Aim: The aim of this study is to investigate the effects of ceteareth-20 and paraffinium liquidium on cell viability and cytotoxicity in human lymphocyte in vitro.
Methods: We studied the cytotoxic and inhibitory effects of ceteareth-20 and paraffinium liquidum on cell proliferation using lactate dehydrogenase (LDH) assay and cell proliferation (WST-1) assay.
Results: The cytotoxicity was enhanced when cells were treated with 1%, 5%, 25% and 50% paraffinium liquidium dilutions (p < 0.05). Moreover, cell number significantly reduced after 24 hours when they were treated with the same concentrations of paraffinium liqudium (p < 0.05). On the other hand, ceteareth-20 at concentrations at 1%, 5%, 25% and 50% showed no significant cytotoxic effect. These results showed that paraffinium liquidum dilutions have cytotoxic and proliferative effect otherwise ceteareth-20 dilutions have only proliferative effects on cultured human lymphocytes.
Conclusion: It can be said that paraffinium liquidum and ceteareth-20 is harmful to use in beauty and cosmetic products. Future studies might allow alternative agents to be used instead of paraffinium liquidum and ceteareth-20.
Paraffinium liquidum, Ceteareth-20, Cytotoxicity
The perception of beauty is being impacted by the culture, society and geography, changing and evolving continuously. There are a lot of beauty and personal care products available, but these products are mainly used by people without paying much attention to the chemicals listed in the ingredients. Currently, a lot of cosmetic products include chemical ingredients to increase their cosmetic properties, conserve their effectiveness, and generate a more viable product. Unconscious use of these chemical ingredients can increase the risk for sensitization [1,2]. Previous studies showed that chemical ingredients in cosmetic products have a potential to cause numerous health problems including irritation [3], inflammation [4], allergic contact dermatitis [5]. Some of the chemical additives used in beauty and personal care products were determined as carcinogenic in animals and in vitro [6-8]. Although the cosmetic product is applied in very low amount or the chemical ingredient present at low concentrations within the product, the product can cause unwanted skin reactions or even more serious health problems [9]. However, many chemical additives are not investigated in terms of their genotoxicity and cytotoxicity.
Paraffinum liquidum is a widely used chemical ingredient in cosmetic and personal care products. It has many common names used in the industry such as heavy mineral oil, light mineral oil, liquid paraffin, liquid petrolatum, mineral oil mist, paraffin oil, mineral oil, petrolatum liquid, petroleum oil, white mineral oil and white oil [10]. Paraffinum liquidum has a complex combination of highly refined, saturated branched-chain and napthenic hydrocarbons that are used in medicinal cosmetics, food and pharmaceuticals [11]. Molecular weight of paraffinum liquidum is determined as 423 g/mol and there is no genotoxicity and cytotoxicity studies in the literature. Another substance of unknown genotoxicity and cytotoxicity is ceterareth-20, which is the polyethylene glycol ether of cetearyl alcohol. It is known as PEG-20 cetostearyl alcohol, peg-20 cetyl/stearyl ether, polyethylene glycol 1000 cetyl/stearyl ether, polyoxyethylene (20) cetyl/stearyl etherceteareth. Ceteareth-20 has a molecular weight of 70.49 g/mol and it is dissolved in water and alcohol to form a colloid solution. Commercial Ceteareth-20 might contain toxic impurities such as 1,4-dioxane [12] and it is used by cosmetic products such as hair dyes [13], shampoos [14], body creams [15]. The aim of this study is to examine the effects of ceteareth-20 and paraffinium liqudium which are content of water based hair dyes, on cell viability and cytotoxicity in human lymphocyte in vitro.
RPMI, FBS, Ficoll, L-glutamine, Penicilin, Streptomycin were obtained from Biochrom AG (Mannheim, Germany) and Biological Industries (Kibbutz Beit Haemek, Israel). WST-1 and LDH kits were purchased from Roche (Mannheim, Germany). Ceteareth-20 and paraffinium liqudium were provided by Doga Pharmacy Company, Istanbul.
PBMCs were isolated from heparinized blood samples using Ficoll solution by density gradient centrifugation. RPMI1640 medium with 2 mmol/L L-glutamine, 10% fetal bovine serum and antibiotics (penicillin and streptomycin) were added into PBMCs. Cells were incubated in the presence of 0.5% CO2 at 37 °C at 1.0 × 106 cells per mL with Ceteareth-20 and Paraffinium liqudium concentrations of 1%, 5%, 12.5%, 25% and 50% for 24 and 48 hours, respectively [16].
Cytotoxic effects of Ceteareth-20 and Paraffinium liqudium extracts were shown by the activity of released lactate dehydrogenase (LDH) from damaged cells. Cells were added as 1 × 106 cell/mL in 96-well plate. Cells were incubated with different concentrations of ceteareth-20 and paraffinium liquidium at 37 °C and 5% CO2 for 24 and 48 hours, respectively. The procedure of LDH kit (LDH; Roche) was implemented and the reaction mixture was put into all wells (100 μL). Then, they were incubated for 30 minutes at room temperature. The 96-well plate was incubated in the darkness 24th and 48th hours and absorbance was measured at 490 nm with ELISA reader (BioTek-PowerWaveS, USA). Assays were dublicated. Cytotoxicity was analyzed according to the following formula: Cytotoxicity (%) = (experimental value - negative control)/(positive control - negative control) × 100 [17].
The cell viability was calculated by using WST-1 assay and cells were added as 1 × 106 cell/mL equality in 96- well plate. They were incubated with different concentrations of Ceteareth-20 and Paraffinium liqudium at 37 °C and 5% CO2 for 24 h. WST-1 was put in a 10 μL/well volume. All producedures were implemented according to commercial company procedure. Then the cells were incubated for 4h in a humidified atmosphere (37 °C, 5% CO2). Absorbance of samples was calculated at 420 nm using ELISA reader (BioTek-PowerWaveXS, USA). Means ± SD values were shown in Figure 1 and Figure 2 [18,19].
LDH activity and WST-1 assay results were analysed using Mann-Whitney U-test. A value of P less than 0.05 was accepted as statistically significant. Results were stated as mean ± SD. For these procedures, SPSS 20.0 version for Windows (SPSS Inc, Chicago, Illinois, USA) was used.
The cytotoxic effects of Paraffinium liqudium (PE) in different doses on cultured human lymphocytes after 24 hour is represented in Figure 1. All of PE concentrations used caused LDH to be released, resulting in cytotoxicity. Increases in the LDH level were observed after the cells were treated with, 1%, 5%, 12.5%, 25%, 50% dilutions of PE when compared with negative control groups. This increase was found to be statistically significant (P < 0.05). The highest cytotoxic effect of PE was observed in the 50% dilution. The cytotoxic effects of different doses of cetereath-20 on human lymphocyte cells after 24 hour represented in Figure 2. Ceteareth-20 at concentrations 1%, 5%, 25% and 50% have no significant cytotoxic effect on cultured human lymphocytes but it has only proliferative effects on cultured human lymphocytes.
Figure 1 and Figure 2 represent the results of the cell viability assessments, including the WST-1 assay of the control and experimental groups. Decreases in the cell proliferation was observed after treatment with different concentrations of Paraffinium liqudium. For cetereath-20; there was significant decreased after treatment at different concentrations of CE. Statistical analysis showed a significant difference in cell viability between the Paraffinium liqudium treated group and control group after 24 hours of incubation. WST-1 results show that the cell proliferation was decreased when cells were treated with both Paraffinium liqudium and cetereath-20 concentrations after 24 hours (P < 0.05).
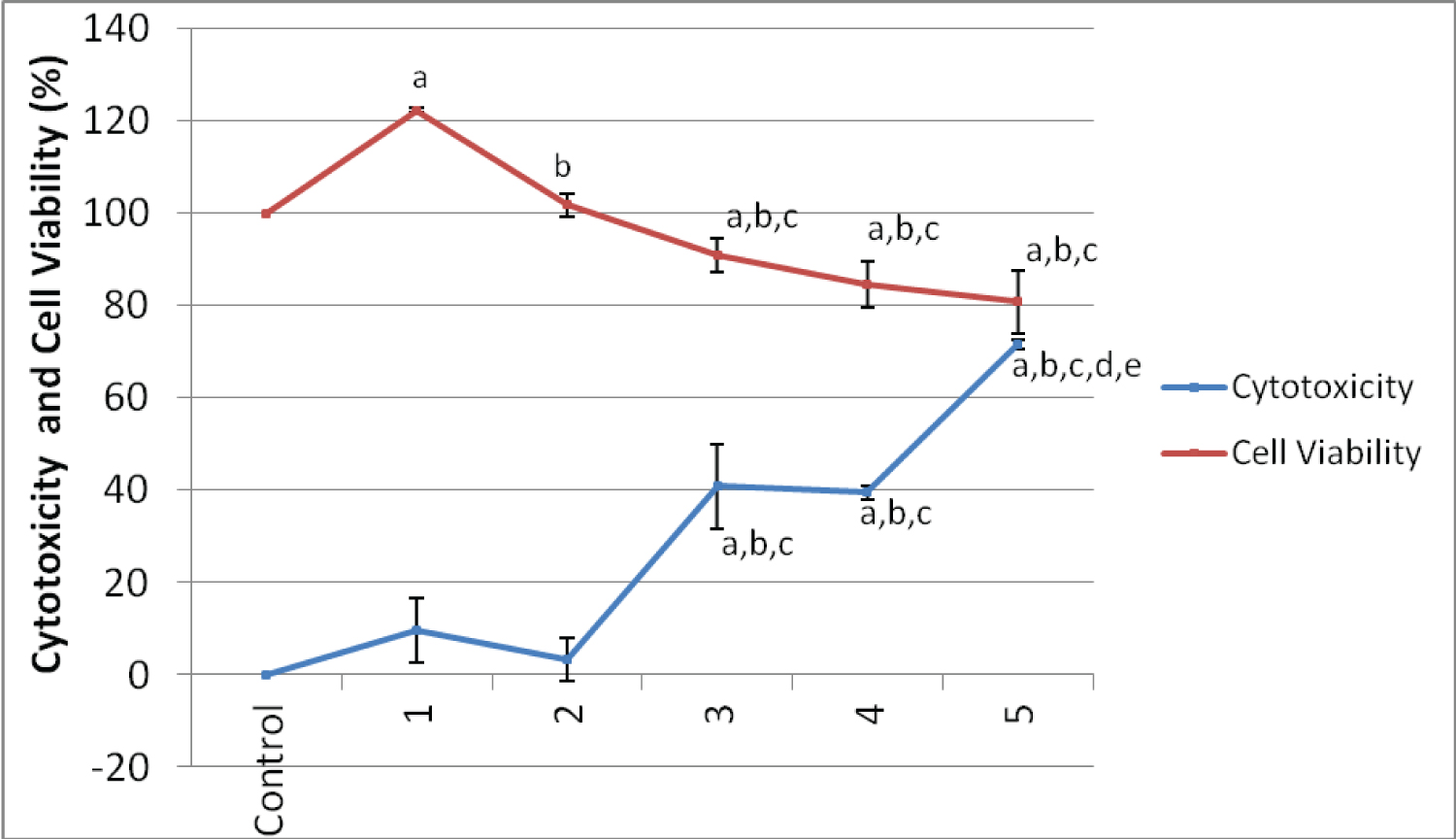 Figure 1: Cytotoxicity effect and Cell Viability at different concentrations of Paraffinium liqudium dilutions (PD) on human lymphocyte cells in vitro. View Figure 1
Figure 1: Cytotoxicity effect and Cell Viability at different concentrations of Paraffinium liqudium dilutions (PD) on human lymphocyte cells in vitro. View Figure 1
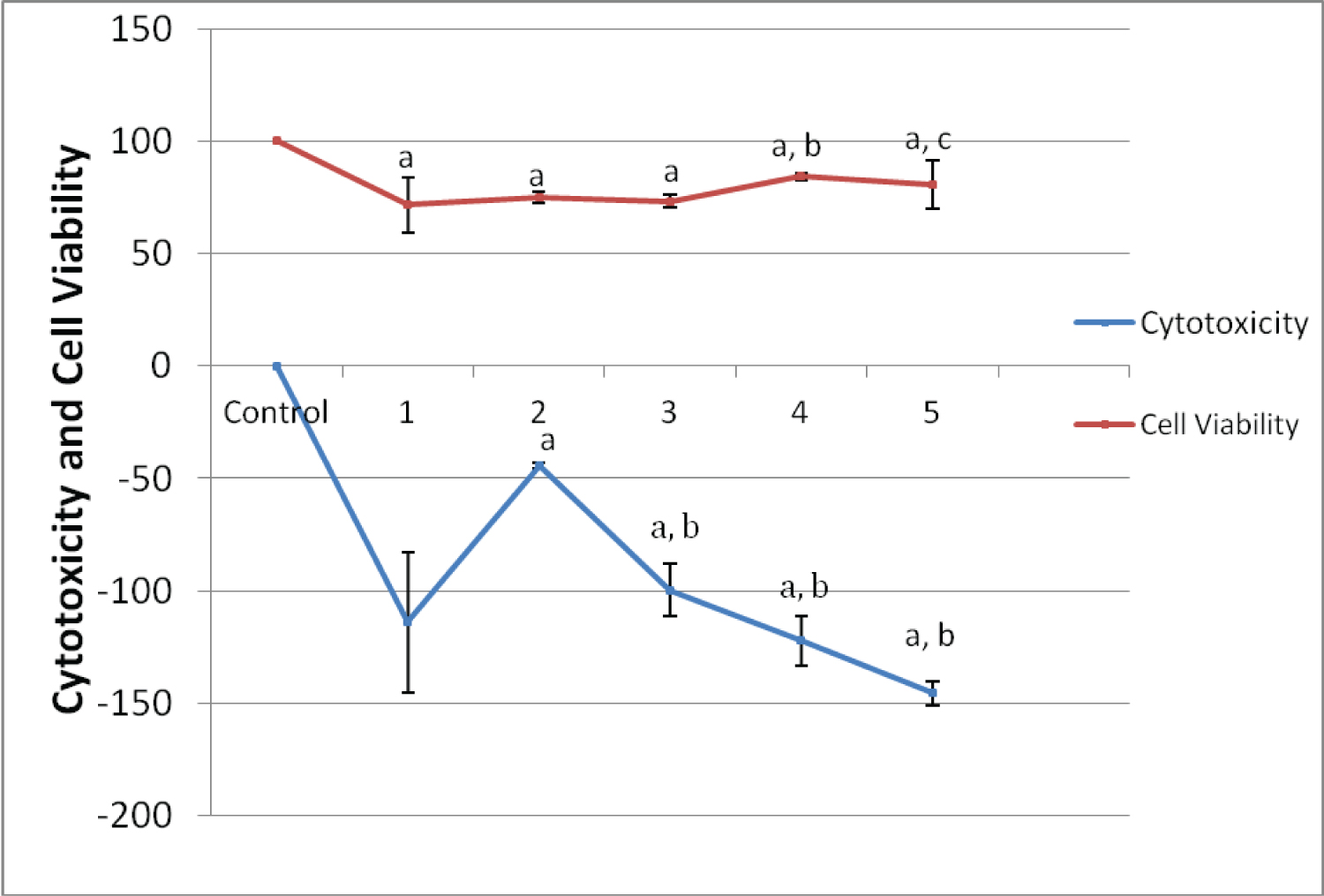 Figure 2: Cytotoxicity effect and Cell Viability at different concentrations of Cetereath-20 dilutions (CD) on human lymphocyte cells in vitro. View Figure 2
Figure 2: Cytotoxicity effect and Cell Viability at different concentrations of Cetereath-20 dilutions (CD) on human lymphocyte cells in vitro. View Figure 2
ap < 0.05 compare with control group, bp < 0.05 compare with PD-1-treated group, cp < 0.05 compare with PD-2-treated group.
ap < 0.05 compare with control group, bp < 0.05 compare with PD-1-treated group, cp < 0.05 compare with PD-2-treated group, dp < 0.05 compare with PD-3-treated group, ep < 0.05 compare with PD-4-treated group.
ap < 0.05 compare with control group, bp < 0.05 compare with CD-2-treated group.
ap < 0.05 compare with control group, bp < 0.05 compare with CD-2-treated group, cp < 0.05 compare with CD-3-treated group.
Beauty and personal care products are directly contacted to the skin and exposure is the most significant topic [20]. People are used to a lot of beauty and personal care products in daily life and these products contain many chemical additives. Many researchers have indicated that cosmetic products can contain potential toxic agents [21-23]. Approximately 10,000 ingredients are found in beauty and personal care products and can be directly associated with too many diseases such as cancer, genetics disorders and birth defects. US FDA (Food and Drug Administation) forbid some chemical additives like glycol, lead, mercury, formaldehyde [24].
Cytototoxicity and genoxocity of cosmetic products can described, distinguished and evaluated both in vivo and in vitro methods [25-27]. Thus, we studied the cytotoxic and inhibitory effects of ceteareth-20 and paraffinium liquidum on cell proliferation, which were evaluated using LDH assay and WST-1 assay in this study. Ceteareth-20 and paraffinium liquidum are found in many beauty and personal care products, but there is no previous investigation of their inhibitory effects on cell proliferation. Baby oils, body and face creams, hair dyes, shampoos contain paraffinium liquidum and ceteareth-20. Many famous brands use them, because they are not soluble in the water. Some researchers have thought that paraffinium liquid is a probable carcinogen or cancer-causing agent which it contains 1,4 dioxane [28]. Ceterareth-20 is used in many application that increases the viscocity [29]. In our study; paraffinium liquidum dilutions had cytotoxic and proliferative effect otherwise ceteareth-20 dilutions have only proliferative effects on human cultured lymphocytes. On the other hand, ceteareth-20 at concentrations 1%, 5%, 25% and 50% have no significant cytotoxic effect but it has only proliferative effects on cultured human lymphocytes. Because of its molecular formula and it can be bind cell membrane on human cultured lymphocytes.
As a consequence we could say that paraffinium liquidum and ceteareth-20 can be harmfull to use in water based beauty and cosmetics cosmetic products. So instead of paraffinium liquidum and ceteareth-20, alternative agents can be used. Next research on paraffinium liquidum and ceteareth-20 will be study by confirming our results with using genotoxicity assays.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
There are no conflicts of interest.