Claudication is a common manifestation of Peripheral Arterial Disease (PAD) and atherosclerosis. Patients with claudication are at increased risk of cardiovascular events and limb-related complications. Optimal medical therapy is effective in relieving patient's symptoms and reducing their risk of cardiovascular events.
A 54-year-old male with hypertension, hyperlipidemia, tobacco use and sedentary lifestyle presented with left lower extremity exertional pain. Lower extremity segmental arterial study was consistent with the presence of Lower extremity arterial disease. He was managed medically with resolution of his claudication symptoms and improvement of his segmental study.
Medical management of patients with claudication is extremely effective and should be provided to all patients with claudication, even those who undergo revascularization to reduce their risk of cardiovascular events, improve their claudication symptoms and general health.
Claudication, Peripheral Arterial Disease (PAD)
The prevalence of Peripheral Arterial Disease (PAD) among adults aged > 40 years is 4.3% and 13.4% among adults aged > 65 years. The prevalence of claudication is 1.9% in males and 0.8% in females aged > 40 years. The majority of patients with PAD are either asymptomatic or have atypical leg symptoms [1-3]. Major risk factors for lower extremity arterial disease are increasing age, tobacco use, diabetes, hypertension and Hyperlipidemia [1,3]. Atherosclerosis is the most common cause of PAD, but other causes like embolism, vasculitis, Buerger's disease, fibromuscular dysplasia and popliteal entrapment syndrome should be considered.
The majority of patients with lower extremity arterial disease presents with atypical leg complaints or are asymptomatic [4]. Patients with claudication usually present with a variety of symptoms like dull aching, tightness, pain, weakness or fatigue in the calves, thighs or buttocks triggered by physical activities. Patients with claudication usually have co-morbid conditions that could cause different types of pain in the same limb like osteoarthritis, bursitis and neurological causes. The location and severity of symptoms depends on the level and degree of their arterial disease. Symptoms resolve after resting for at least few minutes. Symptoms that start or resolve immediately after initiating or stopping physical activities suggest alternative diagnosis especially musculoskeletal and neurological causes. Also, sitting is not required for resolution of claudication and symptoms that resolve only after sitting or lying down are suggestive of musculoskeletal or neurological causes. Some patients will not complain about their claudication especially the elderly patients because they attribute them to their age or other coexisting conditions such as osteoarthritis. It is important to elicit information regarding the onset of symptoms as gradual or sudden, unilateral or bilateral, and whether the symptoms were triggered by usual physical activities to the patient or after the patient started a new type of physical activity. Ischemic rest pain suggests severe peripheral arterial disease and could be present with severe claudication. It usually affects the toes and feet and is worse with recumbence and better with dangling the feet down. Patients with severe PAD may have coldness, paresthesia, weakness and atrophy of affected foot or limb.
Of patients with suspected peripheral arterial disease and claudication needs to be thorough and detailed.
Patients' ability to transfer from chair to bed or vice versa and their ability to walk even several steps should be noted. If a patient is unable to put weight on one leg, this could indicate the presence of musculoskeletal etiology such as arthritis. If a patient has difficulty transferring from lying down in bed to sitting up and transferring to chair secondary to back pain, this could suggest the presence of radiculopathy. Lower extremities should be inspected for any unilateral or bilateral deformities, atrophy, amputation, ulcer, gangrene, skin and nail dystrophic changes, discoloration and old scars. The presence of varicose veins and swelling should be documented. Socks and shoes should be inspected for discharge and proper fitting, especially in patients with foot lesions. Dependent rubor of the feet and pallor are suggestive of severe ischemia.
Of the legs and feet for warmth, coldness, tenderness, and peripheral pulses, is essential. Cold feet could indicate ischemia and warmth could suggest infection. Normal pedal pulses reduce the possibility of significant lower extremity arterial disease. Dorsalis pedis is congenitally absent in some people. Diminished femoral pulse is caused by aortoiliac lesions. The presence of normal femoral pulse with diminished popliteal artery pulse indicates mostly superficial femoral artery pathology. Irregular pulse could indicate cardiac arrhythmias and possible cardiac source of embolism as a cause of PAD. Sensation should be tested, especially in those with diabetes or in those with symptoms like numbness. Patients with peripheral neuropathy could have severe ischemia with minimal symptoms (rest pain equivalent). Palpation of the aorta and peripheral pulsation from the radial, carotid, femoral, popliteal, dorsalis pedis and posterior tibial arteries is essential to detect irregular rhythm, absence (0), diminished (1+) or normal (+2) pulsation and the presence of aneurysm.
If pedal pulses are not palpable or diminished, a hand held Doppler, if available, could be used to localize and investigate pedal pulses further. The location of the DP pulse is lateral to the extensor pollicis longus tendon along the dorsum of mid foot and the posterior tibial pulse is posterior to medial malleolus. Absent doppler signals suggest congenitally absent pulse or occlusion of that artery. Doppler signal is characterized as triphasic, biphasic or monophasic. Monophasic signal suggest the presence of significant arterial disease or inflammation.
For abnormal heart sounds and bruit over the carotid, renal and femoral arteries. The presence of arterial bruit is a specific sign for significant arterial disease.
The most important diagnosis to consider is neurogenic claudication. Usually patients have back pain or history of back pain. The pain may be reduced slightly by bending forward as with using a shopping cart. Pain is usually not relieved by just stopping walking as in patients with IC but requires patients to sit or lie down to relieve their pain. Venous Claudication is usually relieved by elevating the feet and is usually triggered by standing and sitting instead of walking. Musculoskeletal causes like arthritis, bursitis, tendinitis and myositis are common causes of lower extremity pain and could be differentiated from vascular claudication by detailed history and physical exam.
Is usually made after taking detailed history, doing physical examination and noninvasive studies.
Normally the pressures around the ankle are higher than the brachial pressures due to added aortic recoil pressure. The basic principal of ABI is that the ratio of the higher ankle pressure over the higher of the brachial pressure should be one or above. To increase specificity of the test, 0.9 or 0.95 is used as a cut off for diagnosis of PAD. There are significant limitations on the value of ABI related to non-compressible vessels secondary to medial calcifications, as in patients with diabetes and chronic kidney disease. Bedside ABI check is quick, safe, and cheap and can be done without expensive equipment or significant time commitment and can be done by nursing staff with some training. It can be checked using simple a stethoscope and a blood pressure cuff. However hand held Doppler is more sensitive, especially in patients with PAD. A pressure difference between the brachial artery pressures of more than 12 mmHg could be secondary to subclavian or innominate artery stenosis. Other causes are coarctation of aorta, vasculitis and external compression of the subclavian artery by tumor.
Is especially helpful in patients with arterial calcification where ABI can be falsely elevated or cannot be calculated. Digital arteries are not affected by arterial calcification. Normal toe brachial index range is 0.8-0.9. It could be abnormal in patients with digital arterial disease like in patients with Raynaud's phenomenon.
Is done by measuring systolic pressure at the brachial, upper thigh, above knee, below knee and around the ankle. Vertical or horizontal pressure gradient of more than 20 mmHg indicate a hemodynamically significant arterial stenosis. This is helpful in detecting PAD and sometimes the location of possible narrowing.
Normally demonstrate sharp systolic upstroke and decline during diastole. There is a retrograde flow during diastole that causes flow reversal and dicrotic notch. Loss of dicrotic notch is the earliest sign of arterial disease followed by delayed systolic upstroke and the peak is rounded and the down slope bowed away from baselines. It is especially useful in patients with arterial calcification and non-compressible vessels.
Tracings are important for diagnosing small vessels arterial disease like vasculitis and raynaud's. This is important especially in patients with normal ABI but with signs and symptoms of digital ischemia.
Are useful when resting ABIs are normal and the patient has symptoms of claudication or possible pseudoclaudication.
Is used to diagnose PAD and to assess its severity and anatomical location. This includes real time B-mode (gray scale) imaging to best define vessels anatomy such as the presence of aneurysm and plaque as well as its morphology. Pulsed wave and color doppler imaging is used to detect blood flow, velocities and to estimate degree of stenosis.
Are useful to assess severity and anatomy of PAD and choose appropriate endovascular or open revascularization.
Is the gold standard for defining the anatomy of arterial disease. Sometimes patients undergo contrast angiography angiogram without CTA or MRA to minimize the amount of contrast dye received by the patients especially those with chronic kidney disease. Arterial angiography requires less contrast material than CT angiography. In addition, it provides an opportunity for endovascular intervention at the same setting.
A 54-year-old man was seen for left calf pain that occurs after walking about two blocks on flat ground for at least six months. The pain is usually triggered at a shorter distance when walking at a faster pace or uphill and is relieved after resting for a few minutes. The pain extends to his left thigh if he continues to walk. He denied any pain in his feet or legs while resting or standing. He did not seek medical attention prior to this visit because he attributed the pain to being physically inactive. His past medical history includes hypertension, hyperlipidemia and tobacco use. He was on hydrochlorothiazide 25 mg/day for hypertension. He had no allergies. His parents have hypertension and hyperlipidemia. He was single and lived alone. He did not graduate from high school and was working as a cashier in a supermarket. He has a 32 pack-year history of smoking. He does not drink. His review of symptoms was negative.
On examination, his blood pressure was 146/84 with pulse of 67. His weight was 167 pounds. He has no neck vein distension or carotid bruit. Heart: normal S1 and S2, no murmurs, regular rate and rhythm. Chest: Clear to auscultation. The abdomen was soft without tenderness, guarding, mass, rebound or organomegaly. Bowel sounds were normal. No CVA tenderness or inguinal adenopathy. His feet were warm symmetrically, pink and well perfused with no clubbing, cyanosis, edema or signs of embolization or ulcers. On the right side his femoral, popliteal, dorsalis pedis and posterior tibial pulses were easily palpable and 2+. On the left side his femoral, popliteal, dorsalis pedis and posterior tibial pulses were palpable but diminished and 1+. He had left femoral bruit. He had a normal range of motion of the knee and hip joints and there were no skin lesions. Lower extremity arterial segmental study was done and confirmed the presence of mild left lower extremity resting ischemia (Figure1).
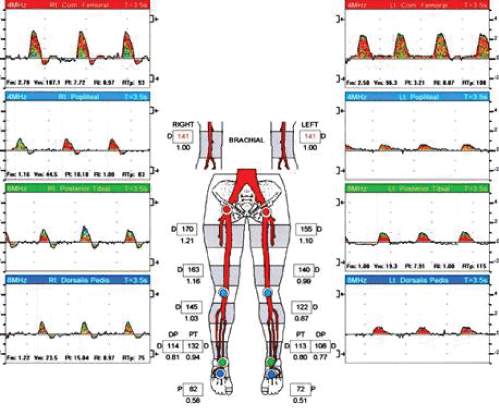 Figure 1: Lower extremity segmental study. View Figure 1
Figure 1: Lower extremity segmental study. View Figure 1
The resting ankle-brachial index is 0.94. The toe pressure is 82 mmHg with a TBI of 0.58 Segmental pressures across the lower extremity demonstrate significant resting vertical pressure gradient. Doppler waveform analysis shows relatively normal arterial waveforms at all levels with preserved diastolic flow reversal. This represents borderline resting right lower extremity arterial study.
The resting ankle-brachial index is 0.80. The toe pressure is 72 mmHg with a TBI of 0.51. Segmental pressure at the high thigh level is normal. There is significant pressure gradient between the above knee popliteal pressure and the ankle level pressures. Doppler waveform analysis shows an abnormal monophasic high thigh waveform with continuous forward flow during the cardiac cycle. This represents mild left lower extremity arterial disease secondary to multilevel arterial disease (inflow disease and infrainguinal).
In this case, the diagnosis of claudication secondary to atherosclerosis arterial disease with associated risks of limb loss, heart disease, stroke and death were discussed in detail with the patient. Therapeutic interventions were discussed in terms of targeting one of two main goals or both. The first goal is to reduce claudication symptoms and risk of future limb-related complications and second is to improve his health in general and to reduce his risk of heart disease, stroke, and death. Within that frame, indications, risks and potential benefits of every proposed intervention were discussed. It was emphasized to him that tobacco use is his number one risk factor and quitting smoking is the most important step he could take to prevent further progression of his peripheral arterial disease, reduce risks of heart disease, stroke and to improve his overall health. He was provided with smoking cessation counseling and was able to quit smoking three months after his first visit using nicotine patches and gum for four months. Blood tests were done as shown in Table 1. He was started on aspirin 81 mg, atorvastatin 40 mg and lisinopril 20 mg once daily. He was referred to dietary service to improve his blood pressure control and to lower his lipids.
Table 1: Three year Data Comparison (Blood pressure, cholesterol, estimated CV risk scores, ABI and walking distance). View Table 1
He was advised to start doing regular exercises like walking and or biking. He was referred to physical therapy and although did not enroll in a physical rehabilitation program, he started to walk regularly. He was reassured that walking through his calf pain is not harmful to his health. Shortly after that he adopted a dog and started walking and later jogging with his dog on a regular daily basis. The time to onset of his left leg pain became progressively longer and the severity of the pain was milder. He was able to gradually increase his exercise level from two blocks at the time of his initial visit to jogging two miles almost every day three years later. Lower extremity segmental study three years later (Figure 2).
 Figure 2: Lower extremity segmental study three years later. View Figure 2
Figure 2: Lower extremity segmental study three years later. View Figure 2
The resting ankle-brachial index is 1.15. The toe pressure is 61 mmHg with a TBI of 0.48. Segmental pressures across the lower extremity do not demonstrate any significant resting vertical or horizontal gradients. Doppler waveform analysis show relatively normal arterial waveforms at all levels with preserved diastolic flow reversal.
The resting ankle-brachial index is 0.96. The toe pressure is 66 mmHg with a TBI of 0.52. Segmental pressures across the lower extremity demonstrate significant resting vertical and horizontal gradients. Doppler waveform analysis show relatively normal arterial waveforms at all levels with preserved diastolic flow reversal.
Claudication is an important symptom of lower extremity arterial disease and more importantly is an indicator of systemic atherosclerosis with associated high cardiovascular events and death. Although limb related complications are frightening to patients, limb related prognosis is usually benign. Within five years, only 1-2% of patients (and likely less for those who adopt a healthy life-style) with claudication progress to develop more severe symptoms like rest pain, ischemic ulcers or gangrene and for whom revascularization is usually effective. However, 15-30% will die mostly secondary to cardiovascular disease and 25% will have a heart attack or a stroke [5,6].
Also, patients with PAD have greater functional decline and mobility loss than those without PAD. Even patients with asymptomatic PAD were found to have poor functional capacities and poorer quality of life in comparison to those without PAD. Patients with claudication are more sedentary and their free-living daily physical activity was significantly lower than healthy subjects. Because of this functional decline, increased risks for cardiovascular events and ischemic complications like ulcers, gangrene and amputations and the associated need for hospitalizations, they have a poor quality of life and higher rates of depression than those without PAD [7-12].
So the main therapeutic goals in patients with claudication are to prevent cardiovascular events especially fatal and nonfatal MI and stroke, reduce limb-related symptoms and complications and improve functional abilities and quality of life. To help patients with claudication make an informed decision regarding their treatment and to motivate them to make necessary and difficult life style changes, the above goals need to be conveyed clearly to patients. Health care providers need to explain to their claudication patients that although their symptoms are related to their legs, the most serious consequences of atherosclerosis are related to ischemic heart disease and stroke. In that sense, Paul Brand's statement that "Pain is God's greatest gift to mankind" applies to patients with claudication if viewed as an opportunity and a "teachable moment" to reduce cardiovascular events, improving overall health and reducing claudication symptoms. We need to help patients shift their focus from only their immediate presenting symptoms to the general risk on their health especially cardiovascular complication. This is not an easy task and requires multidisciplinary team and supportive health care systems placing significant emphasis on prevention.
The most important risk factors for PAD are tobacco use, obesity, diabetes, hypertension, hyperlipidemia and low kidney function [13,14]. Mortality rate was reduced by moderately vigorous exercise and quitting smoking by 23% and 41% respectively [15]. Risks of death, heart attack and amputations are significantly higher in patients with PAD who continue to smoke than those who are able to quit smoking [16-19]. Supervised treadmill exercise programs increased maximal treadmill walking time by 50-200% [20-23].
Furthermore, it was found that in adults who are at risk for development if Diabetes (BMI > 24 and fasting blood sugar 95-125 mg/dl or 145-199 mg/dl at the 2 hours after 75 gm of oral glucose). 150 minutes of exercise every week and 7% weight reduction over 24 weeks resulted in 58% reduction in incidence of DM in comparison to placebo [24,25].
Statin [26-30] and ACEI [31,32] have been found to reduce mortality and possibly improve claudication symptoms. Available FDA approved medication to treat Claudication is cilostazol [33]. Pentoxifylline was not found to be effective in the treatment of claudication. Antiplatelet therapy has been a cornerstone of medical management of patients with symptomatic PAD as recommended by the latest AHA Guidelines. "Antiplatelet therapy with aspirin alone (range 75-325 mg per day) or clopidogrel alone (75 mg per day) is recommended to reduce MI, stroke, and vascular death in patients with symptomatic PAD" [34]. Recent publications and advances in the use of newer antiplatelets and anticoagulants like Ticagrelor and Rivaroxaban in patients with PAD [35,36] could potentially change future guidelines related to the management of patients with PAD. Revascularization (endovascular or open) is indicated in patients with significant lifestyle limiting claudication with inadequate response to optimal medical management. Medical management is crucial in these patients to reduce their risk of cardiovascular events. Revascularization has risks of preoperative mortality and morbidity and questionable long term patency [37,38]. Treatment selection should be individualized depending on patients' characteristic, arterial anatomy and risk of perioperative adverse events. Endovascular procedures should not be performed in patient with PAD to only prevent the progression to critical limb ischemia [36].
This case demonstrated clearly that optimal medical therapy resulted in the resolution of claudication symptoms but significant increase in his level of daily physical activities, risk reduction of cardiovascular diseases and significant improvement of his overall wellbeing. This is the most cost-effective treatment in patients with claudication. Revascularization is indicated for individuals with a lifestyle-limiting disability due to claudication if they do not have adequate response to medical management [5]. Although life-style changes are very challenging and not every patient is capable of achieving these goals, health care providers need to provide every patient with adequate information and support to allow them make informed decisions about their health and to motivate them to achieve their goals. Health care systems require providing essential support services like physical therapy, dietary and smoking cessation counseling. In the 21st century, it is not enough to just tell the patients with claudication to "quit smoking and go walking" or to recommend revascularization without providing them with recommended optimal medical management.
The author has no potential conflict of interest or financial relationship to disclose.