Stress Echocardiography (Echo) and Myocardial Perfusion Scintigraphy (MPS) are effective methods for identification of myocardial ischemia. Aim of this study was to compare adenosine stress MPS and Echo results with gold standard angiography.
Twenty nine patients (18 F, 11 M; mean: 49, 62 ± 10, 9; 45, 5-53, 8-years-old) with suspicion of Coronary Artery Disease (CAD) were included into the study. After performance of basal Echo examination, adenosine infusion was started (140 µgr/kg/dk) and at the third minute after starting the infusion, intravenous Tc-99m MIBI injection was performed and stress Echo findings were recorded at the 10th minute. Stress and rest imaging were performed at the same day. Stress Echo was performed by an experienced cardiologist and MPS results were interpreted by an experienced nuclear medicine physician who didn't know stress Echo results according to 17 segment models. Sixteen patients underwent coronary angiography. Ischemia territory size in both the adenosine stress Echo and MPS were recorded and compared in segment and in patient basis.
There were 34 ischemic segments in MPS and 46 in stress Echo among 493 segments of 29 patients. Both MPS and stress Echo pointed the same ischemic segments in 9 patients and tests were in agreement in 24 patients. Angiography confirmed two tests results in 13/16 patients. Three patients with severe ischemia in the MPS and stress Echo had slow flow pattern in the angiography.
The results of both MPS and stress Echo with same adenosine infusion was in agreement especially in patients with angiography results. These findings can encourage simultaneous studies with same stress agent infusion.
Adenosine, Myocardial perfusion scintigraphy, Stress echocardiography, Ischemia, Tc-99m MIBI
Coronary Artery Disease (CAD) is the most common cause of death all over the world. The most important clinical presentation of this disease is angina pectoris. However there are numerous possible differential diagnoses in the patients with angina pectoris. The identification of patients with myocardial ischemia is an important part of the diagnosis of CAD. Exercise Electrocardiography (ECG) is the first line method for the investigation of the patients with suspicion of CAD despite the low sensitivity of this method. Therefore stress induced ischemia should be demonstrated by various methods like radionuclide imaging. Myocardial Perfusion Scintigraphy (MPS) is an accurate modality which shows the ischemic regions in the myocardial tissue. Other imaging options in the assessment of the myocardial ischemia are stress Echocardiography (Echo) and other morphological imaging methods like Computed Tomography (CT) or Magnetic Resonance Imaging (MRI). Despite all these advances, MPS is still the most important modality in showing the ischemia regarding experience in this field. MPS is considered as a reference method in the recent studies in which different stress Echo applications are introduced [1-3]. As far as we know this is the first comparative study performed with the same stress agent infusion and compared MPS with stress Echo with gold standard angiography. Aim of this study is to compare MPS and stress Echo findings with same Adenosine infusion with gold standard angiography.
Twenty nine patients (18 F, 11 M; mean: 49, 62 ± 10, 9; 45, 5-53, 8-years-old) with suspicion of coronary artery disease (CAD) were included into the study. The patients were referred to Firat University Hospital Nuclear Medicine Department from Cardiology Department for adenosine stress MPS. Exclusion criteria were pregnancy, lactation, < 18 y and all the contraindications of adenosine infusion which include malignant ventricular arrhythmias, sinoatrial and atrioventricular blocks without pacemaker, severe valvular disease, cardiomyoapthy, asthma and severe hypertension systolic > 200 mmHg, diastolic > 120 mmHg [4]. Patients were asked to stop some of their medications (β-blockers, Ca antagonists) one day prior to the examination.
The study was approved by Local Ethic Committee and informed consents of all the patients were obtained. The study was conducted according to the Helsinki Declaration in a prospective manner.
After performance of basal Echo examination, adenosine infusion was started (140 µgr/kg/dk) according to the guidelines [5]. After unset of adenosine infusion at third minute after starting the infusion, intravenous Tc-99m MIBI injection was performed and stress Echo findings were recorded at the 10th minute. Adenosine infusion was stopped in these cases with severe hypotension, sinoatrial or third degree atrioventircular block, severe dispnea or flushing.
Transthoracic stress echocardiography studies were performed with commercially available ultrasound machines (Vivid System 7, GE/Vingmed, Milwaukee, Wisconsin) equipped with multifrequency phased-array sector scan probe (S3-S8 or V3-V7) and with Second Harmonic technology. Echocardiographic images were semiquantitatively assessed using a 17-segment, 4-point scale model of the left ventricle [6]. A wall motion score index was derived by dividing the sum of individual segment scores by the number of interpretable segments. Ischemia was defined as stress-induced new and/or worsening of pre-existing wall motion abnormality. Rest wall motion abnormality was akinetic, hypokinetic (alteration of the wall motion or thickening compared to rest) or dyskinetic myocardium with no thickening during stress. A test was normal in case of no rest and stress wall motion abnormality. The interpreter of the stress echo was subjected to radiation exposure with knowledge of the dose. The interpreter additionally performed the angiography thus he was a radiation worker and carried a dosimeter during the procedures.
Stress and rest imaging were performed at same day. Firstly rest and after a waiting period stress imaging were performed. For both the rest and stress imaging after a waiting interval of thirty minutes from injection of radiopharmaceutical Single Photon Emission Tomography (SPECT) imaging was performed by a double head gamma camera equipped with low energy all purpose collimator (GE, Infina). The images were acquired over a 180° arc in 64 projections each with a time of 30 seconds in 64 × 64 matrix. The butterworth filter with cutoff frequency 0.5 and order 10 was implicated for reconstruction of the images. Short axe, horizontal and vertical long axe slices were obtained and interpreted by an experienced nuclear medicine physician who didn't know the stress Echo results. Additionally the images were evaluated according to the 17 segment cardiac charts by the same nuclear medicine physician and improvement of hypoperfusion in a segment in the rest imaging is considered as ischemia and fix defect as infarct.
Angiography was performed by standard Judkins technique by the same cardiologist that performed the stress echocardiography in a blind manner. Coronary artery disease diagnosis was based on the narrowing of any main artery > 50% in the diameter. Additionally TIMI frame count was calculated which presents the time frame count between the first frame that the entrance of the contrast agent to left ascending artery happens and last frame that the contrast agent reaches the end of the artery [7]. Slow flow pattern was decided regarding the TIMI (> 21 frame).
The results of the two imaging modalities (stress Echo and MPS) were compared in an ischemia positive and negative manner in segment basis and in patient basis.
Patient characteristics are summarized in the following table (Table 1).
Table 1: Patient characteristics. View Table 1
There were 34 ischemic segments in MPS and 46 in stress Echo among 493 segments of 29 patients. Both MPS and stress Echo pointed the same ischemic segments in 9 patients and tests were in agreement in 24 patients. However, only sixteen patients had angiography results (Table 2). The imaging results of the patients with angiography results were as follows; 6 patients with anterior, 6 patients with inferior, one patient with septum defect and three patient with normal perfusion were present also echo showed same regions hipokinesia in 9 patients and normal results in three scintigraphically normal patients. Angiography confirmed two test's results in 13/16 patients (Figure 1 represents a patient with similar results in both tests and confirmation result with angiography). Three patients with severe ischemia on MPS and stress Echo had slow flow pattern on angiography. The sensitivity, specificity, accuracy, negative, positive predictive values of the methods were 100%, 50%, 81%, 77%, and 100% for MPS and 90%, 60%, 81%, 77% and 90% for Echo respectively and are summarized in (Table 3) which shows very similar results for both tests. If we discard the patients with slow flow pattern results in angiography the sensitivity, specificity, accuracy, positive and negative predictive value of the MPS and Echo would be 67%, 43%, 69%, 60%, and 100% versus 100%, 71%, 85%, 75% and 100% respectively.
 Figure 1a: Short axe, vertical and horizontal long axe and bull's eye images of MPS of a patient showing inferolateral wall ischemia whose stress Eco revealed inferior wall ischemia. View Figure 1a
Figure 1a: Short axe, vertical and horizontal long axe and bull's eye images of MPS of a patient showing inferolateral wall ischemia whose stress Eco revealed inferior wall ischemia. View Figure 1a
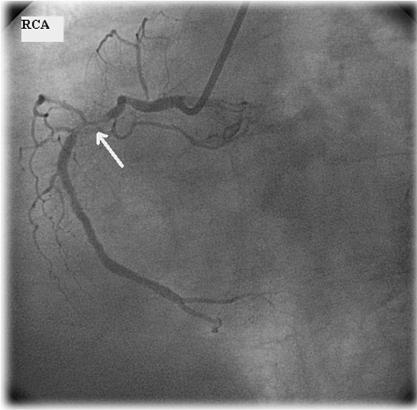 Figure 1b: Coronary angiography of the same patient also demonstrated the 70% narrowing of right coronary artery (arrow). View Figure 1b
Figure 1b: Coronary angiography of the same patient also demonstrated the 70% narrowing of right coronary artery (arrow). View Figure 1b
Table 2: Myocardial perfusion scintigraphy and echocardiography results of the patients with angiography results. View Table 2
Table 3: Diagnostic accuracy of both methods. View Table 3
According to our results MPS and stress Echo modalities with same infusion give comparable results and their results were generally in agreement with the angiography. The specificity and diagnostic accuracy of both methods were low because both MPS and Stress Echo procedures are ischemia imaging protocols and angiography as a gold standard causes increase in false positive results. However the positive ischemia finding with normal angiography shouldn't be considered as a real false positive result. Since all the false positive results are common in both tests, this would confirm that this is not a true false positive result. The diagnosis of ischemia in fact cannot depend on a morphological imaging method like coronary angiography since there are patients with non critical lesions who experience cardiac events, additionally not necessarily all patients that show ischemic changes in the functional imaging methods who have coronary lesions. In fact the most important diagnostic method in the identification of the coronary ischemia is the PET imaging with myocardial agents [8]. However, the availability of PET imaging and myocardial agents are restricted to the developed countries thus we were unable to compare our results with a real gold standard for ischemia. Angiography was an available and reasonable gold standard for this study.
The sensitivity of adenosine echo have been found significantly less than adenosine SPECT and dobutamine echo in a previous study in which gold standard method was the coronary angiography [4]. However, in our study the specificity of both methods was the main problem sensitivities were similar for both tests and in an acceptable range. Another study has included head to head comparison of adenosine and dobutamine stress echo and have found similar diagnostic accuracy for both methods [9]. A comparative study has indicated that both stress contrast myocardial echo and magnetic resonance imaging may differentiate ischemic and non-ischemic cardiomyopathy in a non-invasive manner [10].
The need for myocardial perfusion imaging depends on the fact that myocardial perfusion anomalies are present before the other diagnostic methods demonstrate any change (ECG) [11]. Thus myocardial perfusion anomalies can be considered as the first presentation of CAD in imaging modalities. Exercise ECG is the first line method for identification of the ischemia during stress but has low sensitivity to detect subtle changes since there are many indefinite results in this method. MPS has been considered as the cornerstone of ischemia imaging for many years and is an accurate method for demonstration of CAD [12].
There are many technical advances in the field of myocardial perfusion imaging recently. Although CT angiography has been considered as a morphologic imaging method, recently myocardial imaging by means of stress induced CT is also possible. A recent comparative study with adenosine-induced stress CT has showed good diagnostic performance compared to cardiac MRI and conventional coronary angiography [13]. Additionally adenosine-stress dynamic real-time myocardial perfusion CT and adenosine stress first pass dual energy myocardial CT has been compared in another recent study and both technique revealed comparable results to MR and SPECT with similar radiation exposure [14]. Combination of adenosine stress induced CT and MPS for detection of flow limiting coronary stenosis provided excellent diagnostic accuracy in the field of ischemia imaging [15].
Cardiac MRI is a new way of imaging of the myocardial perfusion and the ischemia. However in a recent study about interobserver variability of stress induced first-pass MR has showed only moderate agreement which has been influenced by experience and systematic reading criteria [16]. Reversible adenosine induced perfusion defects obtained from cardiac MR has found to be an independent predictor of cardiac events according to approximately four years follow up of 203 patients in a previous study [17]. Additionally parameters obtained from cardiac MRI like adenosine stress perfusion, delayed enhancement, and left ventricular function has been observed to provide information regarding prognostification in another study [18].
Stress agents are important part of stress studies in all the imaging methods. Pharmacological stress is considered as an alternative to exercise stress testing in patients without exercise capacity or unable to exercise. A recent comparative study with adenosine and high dose dobutamine/atropine stress MR perfusion analysis has indicated high level of concordance in both agents [19]. One of the most important drawbacks of pharmacologic stress agents is the adverse effects of these agents. There are numerous safety analysis studies as in one of them 16% of 2150 patients have faced with adverse drug reactions thus adenosine stress have been considered as safe and useful method for myocardial perfusion scintigraphy [20]. Adenosine stress SPECT studies have found to be safe in even cardiac transplant patients in a recent report except for higher incidence of atrioventricular blocs compared to general population [21]. Monzen, et al. have performed adenosine application with additional low dose exercise, in order to decrease side effects and improve image quality as a result of decreasing liver uptake. In that study the image quality was improved however, there was not a considerable change in the frequency of the side effects which was not already high [22]. In our study group we didn't observe side effects although we expected them.
Regadenoson is a new stress agent which is introduced as a simpler way of pharmacological stress. Comparative analysis of regadenoson and adenosine revealed excellent agreement between the two agents [23]. Additionally regadenoson applications include a simple injection which is well tolerated by patients with lower cost and do not require dose calculation according to the body weight or renal impairment [24]. Since both adenosine and dypiridamole infusions are time consuming and they both require monitorization, a simpler way of pharmacologic stress application is promising.
In a comparative study with adenosine stress myocardial contrast echocardiography the sensitivity, specificity, and accuracy of this modality compared to X-ray angiography and cardiac MR were 85%, 76% and 82% versus 85%, 74% and 79% respectively [25]. Although it has been documented that interobserver agreement is better in the contrast enhanced echocardiography however we employed a non-contrast protocol in our study [26].
Simultaneous MPS and stress Echo findings with same adenosine infusion were in agreement with each other and also the angiography results. These results can encourage simultaneous pharmacologic stress studies with same injection or infusion in the future.
No conflict of interest, funding, research contract exists. This manuscripts abstract is accepted as an oral presentation in 1st Balcan Congres of Nuclear Medicine which will be in 4-8th April 2012 in Antalya/TURKEY.