We present a case of lung adenocarcinoma with suspected metastasis to the pituitary in a 65-year-old male originally presenting to the ER to rule out a diagnosis of pulmonary tuberculosis based on a lesion found on a recent chest X-ray.
A 65-year-old homeless African American man with a past medical history significant for hypertension, Chronic Obstructive Pulmonary Disease (COPD), diabetes mellitus type II, multiple substance abuse disorder, major depression, and successful treatment of tuberculosis presented to the Miami VA emergency department with depressed mood, fatigue, and suicidal ideation. This Vietnam veteran reported a lifetime of moderate alcohol use, 10 pack-years of cigarette smoking, and 25 years of crack cocaine use, which he quit four years prior to presentation. He was seeking a bed in a homeless shelter when a cavitary lesion was found in the right upper lobe of his lung (as shown in Figure 1) during a routine chest X-ray screening. They recommended he visit our emergency department for diagnosis.
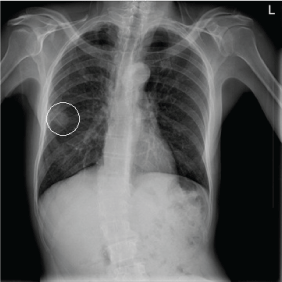 Figure 1: PA chest x-ray upon admission. Tumor is encircled. View Figure 1
Figure 1: PA chest x-ray upon admission. Tumor is encircled. View Figure 1
A chest CT revealed a 1.7 × 2 cm mass with internal but non-central calcification and spiculated margins on the patient's right lung. In addition, a 2.5 × 2.4 cm pleural-based cavitary lesion and a 1.5 × 1.0 cm ill-defined parenchymal opacity were noted in the right upper lobe (as shown in Figure 2).
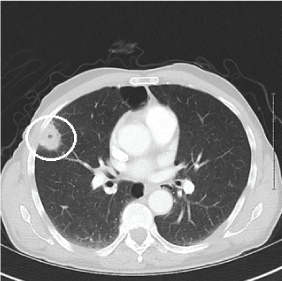 Figure 2: Lung CT w/contrast. Tumor is encircled. View Figure 2
Figure 2: Lung CT w/contrast. Tumor is encircled. View Figure 2
Although the mass was suspected to be lung cancer, the patient had a history of treated tuberculosis and was thus admitted to the hospital to rule out active tuberculosis. Of note, the patient was followed closely by psychiatry and was continued on his home dose of mirtazapine for treatment of depression.
At 6:00 AM on day 2 of hospitalization, the patient was found unresponsive, biting his tongue, and with urinary incontinence consistent with a seizure. He was noted to be severely hypoglycemic with a finger stick glucose reading of 20 mg/dL after receiving 25 units of insulin glargine (his usual home dose). Administration of dextrose showed improvement of his glucose level but still with persistent deficits in mental status. He was transferred to the MICU for further management. The presumed etiology of his seizure was hypoglycemia at the time as he did not have a documented history of epilepsy. He remained altered and unresponsive to questioning for 30 minutes but without signs of overt epileptic activity. EEG was not performed. This unresponsiveness was initially assumed to be due to a postictal state, a second hypoglycemic insult, or some combination of both.
A non-contrast CT brain was ordered, and a possible suprasellar mass was noted with a recommendation to obtain a contrast-enhanced MRI brain for better evaluation. On MRI (Figure 3A and Figure 3B), a 2.5 cm suprasellar mass was confirmed with pronounced invasion into the pituitary gland leading to significant difficulty even delineating individual structures. There was also significant right cerebellar vasogenic edema (Figure 4) and sulcal enhancement (Figure 5) that is highly consistent with malignancy. Subsequently a PET scan (Figure 6) was performed showing the sellar/suprasellar mass to be hypermetabolic. Other findings confirmed by the MRI and head CT included a lesion obstructing the pituitary stalk (Figure 7), and marked bilateral frontal cerebral white matter disease. The patient was subsequently stabilized with improvement of mental status and transferred back to the medical team.
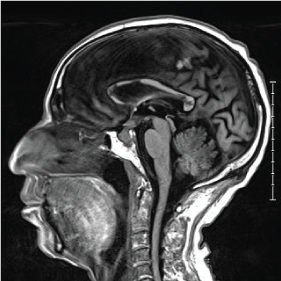 Figure 3a: T1 pre-contrast sagittal MRI. View Figure 3a
Figure 3a: T1 pre-contrast sagittal MRI. View Figure 3a
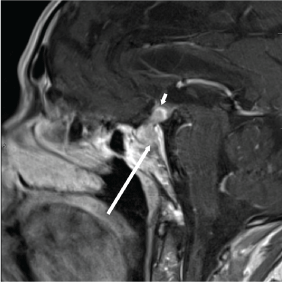 Figure 3b: T1 brain MRI post-contrast. Long arrow shows invasion into clivus, and short arrow shows invasion into the infundibulum and third ventricle floor. View Figure 3b
Figure 3b: T1 brain MRI post-contrast. Long arrow shows invasion into clivus, and short arrow shows invasion into the infundibulum and third ventricle floor. View Figure 3b
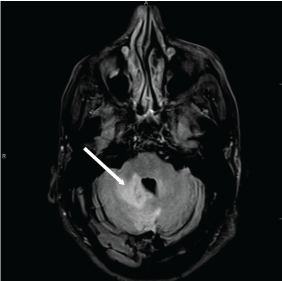 Figure 4: T2 Brain MRI. Arrow shows right cerebellar vasogenic edema and local mass effect on the forth ventricle. View Figure 4
Figure 4: T2 Brain MRI. Arrow shows right cerebellar vasogenic edema and local mass effect on the forth ventricle. View Figure 4
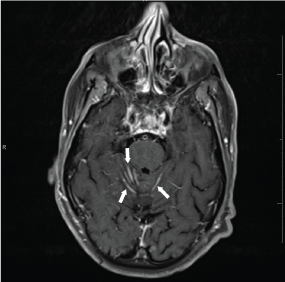 Figure 5: T2 Brain MRI. Arrows indicate sulcal enhancement and likely cerebellar folia surface metastases. View Figure 5
Figure 5: T2 Brain MRI. Arrows indicate sulcal enhancement and likely cerebellar folia surface metastases. View Figure 5
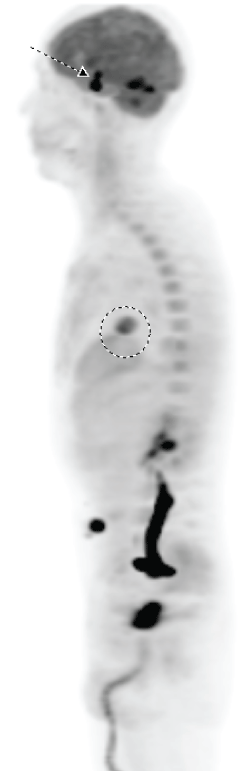 Figure 6: PET scan. Arrow shows suspected brain metastasis and circle shows lung adenocarcinoma. View Figure 6
Figure 6: PET scan. Arrow shows suspected brain metastasis and circle shows lung adenocarcinoma. View Figure 6
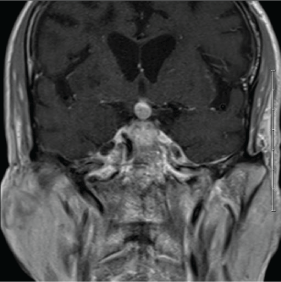 Figure 7: T1 post-contrast coronal MRI showing the lesion obstructing the pituitary stalk. View Figure 7
Figure 7: T1 post-contrast coronal MRI showing the lesion obstructing the pituitary stalk. View Figure 7
Laboratory tests were performed to assess the degree of pituitary functioning including free and total testosterone levels, FSH, LH, TSH, T4, and T3 which were all found to be abnormally low, consistent with Panhypopituitarism (Table 1). A normal prolactin level ruled out a diagnosis of prolactinoma. The patient was subsequently started on Synthroid (levothyroxine) and his thyroid function responded appropriately.
Table 1: Laboratory tests were performed to assess the degree of pituitary functioning including free and total testosterone levels, FSH, LH, TSH, T4, and T3 which were all found to be abnormally low, consistent with panhypopituitarism. View Table 1
Three separate sputum collections were also eventually collected, all of which were negative for acid-fast bacteria, ruling out the diagnosis of active tuberculosis. During daily rounds, the veteran reported no shortness of breath, cough, vision changes or visual field deficits, and the team observed no other abnormal symptoms except persistent weight loss, decreased PO intake, and depressed mood.
A biopsy of the right upper lobe lung lesion was performed and this confirmed the diagnosis of poorly differentiated lung adenocarcinoma with extensive necrosis. Thus it was presumed that the pituitary lesion represented a metastasis of the lung malignancy to the brain. However, due to the patient's refusal of further treatment with radiation and chemotherapy and unlikely significant benefit considering the stage of his cancer, biopsy of the brain lesion was withheld. Subsequently the patient was transferred to palliative care where he succumbed to his illness within a few weeks.
Reported rates of the occurrence of metastases to the pituitary vary greatly. In autopsy series of patients with malignant tumors, rates between 1 and 3.6% were found [1]. However, rates as high as 27% have been reported in autopsy series where the pituitary and surrounding sella turcica were evaluated [2]. Breast and lung cancer are the most frequent origin of metastasis [1,2].
Comparatively, primary pituitary adenomas of clinical significance were found to have a prevalence of 80-90 per 100,000 in the general population in cross sectional studies [3-5]. However, using radiological and autopsy studies which include clinically insignificant incidentalomas, rates as high as 16.7% were found [6].
Though it is difficult to make the distinction on neuroimaging alone, Fassett, et al. reported that neuroimaging can help to distinguish metastatic disease by the presence of thickening of the pituitary stalk, invasion of the cavernous sinus, and sclerosis of the surrounding sella turcica [2]. The average pituitary stalks measures 1.9 mm at its insertion [7], while our patient's stalk was thickened, measuring 4 mm. Our patient's lesion also demonstrates invasion into the cavernous sinus, and involvement of the pituitary and floor of the third ventricle. Moreover, Post reports that metastatic disease can potentially be distinguished by the presence of a visually separate tumor and extensive bone destruction [8]. On sagittal MRI our patient's lesion shows invasion into and destruction of the clivus. These radiographic findings increase the likelihood of the lesion being a metastasis.
Notably, posterior pituitary cancers and metastatic lesions are more common than those of the anterior [9]. One theory for this is that the posterior pituitary gland is supplied with blood from the systemic circulation while the anterior pituitary is supplied by the portal circulation [9]. Growing large enough, tumors from either location may impinge on the anterior pituitary and cause hypopituitarism via mass effect [1].
Only 7% of pituitary metastases are reported to show any symptoms [10]. Common symptoms included diabetes insipidus, anterior hypopituitarism, visual field defects, retro-orbital pain, and ophthalmoplegia. Our patient is particularly notable for panhypopituitarism being the first presenting symptom of his lung cancer. Patient did not exhibit any signs of diabetes insipidus such as polydipsia or polyuria or any visual deficiencies.
Our patient originally presented with neurologic and psychiatric symptoms that are commonly attributed to or associated with old age, homelessness, and veteran status. These included depressed mood, fatigue, and suicidal ideation. While well established as possible consequences of both pituitary disease [11] and cancer, these symptoms are not generally the main reasons patients seek medical care for these disease processes. A retrospective holistic analysis of this patient's presentation, including the psychiatric changes, a hypoglycemic event, and low hormonal values, provides a clear indication of panhypopituitarism. A high level of clinical suspicion among physicians when evaluating patients with such signs, symptoms, and laboratory derangements may lead to earlier and more frequent diagnosis of malignancy involving the pituitary.
Because our patient opted for palliative care, we did not pursue confirmatory biopsy of the brain lesion. The clinical presentation of this patient in combination with his neuroimaging studies suggest that this pituitary lesion is likely, but not definitely, a metastasis. In addition, his rapid clinical deterioration supports the likelihood of metastasis.
Metastasis to the pituitary gland causes a wide range of non-specific symptoms which are often attributed to more prevalent causes. This makes diagnosis difficult without identifying a primary source of the malignancy. Our case stresses the importance of timely diagnostic imaging as well as laboratory testing in order to detect the presence of these lesions. Whenever a patient with symptoms of hypopituitarism is encountered, pituitary metastasis should be considered.