The LRINEC (laboratory risk indicators for necrotizing fasciitis) score was developed in 2004 to help distinguish necrotizing fasciitis from severe soft tissue infections. Some of the laboratory tests, however, may be elevated at baseline in diabetic patients.
To evaluate the use of the LRINEC score in diabetic patients.
A retrospective study was performed on all patients admitted with either a primary diagnosis of cellulitis or necrotizing fasciitis between April 2010 and June 2014. Patients were excluded if they did not have the appropriate presenting lab values to calculate a LRINEC score. Presenting lab values were used to calculate each patient's LRINEC score and diabetic status was evaluated using International Classification of Diseases (ICD)-9 coding. All cases of necrotizing fasciitis were confirmed surgically.
A total of 670 patients met our inclusion criteria with 415 non-diabetic patients and 255 diabetic patients. Twenty-four of the non-diabetic patients (6.1%) and 11 of the diabetic patients (4.3%) had surgically confirmed necrotizing fasciitis. The average presenting LRINEC score in non-diabetic patients with and without necrotizing fasciitis was 6.9 ± 2.4 and 2.5 ± 2.4, respectively. The average presenting LRINEC score in diabetic patients with and without necrotizing fasciitis was 9.8 ± 2.1 and 4.1 ± 2.9, respectively. These scores were significantly higher (p < 0.01) than their non-diabetic counterparts. The sensitivity and specificity of a LRINEC score of 6 in non-diabetic patients was 0.79 (95% CI 0.57-0.99) and 0.86 (95% CI 0.82-0.89), respectively. In diabetic patients, the sensitivity and specificity were 0.91 (95% CI 0.57-0.99) and 0.72 (95% CI 0.67-0.78).
The LRINEC score is significantly higher in diabetic patients when compared with non-diabetic patients. Using a cutoff score of 6 produces a test with a poor specificity in diabetic patients.
Necrotizing fasciitis is a serious, life threatening soft tissue infection that can spread rapidly along fascial planes. This rapid spread often leads to hemodynamic instability, systemic sepsis and can eventually lead to multi-organ failure and death. Given the severity of this infection, early diagnosis and treatment, including surgical debridement, are vital. Diagnosis of necrotizing fasciitis is a clinical diagnosis, and given the consequences in delayed treatment, it should be managed with a high index of suspicion. Unfortunately, the common presenting features of swelling, pain and erythema [1] are non-specific and early necrotizing fasciitis can easily be mistaken for cellulitis [2], which is largely treated non-operatively. There are "hard signs" of necrotizing fasciitis that are more specific including pain out of proportion, rapidly spreading infection, bullae, skin ecchymosis/sloughing, gas in tissue, skin anesthesia, edema beyond erythema and sepsis, but these are only present in 43% of cases [3].
Various adjunct tests have been described to help with this challenging clinical problem including advanced imaging, the finger test, tissue oxygen monitoring, and early histological analysis [4,5]. Another diagnostic aid is the laboratory risk indicators for necrotizing fasciitis (LRINEC score). This test was introduced by Wong, et al. in 2004 as a diagnostic aid using common laboratory test to distinguish necrotizing fasciitis from other severe soft tissue infections [6]. The LRINEC score is a weighted score from 0-13, using C-reactive protein, glucose, sodium, white blood cell count, hemoglobin and creatinine. Table 1 shows the variables and weighted scoring system used to calculate the LRINEC score. As originally described, a score of at least 6 had a positive predictive value of 92% and a negative predictive value of 95% [6]. Separate validation studies have subsequently shown a wide variation in sensitivity (56-100%) and specificity (60-94%), as well as positive predictive (25-57%) and negative predictive value (85-100%) [7-9].
Table 1: Laboratory risk indicators for necrotizing fasciitis scoring system. View Table 1
Immunocompromised hosts are more susceptible to necrotizing fasciitis [9,10] and it has been well documented that diabetes mellitus is a very common comorbidity in patients with necrotizing fasciitis, with rates ranging from 29.7-59% depending on the study [1,7,10-19]. Diabetes is a disease that affects many different organ systems and can cause a wide variety of clinical pathology. This clinical pathology can affect laboratory values, including those used to calculate the LRINEC score [20]. Diabetes is defined by high blood sugar. Diabetic nephropathy can lead to chronic kidney disease, which is associated with elevated creatinine, anemia, and hyponatremia. Because of this interaction, we hypothesized that the LRINEC score would be elevated in patients with diabetes mellitus at baseline, and that using the same cutoff values would lead to a less specific test. The purpose of this study is to evaluate the LRINEC score in diabetic patients.
We retrospectively studied all hospital admissions to the University of California, San Diego medical centers in Hillcrest and La Jolla with a primary admission diagnosis of either cellulitis or necrotizing fasciitis from April 2010 to June 2014. International Classification of Diseases (ICD)-9 codes were used to search our medical record database for cellulitis as a primary admission diagnosis (681.00, 681.10, 681.9, 682.0, 682.1, 682.2, 682.3, 682.4, 682.5, 682.6, 682.7, 682.8, 682.9) and for necrotizing fasciitis (728.86). We evaluated these hospital admissions for age, gender, a diagnosis of diabetes, and the laboratory values used to calculate a LRINEC score. Admissions were excluded if they did not have appropriate laboratory values to calculate a LRINEC score within 24 hours of admission. If multiple laboratory values were available, the earliest value was used to calculate the LRINEC score.
All patients were then separated into two cohorts based on having an ICD-9 coded diagnosis of diabetes mellitus (249.xx, 250.xx). Sensitivities and specificities were then calculated using LRINEC cutoff scores from one to thirteen. All patients with necrotizing fasciitis had their diagnosis confirmed at surgery, with a review of the operative report to include the characteristic dishwater fluid or fascial abnormalities.
We used a t-test with an alpha value of 0.05 to determine significance. All results are reported as the mean ± standard deviation. Receiver operating characteristic curves were also generated to evaluate test sensitivity and specificity at different LRINEC score cutoffs reported with a 95% confidence interval (CI).
A total of 1,381 unique admissions were analyzed. Of these, only 670 (48.5%) had the required laboratory values to calculate an admission LRINEC score. We had 415 non-diabetic patients, 24 of which had a diagnosis of necrotizing fasciitis. Of the 255 diabetic patients, 11 had necrotizing fasciitis. A comparison between the patients admitted for cellulitis and necrotizing fasciitis is presented in Table 2.
Table 2: Comparison between diabetic and non-diabetic patients with cellulitis and necrotizing fasciitis. View Table 2
Average LRINEC scores are shown in Figure 1. The average LRINEC score for the non-diabetic patients with cellulitis and with necrotizing fasciitis was 2.5 ± 2.4 and 6.9 ± 2.4, respectively (P < 0.01). The average LRINEC score for diabetic patients with cellulitis and with necrotizing fasciitis was 4.1 ± 2.9 and 9.8 ± 2.1 (P < 0.01). When comparing non-diabetic and diabetics groups, the average LRINEC score was significantly higher in the diabetic group for both patients with cellulitis (P < 0.01) and necrotizing fasciitis (P < 0.01).
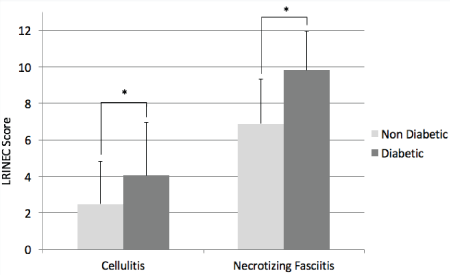 Figure 1: Average laboratory risk indicator for necrotizing fasciitis (LRINEC) scores in diabetic and non-diabetic patients with cellulitis and necrotizing fasciitis.
Figure 1: Average laboratory risk indicator for necrotizing fasciitis (LRINEC) scores in diabetic and non-diabetic patients with cellulitis and necrotizing fasciitis.
*denotes p < 0.01. View Figure 1
Receiver operating characteristic (ROC) curves were generated for both non-diabetic and diabetic patients (Figure 2). The area under the curve (AUC) for the non-diabetic diabetic patients was 0.886 (95% CI 0.851-0.951) and 0.928 (95% CI 0.889-0.956), respectively. The sensitivity and specificity of a LRINEC cutoff of 6 in non-diabetic patients was 0.79 (95% CI 0.57-0.92) and 0.86 (95% CI 0.82-0.89), respectively, which corresponded to a positive predictive value of 0.26 (95% CI 0.17-0.38) and a negative predictive value of 0.98 (95% CI 0.96-0.99). In diabetic patients, this same cutoff yielded a sensitivity and specificity of 0.91 (95% CI 0.57-0.99) and 0.73 (95% CI 0.67-0.78), respectively. The corresponding positive and negative predictive values for this were 0.13 (95% CI 0.07-0.23) and 0.99 (95% CI 0.96-1.00), respectively. The optimal LRINEC cutoff value for non-diabetic patients was 6, with the sensitivity and specificity described above. The optimal cutoff for diabetic patients was 9, which yielded a sensitivity of 0.91 (95% CI 0.57-1.00) and specificity of 0.92 (95% CI 0.87-0.95).
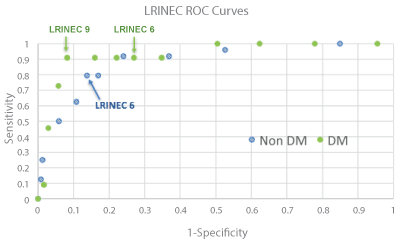 Figure 2: Receiver operating characteristics (ROC) curves for laboratory risk indicators for necrotizing fasciitis (LRINEC) score in diabetic (DM) and non-diabetic patients. Each point represents a LRINEC score cutoff from 1 to 13. For non-diabetic patients, LRINEC cutoffs of 11, 12 and 13 all gave a sensitivity and 1-specificity of 0 and 0 respectively. View Figure 2
Figure 2: Receiver operating characteristics (ROC) curves for laboratory risk indicators for necrotizing fasciitis (LRINEC) score in diabetic (DM) and non-diabetic patients. Each point represents a LRINEC score cutoff from 1 to 13. For non-diabetic patients, LRINEC cutoffs of 11, 12 and 13 all gave a sensitivity and 1-specificity of 0 and 0 respectively. View Figure 2
Our results confirm our hypothesis that the LRINEC score is significantly higher in diabetic patients both with cellulitis and with necrotizing fasciitis. This is not a surprising result given the normal laboratory manifestations in diabetic patients, including hyperglycemia and electrolyte abnormalities seen in diabetic nephropathy. This baseline elevation leads to an increased sensitivity but a decreased specificity for the same LRINEC cutoff. The clinical implication for this is that using the same cutoffs will generate more false positives. In our cohort, almost 30% of our diabetic patients with cellulitis had a LRINEC of 6.
When evaluating this test in the context of our relatively low prevalence of necrotizing fasciitis compared to cellulitis, the negative predictive value for both non-diabetic and diabetic patients is very high using a LRINEC cutoff of 6. Conversely, the positive predictive values for both non-diabetic and diabetic patients is low. Clinically, this means that at least in our patient population, the LRINEC score is far more effective at excluding necrotizing fasciitis as a diagnosis but is a poor test at confirming its presence. This is exaggerated in diabetic patients given the baseline elevated laboratory values. While a low LRINEC score is reassuring given the negative predictive value and generally high sensitivity of the test, in our population a high LRINEC score is not strongly predicative of necrotizing disease. In these cases, a clinical diagnosis is still required, and the disease should always be managed with a high index of suspicion.
One other study has directly compared the differences between necrotizing fasciitis in diabetics versus non-diabetics. This study was a retrospective review of 127 patients with necrotizing fasciitis and showed that patients with necrotizing fasciitis and diabetes were more likely to have multiple comorbidities, renal impairment, and end stage renal failure [20]. These results help to explain our findings of an increased LRINEC score in diabetic patients. This study also showed that diabetic patients were less likely to have tenderness and hypotension than their non-diabetic counterparts, as well as a longer duration of symptoms and hospital stay, lower rate of diagnosis at admission, and high rate of amputation [20]. This lack of traditional findings in conjunction with overall worse outcomes highlights the importance of a high index of suspicion when dealing with necrotizing fasciitis in diabetic patients. In looking at test sensitivity, this study showed that using a LRINEC score cutoff of 6 resulted in a similar sensitivity between diabetics and non-diabetics (78.9% versus 63.3%). Using a score cutoff of 8 resulted in a more sensitive test in diabetic patients (68.4% versus 28.6%), though overall less sensitive test in both patients [20].
There are some definite limitations to our study. The first being the pitfalls of all retrospective reviews, including the reliance on the quality of the input data. In our case, we relied heavily on accurate medical coding. While we did verify necrotizing fasciitis cases from surgical reports, it is still possible that we missed both patients with cellulitis and necrotizing fasciitis from improper coding. The second limitation to this study is the limited number or necrotizing fasciitis cases we had, specifically in diabetic patients. While we did have enough patients to reach clear statistical significant, a larger sample number in this cohort would allow for a more accurate ROC curve. Finally, over 50% of our patients had to be excluded from this study incomplete laboratory values. This may present some selection bias as incomplete laboratory work-up may have been secondary to either very severe disease that did not allow time for additional work-up, or to a less severe clinical presentation that did not warrant additional laboratory work-up. While it is difficult to assess the exact effect of this potential selection bias, one would expect this to affect both diabetic and non-diabetic patients equally.
In conclusion, while the LRINEC score is a powerful tool to help evaluate patients with cellulitis symptoms for necrotizing fasciitis, it is still just an adjunct to a clinical diagnosis. Necrotizing fasciitis is a very serious illness and must be managed in all cases with a very high index of suspicion. That being said in all patient populations, diabetic patients with both cellulitis and necrotizing fasciitis have higher LRINEC scores and using the same cutoff value for diabetic patients as non-diabetic patients will yield a significant number of false positives. In our population, it is a far more powerful tool to rule out necrotizing fasciitis as a disease process, but a poor tool for confirming its presence.
No sources of support were used in the preparation of this manuscript.