At birth Perirenal Adipose Tissue (PrAT) exhibits a brown-like phenotype and then becomes phenotypically white with ageing and obesity, potentially contributing to the onset of chronic disease. Little is known about the composition of PrAT in the later years of life and whether it is modulated by different types of disease.
PrAT samples from 223 autopsies were taken from Newcastle General Hospital between 1972-1973. After histological examination they were graded (scale of 1-5) based on the abundance of multilocular brown adipocytes. The effect of age, chronic disease and obesity on brown adipocyte distribution was then examined.
Samples from individuals aged 0-19 years demonstrated the highest presence of brown adipocytes. PrAT acquired a white phenotype with age until the later years of life (i.e. 80-100 years) when the brown phenotype reappeared. Interestingly the presence of brown adipocytes was also increased in individuals with chronic conditions, particularly diabetes and hypertension compared to controls. As expected, we observed a white phenotype in overweight and obese subjects with lean individuals exhibiting a greater amount of brown adipocytes.
PrAT exhibits a brown-like phenotype during old age and in the presence of chronic disease the mechanisms for which remain to be examined.
Brown adipose tissue, Ageing, Disease, Historical
Brown Adipose Tissue (BAT) is a thermogenic tissue that utilises glucose and lipids to generate heat following activation of the Unique Uncoupling Protein (UCP)1 [1]. The recent re-discovery of BAT in adult humans which is inversely associated with age, BMI and metabolic health has led to a resurgence in research interest [2,3], and it has been postulated that activation of existing BAT or recruitment of brown adipocytes in classical white adipose depots could prevent obesity and related metabolic disease [4]. One major adipose depot is located within the Perirenal region (PrAT), which is brown-like at birth and then becomes phenotypically white with ageing [5]. We present here previously unpublished novel data collected from autopsies between 1972-1973 suggesting that PrAT may reacquire a previously overlooked brown-like phenotype in both old age and certain diseases. We acknowledge that there are a number of limitations to the data presented. Limited original raw data remains for this 1972-1973 cohort so that any relationships to gender, body mass and ambient temperature at time of death remain unexplored. The precise 'n' for each age group is unknown preventing further statistical analysis and furthermore, techniques for quantification of thermogenic genes, particularly UCP1, were not readily available at the time of the study. Further cohort studies examining the distribution of brown adipocytes throughout the life-cycle in multiple depots are thus required to address the above points adequately. Despite this, a potential increase in the presence of brown adipocytes in the oldest subjects is intriguing as a role for BAT in old age is new and speculative.
For a total of 223 autopsies conducted at the Newcastle General Hospital the adrenal gland was removed and the encompassing PrAT was collected for subsequent analysis. Age, sex, weight and height were recorded for each autopsy in addition to other factors that were hypothesised to affect the total mass or function of BAT such as nutritional state (based on the pathological description), cause of death and the presence of other medical conditions. Between 3-8 sections were cut from each sample, fixed in either glutaraldehyde or formal saline and embedded in paraffin with 4µ sections then stained by haematoxylin and eosin. After microscopic examination these were each graded based on the occurrence of brown like adipocytes as per the morphology of each sample (Figure 1A, Figure 1B, Figure 1C and Figure 1D) based on the following criteria:
 Figure 1: Morphological characteristics of Perirenal Adipose Tissue (PAT). A) 'Grade 1' PAT ×10 magnification; B) 'Grade 3' ×40 magnification; C) 'Grade 4' ×10 magnification; D) 'Grade 5' ×40 magnification and the distribution of grades across the life course (E). View Figure 1
Figure 1: Morphological characteristics of Perirenal Adipose Tissue (PAT). A) 'Grade 1' PAT ×10 magnification; B) 'Grade 3' ×40 magnification; C) 'Grade 4' ×10 magnification; D) 'Grade 5' ×40 magnification and the distribution of grades across the life course (E). View Figure 1
'Grade 0' - No multilocular cells present.
'Grade 1' - One or two small foci or multilocular fat cells present within "sheets of white fat".
'Grade 2' - Small foci of multilocular fat cells scattered more frequently through the fat.
'Grade 3' - More numerous, larger foci of multilocular cells present but make up less than one half of the cells.
'Grade 4' - One half to two-thirds of the cells present is multilocular and appears at the centre of lobules and spreading towards the edge.
'Grade 5' - More than two-thirds of the cells present are multilocular brown adipocytes.
To examine the relationship between age and the distribution of brown adipocytes in PrAT samples were age-banded (Figure 1E). As expected, large amounts of BAT (Grades 3-5) were prevalent in subjects < 20 years of age. The progression to adulthood was associated with PrAT acquiring a whiter phenotype becoming increasingly unilocular with advancing age. Interestingly an increase in the abundance of brown adipocytes (i.e. grades 4-5) was demonstrated in the subset of patients from the eldest age groups (i.e. aged 80-100 years) suggesting that the perirenal depot acquires a brown phenotype in the later years of life.
In order to determine the effect of chronic disease on adipocyte distribution samples were grouped by the cause of death (Figure 2A) with the accidental deaths group acting as controls. PrAT of individuals who died accidentally consisted primarily of unilocular white adipocytes (i.e. grades 1-3). The presence of a chronic condition increased the amount of brown adipocytes with this pattern most evident for patients with systemic hypertension and other endocrine diseases, including diabetes. Grouping the samples by their respective donors body mass (Figure 2B) demonstrated that obese individuals have a higher proportion of white adipocytes (Grades 0 and 1) in PrAT and a lower proportion of brown adipocytes (Grade 3-Grade 5) compared to the 'average' or 'thin' groups.
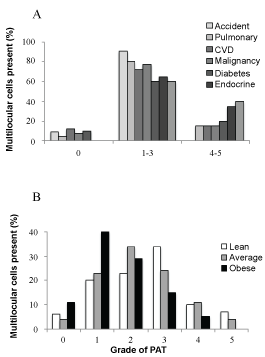 Figure 2: Grading of perirenal adipose tissue by cause of death (A) and body mass (B). View Figure 2
Figure 2: Grading of perirenal adipose tissue by cause of death (A) and body mass (B). View Figure 2
Our results from this historical cohort demonstrate those PrAT displays BAT-like characteristics throughout the life span and that BAT presence is enhanced in the youngest and the oldest individuals. The finding that presence of BAT was maximum in the youngest and declined between 20 and 79 years of age is in keeping with the more recent investigations that have demonstrated that incidence and activity of BAT decreases with age [2]. This report further suggests that the presence of certain chronic conditions are associated with an increased presence of brown adipocytes at death implying that BAT could be retained in the presence of underlying disease including systemic hypertension, diabetes or other endocrine conditions. These results, first discovered over 40 years ago, also illustrate a relationship between body mass and the proportion of brown adipocytes in PrAT. This is not unexpected in view of current research, however, it is worth noting that despite the existence of such work, the first reported association between BAT and body mass was not made until its recent re-discovery in adults [2,3]. This report possibly represents one of the earliest discoveries of the relationship between body mass and brown adiposity and brings to light research that was well ahead of its time.
The authors have nothing to disclose.