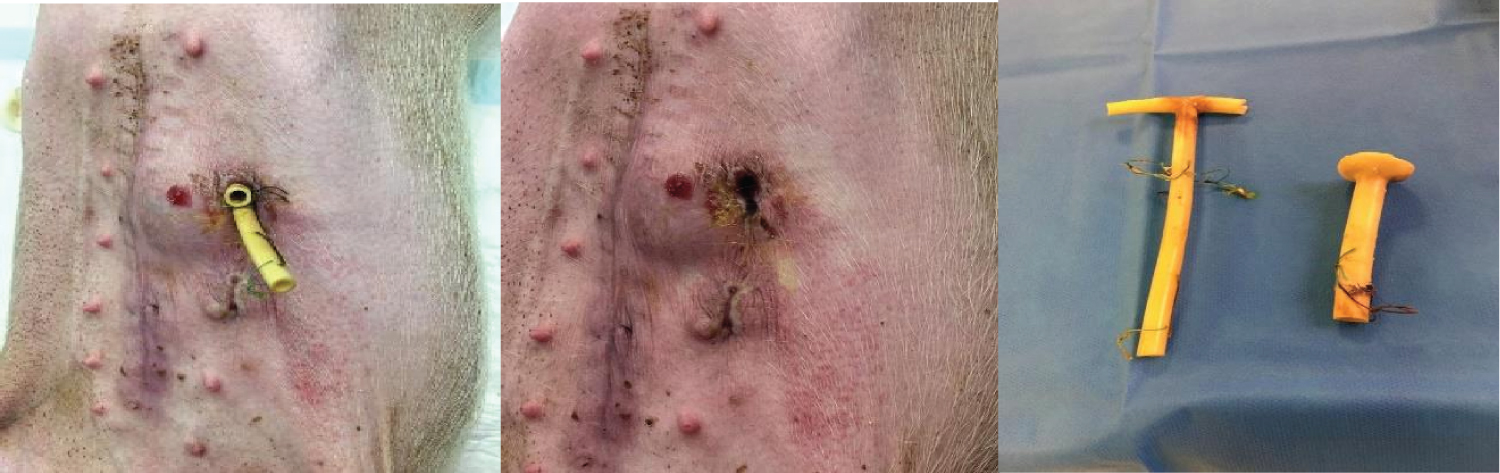 Figure 9: The fistulous orifice in the animal's external abdominal wall. Kehr drain and Pezzer Probe were used in the experiment.
View Figure 9
Figure 9: The fistulous orifice in the animal's external abdominal wall. Kehr drain and Pezzer Probe were used in the experiment.
View Figure 9
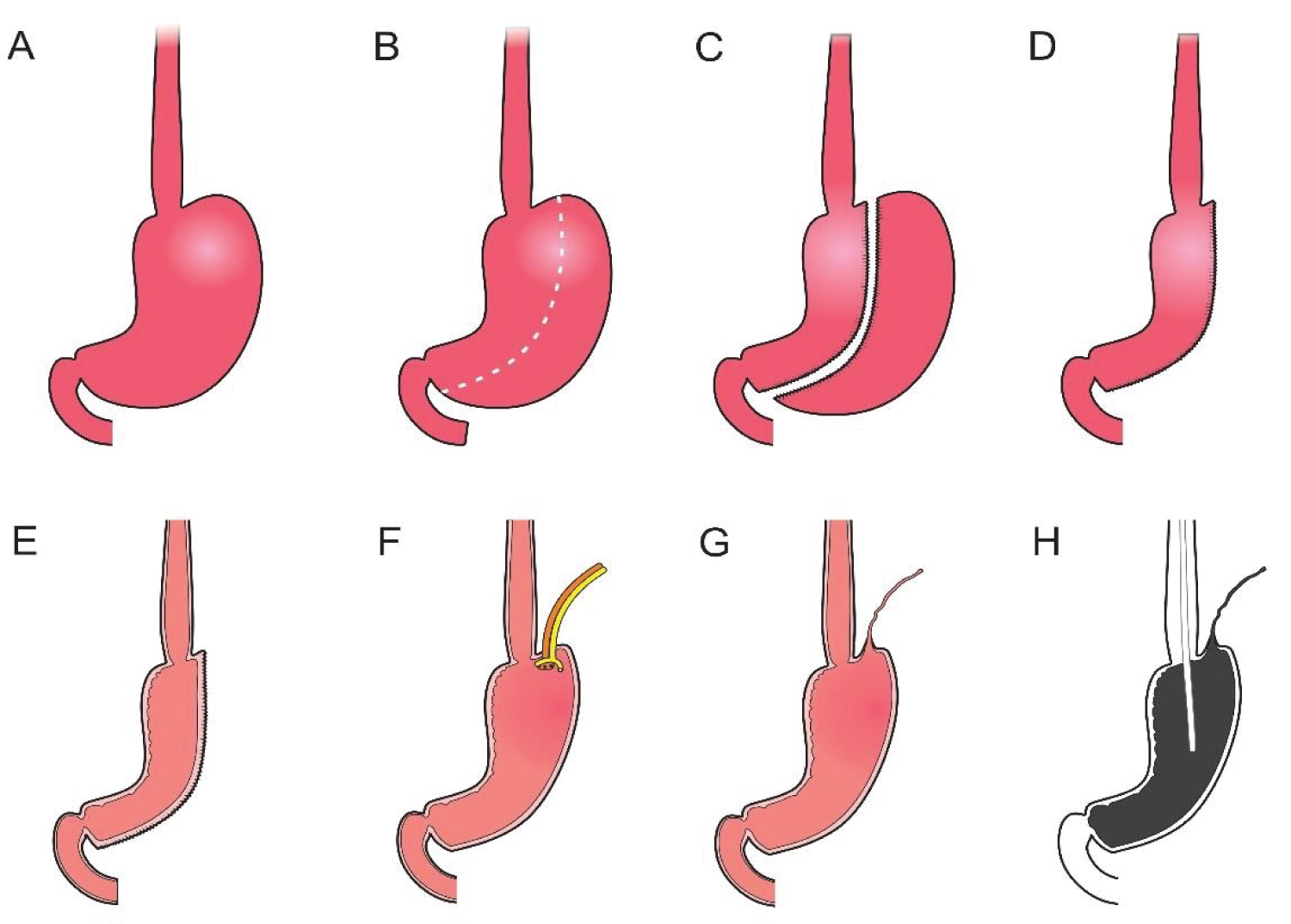 Figure 10: A) Pig stomach; B) Delimitation of the gastrectomy; C) Gastrectomy; D) Reduced stomach; E) Reduced stomach lumen; F) Placement of a Kehr 20Fr "T" Drain next to a 22Fr Pezzer Probe; G) Fistulous path formed; H) Radiological control of the fistulous path formed after the tubes were removed.
View Figure 10
Figure 10: A) Pig stomach; B) Delimitation of the gastrectomy; C) Gastrectomy; D) Reduced stomach; E) Reduced stomach lumen; F) Placement of a Kehr 20Fr "T" Drain next to a 22Fr Pezzer Probe; G) Fistulous path formed; H) Radiological control of the fistulous path formed after the tubes were removed.
View Figure 10
Postoperative complications will always be a reality in the clinical practice of surgeons. Fistulas are a permanent challenge, as they can progress to abscesses, peritonitis, septic shock, and death of affected patients [12-14].
Regarding morbidly obese patients undergoing bariatric surgery, the appearance of the postoperative fistula is added to hypertension, diabetes, and other comorbidities present in these patients, greatly increasing morbidity and mortality [19,20].
Understanding the mechanisms involved in the appearance of postoperative fistulas, risk factors, and the diagnosis and knowledge of early forms of treatment are paramount to increasing survival in obese patients with fistulas when it comes to bariatric surgery [15-17].
In this sense, the experimental model demonstrated in the present study presents itself as a viable and scientifically validated alternative to improve the therapeutic management of fistulas after sleeve gastrectomy [21,22].
We chose to use pigs because they are animals similar to humans in terms of anatomy, genetics, and physiology and, therefore, serve the purpose of replacing them in clinical tests, mainly in the application of new techniques and therapeutic modalities, until reaching the desired safety and efficacy, before being applied in daily clinical practice [3,4,8-10].
Another important aspect is that the present experimental model of fistula after sleeve gastrectomy is similar of what occurs in daily clinical practice, where fistulas arise more frequently at the Angle of His (esophageal-gastric junction) [23].
Added to this is that it is a safe experimental model, easy to perform, low cost, reduced morbidity and mortality, present weight loss, and requires little observation time regarding the formation of the postoperative fistulous path. In addition, to reproduce in a very similar way to what occurs in humans, a fact proven through fistulography and endoscopic control [15,20-22].
Currently, studies demonstrate several strategies in fistula treatment after sleeve gastrectomy. Such research ranges from the use of stem cells, the use of self-expandings tents, interventional radiology, the transformation of Sleeve gastrectomy in to a Roux-Y-shaped by pass, addition to the most used which is the endoscopic approach with the use of catheters to direct the fistula, suction mechanisms, even performing total gastrectomy in cases considered to be of high severity, among others [14,23-25].
It is important to emphasize that the study has limitations, as there was not enough time to complete other stages due to the pandemic caused by COVID-19, which forced the suspension of the different stages of the research, such as the performance of the procedure by video laparoscopy in addition to the biophysical chemical analyzes proposed in the initial project, since the experiments started in November 2019 with an estimated time of duration until June 2020.
However, we believe that we have collaborated in some way to disseminate our preliminary results since we were at risk of losing data and, therefore, of the opportunity to contribute, through the publication of our results, to the dissemination of scientific knowledge.
In conclusion, based on the methodology used, the experimental model of post-Sleeve Gastrectomy fistula is efficient, effective, and reproducible and can be used in future studies to test new diagnostic methods and, above all, innovations in the therapeutic management of postoperative fistulas of the obese patients undergoing mesh surgical technique.
The authors thank the Federal University of Rio Grandedo Norte, Potiguar University, Paris Diderot Foundation, and Liga Contrao Cancer for supporting this study.