International Journal of Respiratory and Pulmonary Medicine
Halotherapy in Patients with Cystic Fibrosis: A Pilot Study
Morhaf Al Achkar1*, David E. Geller2, Amanshe Perera Slaney3 and Daniel T Layish3,4
1Indiana University School of Medicine, Indianapolis, USA
2Florida State University College of Medicine, Orlando, FL, USA
3Central Florida Pulmonary Group, USA
4University of Central Florida School of Medicine, Orlando, FL, USA
*Corresponding author: Morhaf Al Achkar, Indiana University School of Medicine, Indianapolis, IN 46205, USA, E-mail: alachkar@iupui.edu
Int J Respir Pulm Med, IJRPM-2-009, (Volume 2, Issue 1), Research Article; ISSN: 2378-3516
Received: November 28, 2014 | Accepted: December 30, 2014 | Published: January 04, 2015
Citation: Achkar MA, Geller DE, Slanely AP, Layish DT (2015) Halotherapy in Patients with Cystic Fibrosis: A Pilot Study. Int J Respir Pulm Med 2:009. 10.23937/2378-3516/1410009
Copyright: © 2015 Achkar MA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: Cystic fibrosis (CF) is a complex genetic disorder involving the lower and upper respiratory tract. The purpose of this study is to evaluate the effect of Halotherapy on sinusitis symptoms, dyspnea, pulmonary function tests, and quality of life in CF patients.
Study design: This was a pilot open-label before-and-after-study.
Settings: The study was performed at the Salt Room® Orlando Participants were from a single CF care center, and were enrolled in the study between January and June, 2012.
Subjects and methods: Patients with clinically stable CF were included in the study. Participants received 9 sessions of HT, 45 minutes each, completed over a 3-week period. Study endpoints include: FEV1 and FVC, Borg dyspnea index test, Cystic Fibrosis Questionnaire-Revised (CFQ-R), and the Sino-Nasal Outcome Test (SNOT-20).
Results: Twelve patients completed the study protocol. FEV1 and FVC did not change significantly (p= 0.49 and 0.87, respectively). SNOT-20 score improved by 0.62 points (95% CI -1.03 to -0.2, P = 0.007). There was a trend for improvement in Borg Dyspnea index; the mean score decreased by 0.79 (95% CI -1.64 to 0.05, P=0.065). There was significant improvement in the physical domain, the health perception domain, and the digestive domain.
Conclusion: HT is associated with improvement in symptoms of sinus disease in CF, and should be explored as an adjunct treatment for CF patients.
Introduction
Cystic fibrosis (CF) is a complex genetic disorder characterized by dehydration of the respiratory epithelial surface resulting in impaired mucociliary clearance [1,2]. Thick, sticky secretions obstruct the lower airways and sinuses, providing an environment for chronic infection. A significant proportion of CF patients experience sinus symptoms and almost all have radiographic findings of chronic sinusitis. Increasing the volume of airway surface liquid improves mucus clearance in patients with CF [2,3]. Inhaled hypertonic saline is one method used in patients older than 6 years to rehydrate the airways [3]. In clinical trialshypertonic saline inhalation improved pulmonary function [2,4], and respiratory symptoms, reduced pulmonary exacerbations [4,5] and reduced absenteeism from school or work [5]. Halotherarpy (HT; �halos� means salt in Greek) aims to deliver salt particles into the upper and lower airways, and appears to be a promising alternative method.
For centuries, especially in Eastern Europe, people have visited natural salt caves for the healing properties of the air. Halotherapy (HT) simulates conditions in a natural salt cave by dispersing salt particles in a controlled air medium. While similar in principles to hypertonic saline, HT differs in that it delivers dry aerosol microparticles (1-5 �m) of salt rather than a wet solution [6]. Typically, a person visits a facility that provides HT services for a period of 30-60 minutes, where they read or perform relaxing activities while undergoing halotherapy. Breathing through the nose and mouth allows treatment effect to target the upper and lower airways.
While considered spa treatment, Halotherapy�s effectiveness was evaluated in multiple clinical trials. HT was studied in 139 patients with respiratory diseases, among whom 5 had CF. Improvements in flow-volume loop parameters and decreased bronchial resistance measured by plethysmography were reported after 10-20 sessions. The CF patients were reported to have similar response with the treatment [7]. Another recent study showed an increase in lung function and sputum production in 6 CF subjects after only 5 halotherapy sessions [7].
As a pilot work to assess feasibility, evaluate effectiveness, and gather clinical data to better estimate sample size for an experimental study, we used a pre- and post- test study design to assess the effect of HT on the pulmonary and sinus symptoms, dyspnea, and quality of life in CF patients.
Methods
This open-label study was performed at the Salt Room� Orlando, which provided the facilities for HT. Patients from a single CF care center (Central Florida Pulmonary Group, Orlando, FL, USA) were enrolled in the study between January and June of 2012. Study endpoints were measured twice: before the first and after the last HT session. The ethics committee at Quorum Review Board approved the study. Each participant provided written informed consent or assent. The trial was designed and executed by the academic investigators. The Salt Room� Orlando provided the HT sessions and information on participantsattendance but otherwise did not participate in the design and conduct of the study, in the analysis and interpretation of the data, or in the writing or review of the manuscript.
The inclusions criteria were the following: History of CF and the following, age 15 years and older, clinically stable on their medical regimen for at least a month prior to enrollment, forced vital capacity (FVC)>40% of predicted value, forced expiratory volume in one second (FEV1) between 30% and 85% of predicted value, and a score of 10 or above on the rhinologic domain of the Sino-Nasal-Outcome Test-20 (SNOT 20), which is a validated patient-outcome reported measure with four sub-domain: psychological function, rhinological symptoms, sleep function, and ear and/or facial symptoms [8]. Participants who had received antibiotics or corticosteroids for the treatment of a pulmonary exacerbation within 30 days, had taken hypertonic saline within two weeks, or had used HT previously were all excluded from the study. Patients were screened for eligibility in the clinic, prior to enrollment. Eligible participants, based on the available clinical information, returned for a baseline visit within one week of screening. At the baseline visit patients provided written consent, had a medical history and physical exam, performed spirometry, and answered questionnaires, including the Borg dyspnea index test, Cystic Fibrosis Questionnaire-Revised (CFQ-R), and the Sino-Nasal Outcome Test (SNOT-20).
Results
Between January and June of 2012, twelve participants completed the study protocol, including all 9 sessions of HT. Two additional patients met the inclusion criteria but they developed respiratory exacerbations before starting the treatment and were excluded from the analysis. Every participant completed medical history and physical exam, spirometry test, and sets of questionnaires (Borg, CFQ-R, SNOT-20). During the course of the study the participants continued on their standard regimen of treatment for CF.
![]()
Table 1: Baseline Characteristics of the 12 Patients
View Table 1
![]()
Table 2: Effect of Halotherapy on Lung Function, SNOT, and Borg Score
View Table 2
The baseline characteristics of the participants are shown in Table 1. The average duration of follow up was 3 weeks. FEV1 and FVC did not change significantly (p value of 0.49 and 0.87, respectively). SNOT-20 Table 2 score (Figure 1) improved by 0.62 points (95% CI -1.03 to -0.2, P=0.007). There was a trend for improvement in Borg Dyspnea index (Figure 2); the mean score decreased by 0.79 (95% CI -1.64 to 0.05, P=0.065).
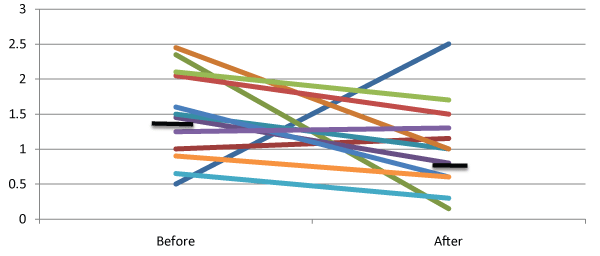
Figure 1: Individual patient SNOT 20 scores before and after HT
View Figure 1
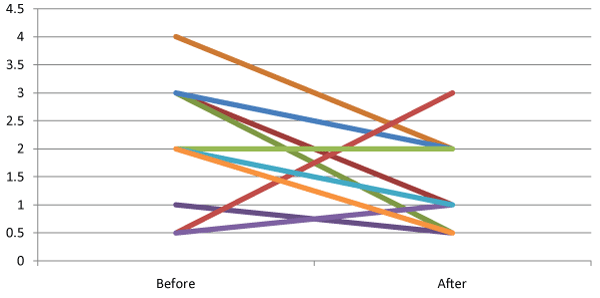
Figure 2: Individual patient Borg scores before and after HT
View Figure 2
Among the CFQ-R Table 3 domains there was significant improvement in the physical domain, the health perception domain, and the digestive domain. The respiratory domain improved by an average of 9.25 points, and while this is well above the recognized clinically important difference of 4 points, the change did not reach significance (p=0.124). All the other domains showed no significant change (Figure 3). There were no reports of chest tightness or wheezing as a direct result of the HT.
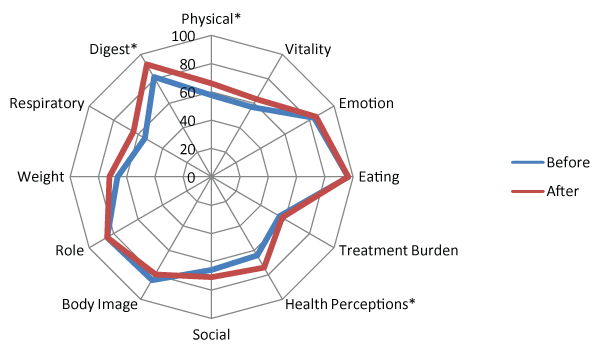
Figure 3:Figure 3: CFQR domains
The score on the CFQ-R domains ranges from 0, the worst score (poor
health), to 100, the best score (good health). Asterisks indicate P< 0.05 for the
comparison between before- and after- treatment.
View Figure 3
![]()
Table 3: Effect of Halotherapy on CFQ-R**
View Table 3
Discussion
This pilot study of HT is the first to include assessment of pulmonary function, dyspnea scores, sinus symptoms, and quality of life exclusively in patients with CF. Hypertonic saline has long been used successfully in CF, and HT is thought to work similarly in clearing the thick mucus secretions. A potential advantage of HT is the osmotic effects of salt particles both in the nose and sinuses as well as the lower airways. Hypertonic saline inhalation functions mainly in the lower airways, and may also be used via lavage or nebulization to the sinuses, but this is a separate procedure. Hypertonic saline can provoke bronchospasm in susceptible individuals; none of the participants in our study reported such symptoms.
The symptomatic improvement in the sinus symptoms may relate to the ability of the salt particles from HT to reach a target in the sinuses and stimulate mucus clearance. In this study, we enrolled only participants with significant baseline sinus symptoms (score >10 on the SNOT-20 rhinosinusitis domain). HT may not have similar effects on patients with absent or minimal sinus symptoms.
The improvement in some of the CFQ-R domains must be interpreted with caution as the study population was relatively small. The improvement in physical activity score may infer improved exercise capacity, which helps preserve pulmonary function [10]. The improvement was also significant in the health perception and digestive domains. One could speculate that the digestive domain may be influenced by better sinus function by improving olfactory sensation, and therefore appetite.
Our study had some limitations. Since this was an open-label study, we could not evaluate whether the improvement in the reported symptoms were due to subjective effects or objective physical benefits. While there were clear trends toward improvement in dyspnea perception, our pilot study was not powered enough to detect the change. The improvement in sinus symptoms was based on patient report, but was not validated by objective measures like sinus imaging. While the study was too short to evaluate outcomes like reduction in antibiotics or surgery, our initial findings suggest that a more detailed and longer-term study may be worthwhile to evaluate those important outcomes. Longer-term studies are also necessary to evaluate the effect of HT on pulmonary exacerbations and lung function. Examining the quantity or rheologic characteristics of sputum might also be considered in further studies, as well as the effects of HT on bacterial colonization.
In conclusion, this exploratory study has demonstrated that HT may have some benefit in CF patients with symptomatic sinus disease. Longer studies, using particularly a randomized controlled study design, are necessary to better evaluate the effects of HT on other outcomes and on patients with asymptomatic sinus disease.
Acknowledgment
We thank the Salt Room� Orlando for the treatment sessions. We also thank the Central Florida Pulmonary Group for performing the pulmonary function tests. The authors thank Global Clinical Research Management for their assistance with data organization.
Conflict of interest
Daniel Layish MD sits on the Board of Directors of the Salt Therapy Association and serves as Medical Advisor for the Salt Room� Orlando. Dr. Geller is currently employed by AbbVie, Inc in North Chicago, IL.
References
-
Flume PA, Robinson KA, O'Sullivan BP, Finder JD, Vender RL, et al. (2009) Cystic fibrosis pulmonary guidelines: airway clearance therapies. Respir Care 54: 522-537.
-
Donaldson SH, Bennett WD, Zeman KL, Knowles MR, Tarran R, et al. (2006) Mucus clearance and lung function in cystic fibrosis with hypertonic saline. N Engl J Med 354: 241-250.
-
Ong T, Ramsey BW (2013) Modifying disease in cystic fibrosis: current and future therapies on the horizon. CurrOpinPulm Med 19: 645-651.
-
Elkins MR, Robinson M, Rose BR, Harbour C, Moriarty CP, et al. (2006) A controlled trial of long-term inhaled hypertonic saline in patients with cystic fibrosis. N Engl J Med 354: 229-240.
-
Elkins MR, Robinson M, Rose BR, Harbour C, Moriarty CP, et al. (2006) A controlled trial of long-term inhaled hypertonic saline in patients with cystic fibrosis. N Engl J Med 354: 229-240.
-
Chervinskaya AV, Zilber NA (1995) Halotherapy for treatment of respiratory diseases. J Aerosol Med 8: 221-232.
-
Graepler-Mainka U (2011) Dry powder inhalation with NaCl for increasing secretolysis in cystic fibrosis patients-a pilot study. Preneted at the European Cystic Fibrosis Conference, Hamburg, Germany.
-
Pynnonen MA, Kim HM, Terrell JE (2009) Validation of the Sino-Nasal Outcome Test 20 (SNOT-20) domains in nonsurgical patients. Am J Rhinol Allergy 23: 40-45.
-
Chaaban MR, Kejner A, Rowe SM, Woodworth BA (2013) Cystic fibrosis chronic rhinosinusitis: a comprehensive review. Am J Rhinol Allergy 27: 387-395.
-
Hulzebos E, Dadema T, Takken T (2013) Measurement of physical activity in patients with cystic fibrosis: a systematic review. Expert Rev Respir Med 7: 647-653.





