Pulmonary Vein Antrum Isolation (PVAI) targeting PV triggers is an established treatment for paroxysmal Atrial Fibrillation (AF). Ablation lesions are typically delivered around the entire circumference of each individual PV. This study tested a strategy where electrical inputs to the PVs were mapped using the NavX system and selectively targeted before ablation. The objectives of the study were to evaluate: 1) Likelihood of PV isolation using this strategy, 2) Correlation between manually tagged PV potentials and Complex Fractionated Electrograms identified using the NavX CFE Mean Map in sinus rhythm and during coronary sinus pacing, 3) Long term success of this strategy.
Ablation was initially guided by identification of PV antral regions activating early in sinus rhythm and during Coronary Sinus (CS) pacing, and exhibiting local fractionated electrograms, mapped using the NavX system, and a circular mapping catheter. If PV isolation could not be established using this approach, it was completed using the conventional ablation approach. For analysis each vein was divided into quadrants from 12 o'clock looking from the Left Atrium (LA) into the vein. Manually identified fractionated potentials were compared with the NavX Complex Fractionated Electrogram (CFE) Mean map acquired in sinus rhythm and during coronary sinus pacing. CFE Mean Map parameters were set to a Width of 20 ms, Refractory of 30 ms, and P-P sensitivity of 0.05 mV. Manually tagged PV potentials were used as the gold standard. AF recurrences were assessed using ambulatory monitoring at 1, 3, 6 & 12 months post ablation.
Twenty consecutive patients with paroxysmal AF (age 59 ± 10 y, 60% male, LA 40 ± 6 mm) were enrolled in the study. Distribution of CFE closely paralleled activation. In addition, CFE were identified on the septum in sinus rhythm in 60% of the patients and on the posterior wall during CS pacing in 35% of the patients. The sensitivity-specificity of the auto-identified CFE that predicted manually tagged fractionated potentials were 60%-68% in Sinus Rhythm (SR), and 80%-70% during CS pacing. Mapping and ablation fluoroscopy time was 30 ± 12 min with Radio Frequency (RF) time of 27 ± 10 min. Completion of PVAI required an additional 22 ± 16 min of fluoroscopy and 31 ± 28 min of RF energy delivery. Total procedure time was 209 ± 36 min. Only 13 (4%) of all PV quadrants in n = 5 (25%) patients did not have to be ablated to achieve PVAI. Three patients (15%) had AF recurrences between the first 3 months and 12 months following ablation. Two of these patients cumulatively had 7 of the 13 non-ablated quadrants in the study.
While mapping earliest PV activation during sinus rhythm and CS stimulation is feasible, ablation guided by this approach may avoid unnecessary RF energy delivery in only a small proportion of the PV antral segments, potentially leading to higher ablation failure rates. NavX CFE mean algorithm accurately identifies areas of fractionation at PV antra during sinus rhythm and distal CS stimulation corresponding to PV potentials.
Atrial fibrillation, Catheter ablation, Pulmonary vein antrum isolation
ACT: Activated Clotting Time; AF: Atrial Fibrillation; AAD: Anti-Arrhythmic Drug; ICE: Intracardiac ECHO; LA: Left Atrium; PV: Pulmonary Vein; PVAI: Pulmonary Vein Antrum Isolation; QoL: Quality of Life; TEE: Transesophageal ECHO; OAC: Oral Anti Coagulation
Atrial Fibrillation (AF) is the most common cardiac arrhythmia [1]. It increases mortality risk, causes disability, reduces quality of life, and presents treatment challenges [2-4]. Sinus rhythm is typically preferred in clinical setting as opposed to AF, in order to control patients' symptoms.
Established evidence implicates triggered activity in the Pulmonary Veins (PV), in initiating and maintaining AF [5,6]. Ablations of such foci of ectopic electrical activity have prevented AF recurrences and in some cases terminated chronic AF, altogether [7-11].
Early ablation strategies included direct focal mapping and elimination of pulmonary vein triggers in a time consuming process that resulted in low efficacy and in significant risks for pulmonary vein stenosis. Main stream approaches to ablation in patients with AF include electroanatomically guided pulmonary vein ablation [9,11] and circumferential pulmonary vein (antrum) isolation using a circular mapping catheter [12], intracardiac echocardiography, and/or other non-fluoroscopic modalities as well as cryoballoon ablation. While these strategies achieve success in 60%-90% of patients, at low risk of hemorrhagic and embolic complications, and pulmonary vein stenosis following the procedure [12-14], most investigators feel that pulmonary vein isolation is an important endpoint of ablation that optimizes procedural success. Strategies aiming to isolate the pulmonary veins generally involve the need for double transseptal access in order to instrument the Left Atrium (LA) with a circular mapping catheter, as well as an ablation catheter. This may increase the risks associated with transseptal puncture and prolong fluoroscopy time during procedures. There has been no prospective testing of a strategy where electrical inputs to the pulmonary veins are mapped, with areas responsible for conduction into of the pulmonary vein antrum identified (on a 3D representation of the LA) and subsequently ablated using a single transseptal access.
NavX electroanatomical mapping system (St Jude Medical, Minneapolis, MN, USA), useful in the strategy described above, can be implemented to map conduction into the veins and help limit ablation lesions to solely the vein regions that propagate electrical impulses between the body of the left atrium and the PV antrum. It can also identify fractionated atrial electrograms using a CFE Mean Map software, which averages the timing intervals between multiple electrogram components over a user-defined time interval.
The objective of this feasibility pilot study was to test the strategy where electrical inputs to the PVs, defined by earliest PV activation in sinus rhythm and during distal Coronary Sinus (CS) stimulation, and/or by local fractionation, are selectively targeted to achieve PV antral isolation. A secondary objective was sensitivity-specificity analysis of the predictive ability of the CFE mean map to identify sites exhibiting local fractionation tagged by the operator, as well as fractionated areas identified using the NavX CFE algorithm.
Twenty consecutive patients with paroxysmal AF meeting criteria for ablation were prospectively enrolled into the study after signing consent. All patients completed 1-year follow-up. The study was approved by the institutional Ethics Review Board. The data was collected prospectively.
Patients received continuous oral anticoagulation with warfarin for at least three weeks prior to the experimental study. Warfarin was stopped 3 days prior to the procedure and the patients received low molecular weight heparin for bridging. They had a Transesophageal Echocardiogram (TEE) within 24 hours prior to the procedure to rule out left atrial thrombus or spontaneous echo-contrast.
All anti arrhythmic agents were discontinued at least 5 half-lives before the procedure or 6 months prior in the case of amiodarone.
During the procedure, a decapolar non-deflectable CS catheter was placed using the right internal jugular vein. An intracardiac echocardiography probe (ICE, Siemens AG Inc., Mountain View, CA, USA) was placed in the right atrium via the left femoral vein. Transseptal access to the left atrium was then established under fluoroscopic and ICE guidance. Patients received 5000 U of unfractionated heparin IV with each of two transseptal punctures. Heparin infusion at 1000 U/hour was initiated with the first puncture. Activated Clotting Time (ACT) was maintained in the range of 350-400 seconds.
Transseptal access was used to advance a decapolar circular mapping catheter (Lasso, Biosense Webster, Diamond Bar, CA, USA) and an irrigated tip ablation catheter (Celcius Thermocool, Biosense Webster, Diamond Bar, CA, USA) into the left atrium via braided SL-1 introducers (St Jude Medical, Minneapolis, MN, USA).
Once in the left atrium, 3D electroanatomic maps were constructed using NavX to display electrical activation in Sinus Rhythm (SR) and during distal CS stimulation. The NavX non fluoroscopic navigation system has been described elsewhere [15]. Briefly, the roving circular mapping catheter was placed 2-4 cm into each PV and slowly pulled back. Along pullback, multiple locations were recorded to tag the vein. The ostium was identified by fluoroscopic visualization of the catheter entering the cardiac silhouette with simultaneous impedance decrease and appearance of atrial potentials. Separate chamber geometries were collected for each pulmonary vein and for the left atrial appendage.
The NavX Local Activation Time map (LAT) was set up using a robust atrial reference signal from the proximal CS electrogram. The Roving Activation Interval (RAI) for the Complex Fractionated Electrogram (CFE) mean map was set to encompass only the atrial activation within the window of interest re: the reference signal, e.g. 100 ms to +200 ms. Fractionated potentials in the roving catheter electrograms acquired during sinus rhythm and coronary sinus pacing could then be quantified using the CFE Mean map. CFE Mean Map parameters were set to a Width of 20 ms, Refractory of 30 ms, and P-P sensitivity set just above baseline noise, at 0.05 mV. Using this approach, LAT mapping resulted in a simultaneous acquisition of activation and fractionation maps in sinus rhythm and during distal CS stimulation. Each vein was divided into Quadrants for later analysis, looking from the LA into the vein and starting at 12 o'clock for Quadrant 1 (Figure 1).
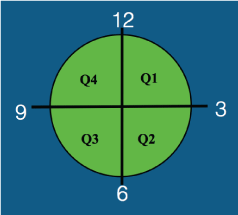 Figure 1: Representation of the pulmonary vein antrum for the analysis. Each vein was divided into quadrants clockwise from 12 o'clock, looking into the vein from the left atrium.
View Figure 1
Figure 1: Representation of the pulmonary vein antrum for the analysis. Each vein was divided into quadrants clockwise from 12 o'clock, looking into the vein from the left atrium.
View Figure 1
Segments of earliest wave front propagation into the pulmonary vein antra were labeled on both maps, for sinus rhythm and for distal CS stimulation (Figure 2) using manual propagation of the activation front through the left atrium. Each acquired point was then examined manually by the operator for local electrogram fractionation defined as 3 or more high frequency local deflections (Figure 3). Fractionated sites were tagged for later comparison with the NavX CFE Mean Map. CFE were mapped during sinus rhythm and CS pacing. Only areas responsible for PV activation were ablated. No ablation was carried out beyond what was necessary to achieve PV isolation.
 Figure 2: Wavefront propagation into pulmonary vein antra. Wavefront propagation was examined during manual propagation analysis of each map. Green lines represent early activating segments in sinus rhythm. Red lines represent early activating segments during distal CS stimulation. Green arrows illustrate the direction of wavefront propagation into the pulmonary veins. View Figure 2
Figure 2: Wavefront propagation into pulmonary vein antra. Wavefront propagation was examined during manual propagation analysis of each map. Green lines represent early activating segments in sinus rhythm. Red lines represent early activating segments during distal CS stimulation. Green arrows illustrate the direction of wavefront propagation into the pulmonary veins. View Figure 2
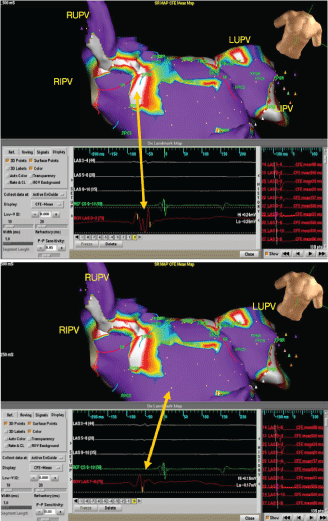 Figure 3: Correlation between manually tagged fractionated potentials, defined as 3 or more local deflections and the CFE mean map. Manually identified sites exhibiting local fractionation have been tagged and correspond to the white areas on the CFE mean map automatically identifying fractionation. A) Manually identified fractionated area during sinus rhythm corresponds to the CFE Mean map and B) Manually identified non-fractionated area during sinus rhythm corresponds to the CFE Mean map. View Figure 3
Figure 3: Correlation between manually tagged fractionated potentials, defined as 3 or more local deflections and the CFE mean map. Manually identified sites exhibiting local fractionation have been tagged and correspond to the white areas on the CFE mean map automatically identifying fractionation. A) Manually identified fractionated area during sinus rhythm corresponds to the CFE Mean map and B) Manually identified non-fractionated area during sinus rhythm corresponds to the CFE Mean map. View Figure 3
Ablation lesions were delivered using power of 35-40 Watt for ~20 sec per lesion with the endpoint of disappearance of all local potentials in the areas of the pulmonary vein antrum defined by early activation in sinus rhythm and during CS pacing, or by the presence of fractionated potentials (Figure 4). After the initial set of lesions had been delivered, pulmonary vein antrum isolation was verified using the circular mapping catheter and additional ablation lesions were delivered as necessary to achieve PVAI (entrance block). Exit block was not routinely verified.
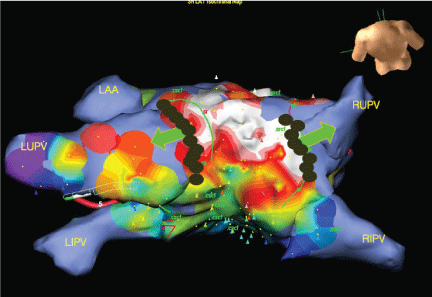 Figure 4: Part of the study lesion set targeting the segment of early wavefront propagation into the pulmonary veins in sinus rhythm. Green lines represent the early activating segments in sinus rhythm. Red lines represent early activating segments during distal CS stimulation. Green arrows illustrate the direction of wavefront propagation into the pulmonary veins. View Figure 4
Figure 4: Part of the study lesion set targeting the segment of early wavefront propagation into the pulmonary veins in sinus rhythm. Green lines represent the early activating segments in sinus rhythm. Red lines represent early activating segments during distal CS stimulation. Green arrows illustrate the direction of wavefront propagation into the pulmonary veins. View Figure 4
Sites tagged by the operator as exhibiting fractionation were then compared to the fractionated areas identified using the CFE Mean Map. Each patient received IV protamine upon withdrawal of the catheters into the right atrium to achieve ACT of < 200 for sheath removal.
Patients were discharged home the day after ablation on oral anticoagulation with warfarin, without a bridging agent. Antiarrhythmic medications in patients taking these prior to ablation were resumed and continued for the first two months following ablation.
A spiral CT of the chest was used to rule out PV stenosis 3 months following ablation.
Recurrence of AF was assessed by a 24-hour Holter monitor assessment at 1, 3, 6 & 12 months following ablation. Additional monitoring with a loop event recorder occurred in the presence of symptoms. Any episode of AF > 30 second's duration was defined as a recurrence. All data for this study was captured and analyzed prospectively from consecutive patients undergoing pulmonary vein isolation procedure at our institution.
Baseline characteristics of the patients in the study are illustrated in Table 1. These are representative of the population of patients with paroxysmal AF ablated at our center [16].
Table 1: Pre-procedural characteristics, Mean ± SD or n (%). View Table 1
Left pulmonary veins activated preferentially along the posterior wall in sinus rhythm (Quadrants 3 and 4) and the ridge with LAA (Quadrants 1 and 2) during distal CS stimulation (Figure 5).
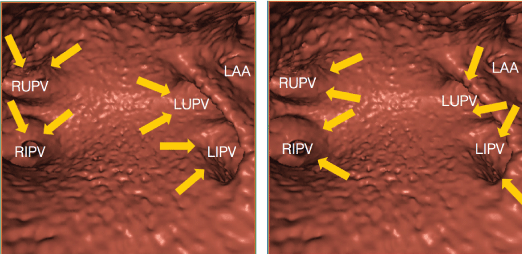 Figure 5: Pattern of activation of the pulmonary veins. Yellow arrows indicate direction of local activation at PV antra in sinus rhythm and during CS pacing. A) During Sinus rhythm and B) During Distal CS stimulation.
Figure 5: Pattern of activation of the pulmonary veins. Yellow arrows indicate direction of local activation at PV antra in sinus rhythm and during CS pacing. A) During Sinus rhythm and B) During Distal CS stimulation.
LUPV: Left Upper PV; LIPV: Left Inferior PV; RUPV: Right Upper PV; RIPV: Right Inferior PV; LAA: Left Atrial Appendage. View Figure 5
Right pulmonary veins were activated preferentially along the roof (Quadrants 4 and 1) during sinus rhythm and along the posterior wall during distal CS stimulation (Quadrants 1 and 2) for all patients in the study (Figure 5).
Distribution of fractionation closely paralleled that of activation. In addition, fractionated potentials were identified on the septum in sinus rhythm in 60% of the patients and on the posterior wall during distal CS stimulation in 35% of the patients.
Sensitivity-specificity of the CFE mean map predicting manually identified fractionated PV potentials were 60%-68% respectively in SR and 80%-70% during distal CS stimulation. These were 70%-70% when evaluating the ability of the CFE mean map to predict manually tagged fractionated potentials during sinus rhythm or coronary sinus pacing (Table 2).
Table 2: Sensitivity and Specificity of the CFE mean map to predict manually identified fractionation. View Table 2
Procedural parameters are shown in Table 3. Fluoroscopy and procedure times during study mapping and ablation were similar to those required to complete PVAI after the study protocol. Total procedure time was 209 ± 36 min. Total procedure and fluoroscopy times were in line with previously reported values.
Table 3: Procedural characteristics, Mean ± SD. View Table 3
Only 13 (4%) of all pulmonary vein Quadrants in 5 (25%) patients did not have to be ablated to achieve PVAI. Three patients (15%) had AF recurrences between the first 3 months and 12 months following ablation. Two of these patients cumulatively had 7 of the 13 non-ablated quadrants in the study.
This study explored a novel approach of sequential rather than concurrent mapping and isolation of the pulmonary vein antra guided by the NavX non-fluoroscopic 3D electroanatomical mapping system. Unfortunately, ablation could be avoided in only a small number of pulmonary vein antral segments. Patients who demonstrated antral isolation without the need for further ablation, had a higher likelihood of arrhythmia recurrence. In the remainder of the study group, completion of PVAI involved ablation of all other antral segments.
These findings support the perspective that complete pulmonary vein antral isolation is of paramount importance to AF ablation success. In most pulmonary veins, activation can proceed into the vein across any of the segments, depending on the source of the atrial rhythm. The areas showing earliest pulmonary vein antral activation frequently demonstrate local fractionated potentials, consistent with the "pulmonary vein potentials" typically targeted for ablation. While we don't have sufficient proof from this study that additional ablation required to complete the PVAI lesion set added to the success of the ablation, this may be hypothesized as most recurrences occurred in patients with evidence of pulmonary vein antral isolation, despite having segments of the antrum that were not ablated. These findings suggest that circumferential pulmonary vein antral isolation is superior to segmental ablation [13], contradicting some published evidence that indicates ablation of the antra can be limited to a few segments without sacrificing the clinical success of the procedure [17].
Similar potentials are also seen outside of the pulmonary veins at the sites of earliest left atrial activation, such as the septum during sinus rhythm, or the posterior wall during stimulation of the distal coronary sinus. The importance of these sites, as substrates for atrial fibrillation, has been previously demonstrated [18]. As observed in the results of this study the CFE mean map algorithm correlated very well with the human manual operator manual tagging fractionated signal corresponding to PV potentials which was used as the Gold Standard (Table 2). It is possible that the proprietary analytical system that presents "AF Nests" during sinus rhythm, that have been targeted for ablation [19], effectively identifies the same regions. In that case, such areas of interest could also be labeled with reasonable accuracy using the NavX CFE Mean algorithm. While there are a number of studies including STAR-AF II by Verma, et al. which demonstrated that ablation of CFEs mapped during AF have limited efficacy and in some cases, reduces the success of AF ablation, in this study we did not intend to map or ablate areas tagged as exhibiting CFE beyond PV segments ablated to ensure isolation. Fractionated activity corresponding to PV potentials was mapped in sinus rhythm and during CS pacing rather than in AF. We then applied the NavX CFE Mean Map to the sinus rhythm and CS paced left atrial maps to find reasonable correlation between manually annotated PV-potential fractionation and fractionation as detected by the algorithm. Additionally, it is possible that the lesions delivered during the study were not transmural and complete. If this were the case, PVAI might have been achieved without additional ablation had complete lesions been delivered to the areas identified. For clinical purposes however, our technology remains limited and lesions were delivered using well-established RF energy settings.
This was a prospective feasibility pilot study enrolling a small number of patients. Therefore there were a number of inevitable biases despite the small numbers we felt that further investigation would not be warranted based on our findings.
While mapping earliest PV activation during sinus rhythm and CS stimulation is feasible, ablation guided by this approach likely avoids unnecessary RF energy delivery in only a small proportion of PV antral segments. CFE Mean algorithm accurately identifies areas of fractionation at PV antra during sinus rhythm and distal CS stimulation. Full circumferential Pulmonary Vein Antrum ablation leading to isolation remains the preferred ablation strategy for AF refractory to medical therapy.
The Study was funded by a Research Grant from St Jude Medical. The Authors do not have any further individual disclosures with respect to this study.