Oreochromis niloticus were subjected to 30 days of α-Olefin Sulfonate sublethal toxicity test after which samples of brain, liver, and blood were collected for biochemicals evaluations. The brain under control treatment showed higher specific acetyl cholinesterase activity than the blood. Nonetheless, AChE activities in the brain and erythrocytes of O. niloticus exposed to sublethal concentration of α-OS revealed that the toxicant decreases the activities of the enzyme in both tissues, and the inhibition is concentrations and time dependent and was more in the brain than the erythrocytes. Though, there were inhibitions in all the treatments but were not significant (P ≥ 0.05) when compared with the control except 2.0 mg/L in both tissues. The Catalase (CAT) and glutathione peroxidase (GPx) activities were not significantly (p ≥ 0.05) decreased on the day 2nd of exposure α-OS, but drastically increased compared to their control on day 30th. The ALP activity increases with doses and exposure durations. There were slight changes in the cortisol levels in the control group, while in the exposed groups, the cortisol levels increase spontaneously with the dosage and duration of exposure to α-Olefin. Organophosphates are major agrochemicals that strongly affect different neuroenzymes, antioxidants enzymes and the endocrine of various fish species, but surfactants possess similar attributes to these pesticides. In the present study, AChE activities in the brain, and blood together with the liver functions enzymes, and cortisol levels in the serum after exposing fish to α-OS showed that the enzymes can be used as a biomarker for observations of surfactant contamination in natural water. This study showed that α-OS poisoning may have similar effects on human health as well, and can ultimately cause serious damage to human life. As major sources of surfactant exposure are anthropogenic, hence concerned authorities must impose strict mitigation measures to control this hazard.
Oreochromis niloticus, α-Olefin sulfonate, Neurological stress, Endocrine disruption, Acetyl cholinesterase activity, Cortisol, Blood
Various products created with chemicals improve the quality of modern living, but they also raise worries about the potential dangers they pose to the environment. Anionic, non-ionic, cationic, and amphoteric are the most widely employed surfactant classes today in both commercial and residential settings. Surfactants, which are commonly present in many industrial and domestic cleaning formulations, continue to find their way into our rivers.
Detergents are normally discharged into the environment via sewage treatment infrastructure (sewers, sewage treatment plants) or directly in cases where no treatment systems are available [1]. Their usage and disposal frequently contaminate the aquatic habitat, which typically serves as an effluent recapture system. When detergents are dumped into rivers or lakes without being purified, they can contaminate water and be hazardous to aquatic organisms. They are among the commercial chemical products that pollute water and are toxic to aquatic organisms [2].
Surfactants' surface action against biological membranes is what causes their ecotoxicity [3], and are divided into cationic, anionic, non-ionic, and amphoteric categories based on their chemical makeup, which determines how they affect aquatic and terrestrial life [4]. α-Olefine sulfonates (AOS) are utilized in a variety of ways including laundry powder detergents, liquid dishwashing detergents, and hair shampoos. Chemicals like norust CR 486 (corrosion inhibitor) and Neatex (detergent) are used to clean oil pipelines because they contain AOS that aids with the removal of oil stains and soils from surfaces [3].
Indiscriminate deposition of effluents/toxicants into an aquatic ecosystem can impair respiration in aquatic organisms, leading to asphyxiation (which is an indication of unconsciousness or depth caused by failure of blood to become properly oxygenated in lungs) and may eventually result in organ architectural degradation such as liver dysfunction and enormous stress [5,6]. Stress reactions and behavior appear to show that fish experience pain and suffering, primarily near the end of their lives [7]. The response varies depending on the nature of the stress and the species of fish [8]. The stressor drives changes in stress hormones such as plasma cortisol, which rise quickly and return to normal after a period.
Anionic surfactants have been shown to cause inflammation and oxidative damages [9-11], and toxicity studies of AOS on aquatic animals, as well as the influence of aquaculture-related stresses, have shown that fish can be predisposed to disease when exposed to this surfactant [12]. Fish showed a range of physiological changes in response to stressors, with the main stage involving catecholamine release and activation of the corticotrophin interregnal axis, and the secondary stage involving haematological, metabolic, blood enzymatic, and osmoregulatory alterations [13]. In addition to stimulation of oxygen uptake and transfer, mobilization of energy substrates, reallocation of energy away from growth and reproduction, and primarily suppressive effects of immune functions, the tertiary stage resulting from chronic exposure of fish to stressors is susceptibility to pathogens and behavioral changes [14].
The tilapia is one of the most widely cultivated freshwater fish in Nigeria. Despite being an exotic species, this fish is immune to water-soluble contaminants and insoluble particles at the air-water interface [15]. Precipitated pollutants may be less accessible to tilapia, as they have less contact with this part of the aquatic environment and spend the majority of their time in the upper water column. Due to its ease of handling, adaptation to confinement, laboratory maintenance, susceptibility to various pollutants, and economic importance, the species O. niloticus has been widely used for environmental studies as well as assessing the toxicity of contaminants in aquatic ecosystems.
The goal of this study was to see how sublethal concentrations of Alpha Olefine Sulfonate affected acetyl cholinesterase activity and cortisol levels in the plasma and serum of O. niloticus, a commercial and ecologically significant fish species in the Niger Delta of Nigeria.
The fish experiments were conducted following the applicable standards and laws. The O. niloticus utilized in this study were observed from the egg stage till the appropriate maturity stage was reached. The fish was spawned in clay ponds to create pure fry. Spawning took place in April and May 2021 in a well-built shallow pond filled with one male to five females. The following conditions and physicochemical parameters of the water in the aquariums were maintained throughout the experiment: The optimal temperature range of 25-29 °C was maintained, daily illumination of 12-14 hours with a 100-W fluorescent bulb suspended 50 cm above the water surface was maintained, the breeding stocks were fed a protein- and vitamin-rich diet, and the spawning aquariums were cleaned every 4-5 days.
The mating procedure begins when the male selects the best-developed female and pushes the remaining females to the farthest corner of the tank. The entire spawning and fertilization process take up to two hours. The female immediately departs the aquarium's nest area with the fertilized eggs in her mouth after spawning.
During the incubation stage and until the newly hatched fries have completely absorbed the yolk sac, the females do not eat because their mouths were full of eggs and then newly hatched fry. Immediately following the female's departure from the nest, the male chooses, and another female repeats the same process.
After the fry absorbed the yolk sac, they were transported to a nursing pond until they reached maturity. Fry consume on their own, acquire the form of fish, and grow to a length of 1-2 cm. When a fry reaches a size of 10-15 cm or roughly the size of a finger, it is referred to as a fingerling. It takes approximately 45-60 days for fry to reach fingerling size. Fry and fingerlings were fed a tailored diet in the form of soybean powder or finely powdered cake and rice bran 4-5 times per day at a ratio of 1:1. After two to three weeks of stocking, the nursery pond is drained and advanced fries were transferred to the rearing pond for fingerling development.
A greenhouse was built to represent the fish's natural habitat, and it was cleaned daily. Clayey loam soil was utilized to construct fifteen earthen ponds, each measuring 27 1/4 inches by 24 1/8 inches by 29/12 inches. Each of the final ponds received ten fingerlings, which were fed a finely powdered cake and rice bran three times per day for 12 weeks. After cleaning the aquariums with a manual pumping device to remove stale water, the water was replaced twice per week. After the 12th week, no deaths were observed.
At the end of the 12th week, each of the final ponds and their replicates was exposed for 30 days to the range of α-Olefin Sulfonate concentrations detected in the field (0.5, 1.0, 1.5, and 2.0) mg/L and the control. Throughout the tests, both control and experimental fish were fed roughly 3% of their body weight twice every day. Water and toxicants were entirely replaced every 24 hours, and the earthen ponds were kept in immaculate condition.
Throughout the investigation period, daily measurement of the water's physicochemical properties was taken. After each study period, a fish is retrieved from each pond, transported to the laboratory in a container with adequate ventilation, and anesthetized with MS222 (Ethyl 3-aminobenzoate methanesulfonate salt, Sigma).
In the laboratory, biochemical profiles were measured on days 2 and 30. The unutilized fish were returned to the main pond.
AChE activity was measured using a commercial kit manufactured by Bohringer Mannhim, which was based on the Ellman spectrophotometric assay procedure adapted for microplates as described by Galloway, et al. (2003) and used acetylthiocholine iodide as a substrate.
Using a 1-ml insulin syringe, blood was drawn from fish and put into heparinized tubes containing 66 L of 0.1 M sodium-EDTA. The erythrocyte was centrifuged at 4 °C for an hour, separated, and then resuspended in 0.5 mL of potassium phosphate (12.5 mM, pH 7.4) after two washes with 0.9% NaCl for the enzyme test [16].
The brain was homogenized (1:10, w/v) at 9500 rpm for 1.5 minutes in a homogenization buffer composed of 100 m KCl and 1 m EDTA (pH 7.4), and the supernatant was then separated by centrifuging the homogenates at 10,000 g for 30 minutes at a temperature of 4 °C.
Throughout the experiment, 50 M Tris-HCl with a pH of 7.4 was used as a buffer. Acetylcholine (ATC) was produced in 50 M Tris-HCl in 96-well plates containing 5, 5-dithiol-bis-2-nitrobenzoic acid (DTNB). I. 210 L of Tris-HCl (pH 7.4) buffer, 20 L of 0.1 M DTNB, and 10 L of theliver's acetyl cholinesterase enzyme. The supernatant was transferred to another 96-well plate II and incubated at 28 °C for 15 minutes. The mixture was then treated with 10 L of ATC (2.5 M) in 96-well plate II, followed by a 10-minute incubation.
Thiocholine is produced when the cholinesterase enzyme hydrolyzes the substrate acetylthiocholine. Thiocholine and Ellman's reagent (DTNB) react to form 2-nitrobenzoate-5-mercaptothiocholine. A reading was taken at 405 nm using a microplate reader (Tecan Multimode Microplate, United Kingdom) with fluorescence excitation at 485 nm and emission at 535 nm [17]. The enzymatic activities of erythrocytes were measured in mol/min percent hematocrit, while those of the brain were measured in mol/min/mg protein. Each unit of activity corresponded to 1nmol of substrate hydrolysis per minute. The hematocrit level was measured using capillary hematocrit tubes.
The liver was carefully excised from the fish sample and the supernatant used for the analysis follow the same procedure as described above.
Catalase was evaluated by measuring the decrease of H2O2 concentration at 240 nm [18], with one unit of CAT activity equal to the decomposition of one mol of H2O2/min/mg protein.
The activity of glutathione peroxidase (GPx) was measured using Tert-butyl hydroperoxide and GSH as substrates as described by [19]. The changes in absorbance intensity at 340 nm were used to monitor the conversion of NADPH to NADP+ caused by the reduction of GSSG to GSH by GR. Using a molar extinction coefficient of 6.22 × 103 M-1 cm-1, one unit was expressed as one µmol mol of NADPH consumed each minute.
The alkaline phosphatase (E.C. 3.1.3.1) analysis was carried out with slight changes, as previously described by Garen and Levinthal (1960) using the Reitman and Frankel colorimetric method. Para-Nitrophenylphosphate (p-NPP), sample, 0.1 MgCl2, and bicarbonate buffer (0.1 M, pH 9.2) are all included in the enzyme assay mixture. After that, the reaction mixture was incubated for 15 minutes at 25 °C before being stopped with 0.1 N NaOH. At a constant temperature of 25 °C, one unit of ALP activity was defined as nanomoles of p-nitrophenol released/min/mg protein.
The plasma cortisol levels in the investigated fish were measured using a human cortisol ELISA test kit. For standard curve calibration, seven cortisol standard solutions in human plasma (0, 20, 50, 100, 200, 400, and 800 ng.ml-1) were used. The recovery test used cortisol standard solutions of 50, 100, and 200 ng.ml-1, and the linearity test used four dilutions of fish plasma samples (1/2, 1/4, 1/8, and 1/16). Plasma samples and standard solutions were added to each well fish before adding the enzyme-substrate, and the content was conjugated to peroxidase. After stopping the enzymatic reaction with 0.5 M phosphoric acid, the absorbance at 450 nm was read.
Microsoft Excel 2010 (Roselle, IL, USA), SAS (SAS institute Inc, 1985), SPSS version 16.0 (Chicago, IL, USA), and SPSS, version 10 (SPSS, 2016) software were used for the statistical and graphical evaluations. For comparisons between the treatment and control groups at a probability level of 0.05% and 0.01%, post hoc analysis was used to determine the least significant difference (LSD).
AChE activities in the brain and erythrocytes of O. niloticus exposed to sublethal concentration of AOS revealed that the toxicant decreases the activities of the enzyme in both matrixes. Inhibition is concentration dependent on day 2. At the concentration of 0.5, 1, 2 mg/l of AOS in the brain and the red blood cells the activities of the enzyme were; 45.12, 44.9, 41.23, (unit) and 26.11, 25.12, 24.2, and 20.23 respectively (Figure 1). On day 30 the same trend was observed. The high inhibition of 23.40 mol/min/mg protein and 17.10 mol/min percent hematocrit were reported for brain and erythrocytes respectively (Figure 1). No significant (P ≥ 0.05) in both matrixes, duration, and treatments except 2.0 mg/L of α-OS in both tissues. In the presence of α-OS, the specific activity of catalase and GPx diminished at the initial exposure as observed on day 2nd in doses dependent manner. On day 2, the GPx activities at 0.00, 0.50, 1.00, 1.50 and 2.00 mg/L of AOS were: 3.53 ± 0.11, 3.10 ± 0.50, 2.70 ± 0.30, 2.62 ± 0.20 and 2.13 ± 0.40 µM/min/mg protein, while on day 30th, the activities were; 3.51 ± 0.23, 4.60 ± 0.57, 5.30 ± 0.91, 6.10 ± 0.52 and 6.60 ± 0.42 µM/min/mg protein.
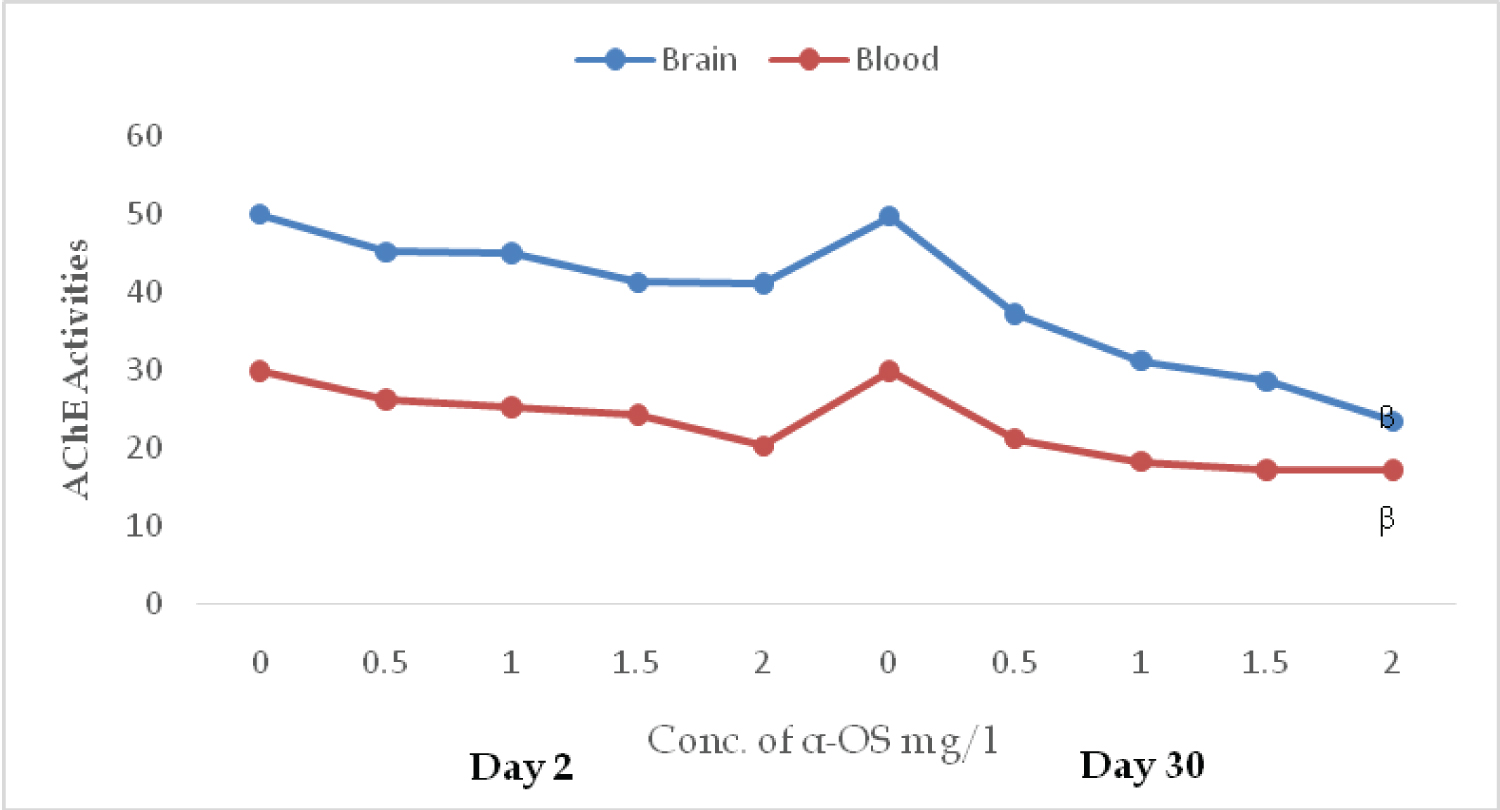 Figure 1: AChE activities in the brain (µmol/min/mg protein) and erythrocytes (µmol/min % hematocrit) in O. niloticus exposed to sublethal concentrations of alpha olefin sulfonates; A symbol shows a significant differences β(p < 0.05 between the control and treatment. View Figure 1
Figure 1: AChE activities in the brain (µmol/min/mg protein) and erythrocytes (µmol/min % hematocrit) in O. niloticus exposed to sublethal concentrations of alpha olefin sulfonates; A symbol shows a significant differences β(p < 0.05 between the control and treatment. View Figure 1
The CAT activities on days 2nd and 30th with the same concentrations of α-OS were 13.20 ± 0.14, 6.50 ± 0.30, 8.20 ± 0.31, 7.10 ± 0.22, 4.80 ± 0.15, and 13.50 ± 0.60, 16.10 ± 0.73, 18.40 ± 0.20, 21.80 ± 0.11, and 23.40 ± 0.60 respectively. Although the catalase and GPx activities were not significantly decreased on the day 2nd of exposure, their activities were drastically increased compared to their control on day 30th (Figure 2).
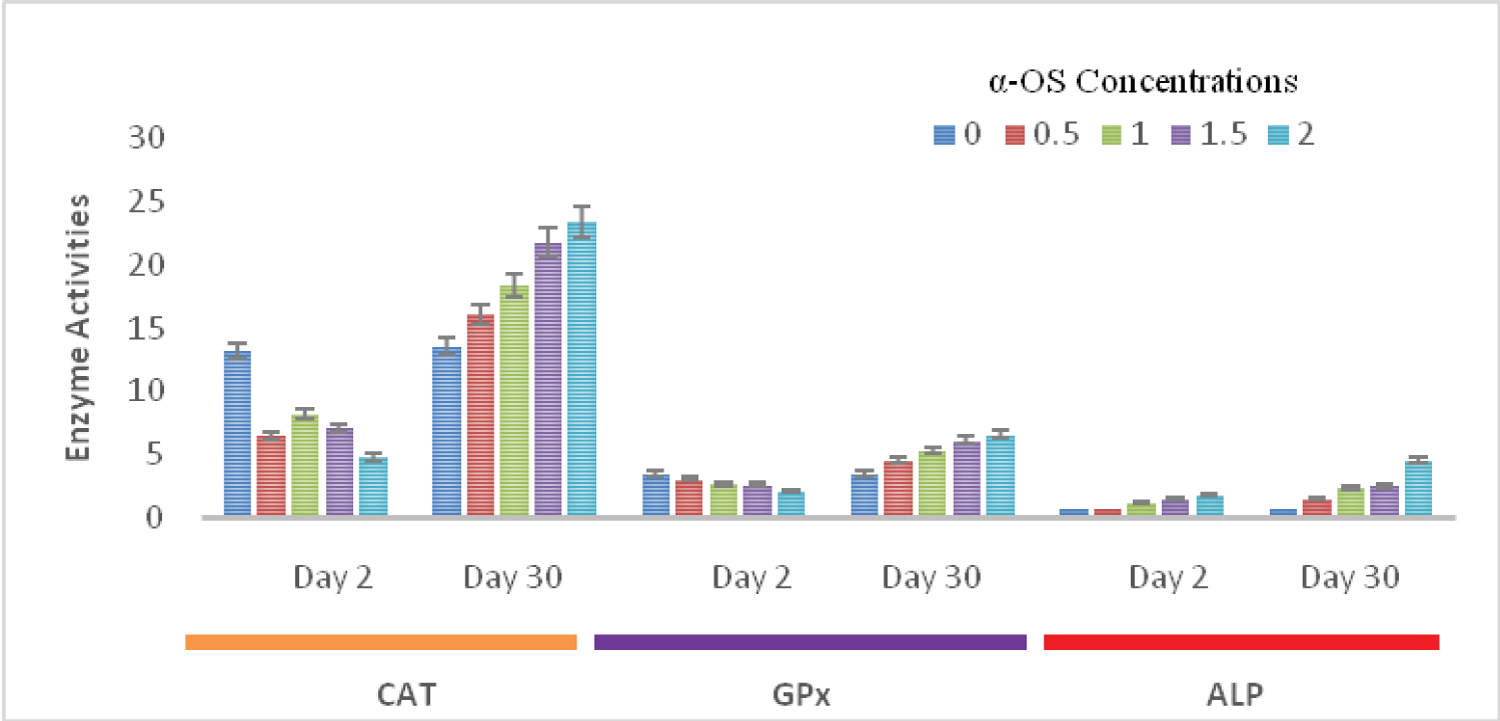 Figure 2: CAT (mM/min/mg protein),GPx (µM/min/mg protein) and ALP (IU/min/ mg-1 protein) activities in the liver of O.niloticus exposed to sublethal concentrations of alpha olefin sulfonates; Data presented as mean ± SE.
View Figure 2
Figure 2: CAT (mM/min/mg protein),GPx (µM/min/mg protein) and ALP (IU/min/ mg-1 protein) activities in the liver of O.niloticus exposed to sublethal concentrations of alpha olefin sulfonates; Data presented as mean ± SE.
View Figure 2
The ALP activity increases with doses and exposure durations. On days 2nd and 30th the ALP activities were 0.70 ± 0.30, 0.81 ± 0.42, 1.20 ± 0.18, 1.52 ± 0.15, 1.86 ± 0.23, and 0.71 ± 0.03, 1.46 ± 0.03, 2.38 ± 0.35, 2.53 ± 0.17, 4.60 ± 0.25 respectively. ALP activities in all the treated fish groups were significantly higher (p ≤ 0.05), when compared with the control on days 2nd and 30th (Figure 2).
There were slight variations in the cortisol level in the control group. In the exposed group, the cortisol level increases spontaneously with the doses and duration of exposure. The range of cortisol in the control group was (3.20 ± 0.20 - 3.87 ± 0.04), while the range of cortisol secretion in fish treated with 0.50 mg/L of α-OS was (4.50 ± 0.11) ng/mL to (7.30 ± 0.15) ng/mL, while it was (4.90 ± 0.10 - 8.40 ± 0.25) ng/m and (5.60 ± 0.18 - 9.30 ± 0.90) ng/mL in fish treated with 1.0 mg/L, 1.50 mg/L, and 2.00 mg/L. On day 2, there was no discernible difference between the control and various treatments (p ≥ 0.05). However, on day 30, it significantly varied (P ≤ 0.05), (P ≤ 0.01) and was concentrations-dependent (Table 1).
Table 1: Responses of hydrocortisone (ng/ml) in the plasma of O. niloticus exposed to sublethal concentrations of Alpha Olefin Sulfonates (mg/L). Data presented as mean ± SE. View Table 1
It is necessary to understand the connections between chemical exposure, biomarker responses, and the negative effects of using a biomarker in biomonitoring. In the case of AChE, these attributes have been well established, and numerous studies found a connection between AChE inhibition and exposure to organophosphorus compounds in a population of exposed workers [20]. To evaluate the effects of these pollutants on physiological, biochemical, and genetic parameters in various fish species, toxicity tests with organophosphate and carbamate pesticides have been widely used [21-23], whereas surfactant research, on the other hand, had been extremely limited, though, surfactants may not be lethal to fish, but they may have negative effects on development, metabolism, and their offspring.
Organophosphates and carbamates affect neuro-enzymes and fish growth, but surfactants have similar properties. In the current study, AChE activities in the brain and blood were examined after fish were exposed to α-OS. It was discovered that changes were observed in the brain and erythrocyte activity in the fish exposed to the surfactant, likely reflected in movement disturbances, with fish lethargic and immobile in the swimming habit, and that these changes help to explain behaviour changes induced by α -OS. Similar findings were made in Anguilla anguilla after exposure to fenitrothion and thiobencarb [24], and silver catfish fingerlings exposed to 10 mg L-1 clomazone for 96 h [25].
Acetylcholine is a neurotransmitter that is degraded by AChE in cholinergic synapses, as well as pseudocholinesterases that are involved in the detoxification of some xenobiotics [26]. Due to excessive ACh buildup at synapses and neuromuscular junctions brought on by its inhibition, ACh receptors may become over stimulated, which may ultimately result in respiratory failure and death [27]. These results suggest that AChE activity may serve as a biomarker for the detection of surfactant contamination in natural water.
The liver is the main organ involved in the metabolism of xenobiotics in animals, and the antioxidant enzymes in the liver are thought to be indicators of hepatotoxicity brought on by different substances. ROS are produced during the xenobiotics' transformation in processes mediated by monooxygenases from the CYP 450 families. To aid in the detoxification of oxygen free radicals and help avoid or reduce macromolecule damage, antioxidant enzyme activities frequently rise when oxidative stress is induced in fish [28,29].
However, it has been demonstrated that free radicals can partially inactivate various enzymes in the early phases; for example, hydrogen peroxide can inactivate CAT, as observed in this investigation. CAT and GPx activity decreased on the second day and increases with time, and the highest activities were observed on the 30th. These enzymes' initial subdued and abrupt increase in activity could be related to an adaptive response. The decrease in CAT and GPx activities at the early stage indicated that the tissue was involved in neutralizing the impact of peroxides formed during the α-OS stress. Visweswaran and Krishnamoorthy [30] reported fluctuations in GPx activity in Tartrazine-treated rats' testis.
Similarly, the decrease in GPx activity correlated with a decrease in catalase enzyme synthesis and can be linked to H2O2 generation under α-OS stress, as described by Vijayavel, et al. [31] in naphthalene-exposed Scylla serrate. Bindu and Babu [32] noticed a similar observation when an Oreochromis mossambicus was given sub-lethal dosages of anionic, cationic, or nonionic surfactants at 1 mg/L. A significant (p ≤ 0.05) dose- and time-dependent increase in GPx activity suggests that α-OS promoted ROS generation in the test fish, which increased the enzyme's level in the liver. Although it is primarily responsible for the elimination of organic peroxides, it is also involved in the metabolization of H2O2 [33].
Dose- and time-dependent increase in CAT activity in the liver under the stress of the surfactant, may have been a consideration to provide defense against lipid hydroperoxides and H2O2. Similar observations were reported when the liver of Carassius auratus was exposed to 3,3'-dimethylbenzidine, nitrobenzene, cadmium, naphthalene, and phenanthrene [34-37].
The results of this study's analysis of liver enzymes reveal that consuming surfactants also raised the serum ALP level, which was a sign of stress, and myocardial infarction. The rise in plasma ALP could be the result of toxic damage brought on by AOS, which induced tissue healing by stimulating protein synthesis and respiration. Similar findings were reported by [38] in a freshwater fish subjected to diethyl phthalate. Changes in fish plasma enzyme activity have been observed as a result of toxicants or pollutants impacting numerous cells, immune systems, tissues, and organs [39,40]. The serum ALP activity in this study was comparable to the findings of [41], who reported a considerable rise in ALP that could be indicative of broad pathological alterations and injury to specific organs of C. gariepinus. Conversely, Inyang, et al. [42] reported a decrease in plasma ALP, in the fishes, although, a sublethal dose of diazinons was administered to the C. gariepinus.
The physiological process that is activated in response to environmental stressors, such as pollutants, is known as homeostasis, and it includes the endocrine response to pollutants. Vertebrates need the hypothalamic-pituitary-adrenal (HPA) axis to be able to respond to stressors. Since fish have interrenal cells in their head kidneys rather than adrenals, this axis is known as the HPI axis in fish, and its output is cortisol, which has both glucose- and mineral ocorticoid-regulating properties. The significant (p ≤ 0.01) rise in plasma cortisol levels during the treatments of O. niloticus to AOS in this study may have been caused by the release of cortisol from the interrenal tissue as a stress-reduction strategy, an abnormal plasma chloride level, or the attempt to return the values to normal. Likewise, since corticoids have been linked to electrolyte balance and gill ATPase activity, elevated cortisol levels are likely related to either causing abnormal chloride and ATPase levels or to the process of attempting to restore the values to normal [43]. According to Scott, et al. [44] when fish were exposed to an alarm substance, a chemical released from skin epithelium, plasma cortisol levels in rainbow trout increased. This increase was prevented by cadmium. Additionally, Hontela, et al. [45] found that copper has a direct toxic effect on adrenocortical cells, which disrupts cortisol secretion at high concentrations. According to [45], the interrenal tissue of teleost fish typically secretes the steroid hormone cortisol in response to nearly all types of environmental stress. Therefore, exposure to toxins that impair cortisol secretion may affect social interactions and restrict the activities of the organism.
This study demonstrated that α-OS acts as an endocrine disruptor and has a significant impact on the fish control system. These enzymes have the potential to be useful biomarkers for detecting surfactant pollution in aquatic bodies. The findings also revealed that the presence of surfactants in water bodies could be detrimental to the health of aquatic animals. To explain the specific differences in the relationship between AChE inhibition, disruption of endocrine, and physiological perturbation associated with alterations of marker enzymes in fish and other aquatic inhabitants, additional research is required.
The impact of surfactant pollution on the aquatic environment has been overlooked. Surfactant toxicity poses a serious threat to ecological and environmental balances. Surfactants like Alcohol Ethoxy Sulfates, Sodium Dodecyl Sulfate, and Nonylphenol Ethoxylates may harm aquatic. Aquatic organisms may have a lower survival rate as a result of their physiological changes. To prevent aquatic ecosystems from being harmed by contaminants that have been carelessly dumped into waterways, precautions must be taken, and more research must be conducted to strengthen the findings that have already been made. For a more complete understanding of their harmful effects on aquatic ecosystems, additional research is needed into their potential residual effects and the use of environmentally friendly surfactants.
Not applicable.
Not applicable.
The manuscript meets all applicable standards concerning the ethics of experimentation and research.
Not applicable.
I would like to state that the research was self-funded and conducted in my research laboratory. The authors hereby certify that the publishing of this paper does not involve any conflicts of interest.
Data generated as part of this study is available upon request from the corresponding author.
All the authors have substantial contributions to the final manuscript and approved this submission.