Amniotic fluid embolism (AFE) is a rare event in pregnancy, especially after first and second trimester abortions. Co-existence of a fetus and a molar pregnancy is also rare with few isolated case reports.
A 33-year-old G2P1001 at 13w0d (by LMP) with a twin pregnancy complicated by a molar component presented for management with suction dilation and evacuation (D&E). The patient was asymptomatic until the day of scheduled surgery. In the operating room the patient suddenly became hypotensive, tachycardic, hypoxic, and pulseless. The patient was intubated, resuscitative measures began, and bedside transesophageal echocardiogram (TEE) showed massive pulmonary embolism (PE). The patient was taken to the ICU and started persistently bleeding from the vagina and IV sites. Lab work was consistent with severe DIC. Massive transfusion protocol was started with IVC filter placement and bilateral uterine artery embolization. The patient remained hemodynamically unstable and underwent a total abdominal hysterectomy and bilateral salpingectomy. The patient successfully recovered, and was discharged in stable condition.
This case demonstrates the rare occurrence of AFE in early pregnancy with a co-existent partial molar pregnancy. This case shows the importance of recognizing the diverse presentations and sequelae of molar pregnancies, as patients can develop life-threatening complications such as AFE and DIC. The most important aspect to survival of patients with DIC and AFE is treatment with a multidisciplinary team approach.
Amniotic fluid embolism, Disseminated intravascular coagulation, Molar pregnancy
Disseminated intravascular coagulation (DIC) is a disruption of hemostasis and imbalance between thrombosis and thrombolysis. DIC can result in life-threatening bleeding, clot formation, and multi-system end organ damage [1]. Although the prevalence of DIC in pregnancy is low, ranging from 0.03-0.35%, the prevalence increases in pregnancies complicated by comorbidities such as placental abruption, postpartum hemorrhage, pre-eclampsia, hemolysis elevated liver enzymes and low platelets (HELLP syndrome) and pregnancies complicated by amniotic fluid embolism syndrome (AFES) [2]. Just as there are numerous comorbidities associated with DIC, patients with DIC can present with varying symptoms. Some patients present with severe vaginal bleeding, occult intrauterine or abdominal bleeding, or oozing from IV sites and/or mucosal surfaces while others present with signs of shock or multiple organ dysfunction. Laboratory findings suggestive of DIC include prolonged coagulation times, reduced fibrinogen, and thrombocytopenia. Labs should be correlated with clinical findings as they are not specific to DIC and often suggest other pregnancy-related complications mimicking DIC [1]. Management of DIC in pregnancy constitutes immediate treatment, which involves a multidisciplinary team based approach [2]. Surgery should not be delayed in an emergency, especially when initial resuscitative measures fail. This often requires a hysterectomy to conserve blood loss.
Amniotic fluid embolism syndrome (AFES) is another complication of pregnancy. 80% of individuals with AFE develop DIC [3]. AFES occurs when there is a breach between maternal and fetal compartments, resulting in amniotic fluid entrance into maternal circulation. This can result in death [4]. Entrance into maternal circulation can occur through uterine trauma (miscarriage, abortion, amniocentesis), endocervical veins, or site of placental tissue. AFES is a rare and unpredictable event, with an incidence of approximately 1 in 40,000 deliveries and a reported mortality rate ranging from 20% to 60% [3,5]. If AFE occurs after a second-trimester abortion, the mortality rate is as high as 80% [6]. The true incidence of AFES is difficult to determine, because it is a diagnosis of exclusion and is often underreported in nonfatal cases [4]. A case report showed that AFES was diagnosed by transesophageal echocardiogram showing pulmonary vasoconstriction at time of caesarian delivery [7]. Although multiple medical comorbidities can coincide with AFES, there is no clear etiology. Risk factors for AFES include age older than 35, cesarean delivery, operative vaginal delivery, placenta previa or abruption, eclampsia, fetal distress, African American race and other minority ethnic groups [4,8]. AFES can cause cardiogenic shock, respiratory failure, DIC, coma, seizures, or an inflammatory response [3]. These symptoms usually occur during labor and delivery or immediately postpartum. It is rare for AFES to occur beyond 48 hours postpartum or following a first or second trimester abortion. In some cases, clinical symptoms are separated into two phases: Acute respiratory failure and cardiac arrest, followed by severe hemorrhagic shock with DIC [3]. Treatment of AFES is supportive, involving immediate tracheal intubation with ventilatory assistance and cardiopulmonary resuscitation [2].
Yet another potential complication of pregnancy is a molar pregnancy. A molar pregnancy is part of a group of diseases referred to as gestational trophoblastic disease. Molar pregnancies have the potential to invade the uterus or metastasize. There are two types of molar pregnancies: Complete and partial. They differ by morphology, histopathology, karyotype, and their associated risk of malignancy. In the United States, hydatidiform moles are observed in approximately 1 in 600 therapeutic abortions and 1 in 1,500 pregnancies [9]. Molar pregnancies are usually diagnosed in the first trimester. Common presenting symptoms are abnormal bleeding, uterine enlargement greater than expected for gestational age, absent fetal heart tones, cystic enlargement of the ovaries, hyperemesis gravidarum, and an abnormally high level of HCG for gestational age [9]. Work-up for suspected molar pregnancy involves labs (CBC, coagulation factors, liver and renal function tests, type and screen, HCG level) in addition to pre-evacuation chest x-ray to test for metastatic disease. Complications of hydatidiform moles occur in 25% of patients with uterine enlargement of more than 14-16 weeks' gestational size, including anemia, infection, hyperthyroidism, and coagulopathy [9]. Pulmonary complications and respiratory distress syndrome are frequently observed near the time of molar evacuation among patients with marked uterine enlargement [9,10].
Co-existence of a fetus and a molar pregnancy is rare, occurring in 1 in 22,000-100,000 pregnancies. This co-existence can lead to hyperthyroidism, hemorrhage, and pregnancy-induced hypertension [9]. Additionally, twin pregnancies with a fetus and a molar pregnancy carry an increased risk for post-molar gestational trophoblastic disease, with a higher number of patients having metastatic disease and requiring multi-agent chemotherapy for treatment [11]. The preferred method of evacuation of a molar pregnancy is dilation suction curettage (DCE) rather than medical induction of labor or hysterectomy. The latter increases blood loss and may increase the risk for malignant sequela [9]. The risk of post-molar gestational trophoblastic disease after hysterectomy remains approximately 3-5%, thus patients should be monitored postoperatively with serial HCG measurements. Patients with prior molar pregnancies have a 10-fold increased risk (1-2% incidence) of a second hydatidiform mole in a subsequent pregnancy [12,13].
This case shows the rare incidence of AFE in early pregnancy complicated by the existence of a partial molar pregnancy, and the importance of early recognition and multidisciplinary team approach for the treatment of AFE and DIC.
A 33-year-old G2P1001 at 13w0d (by LMP) with past medical history of asthma presented to triage 3 days after arrival from Sierra Leone for follow-up management of a twin pregnancy complicated by a suspected molar pregnancy. Patient had no abdominal pain or vaginal bleeding on initial presentation and stated that her ultrasound in Sierra Leone showed a heartbeat of one fetus and an adjacent molar pregnancy (ultrasound was not available for review). She had stable vitals and repeat ultrasound showed a single intrauterine pregnancy without fetal heart tone and an adjacent echogenic structure suspicious of molar pregnancy (Figure 1). Beta HCG was 36,263 and Hgb was 10.4 g/dL. No additional studies were performed and the patient was counseled to follow-up for outpatient dilation evacuation and curettage (DCE) and was discharged in stable condition.
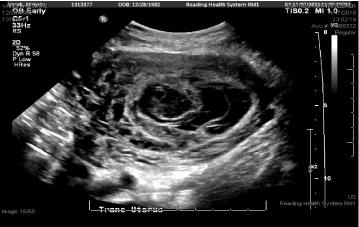 Figure 1: L Partial molar pregnancy. View Figure 1
Figure 1: L Partial molar pregnancy. View Figure 1
She presented the next day to sign surgical consents and was scheduled for D&E to be performed 2 days later; she again did not have any vaginal bleeding or symptoms and vitals remained stable. Prior surgeries included one cesarean section for placenta previa. On the day of surgery, patient had a CBC and type and screen prior to the procedure. In the operating room, she was placed under MAC anesthesia and the table was placed in slight Trendelenburg. Immediately after pelvic examination of a 14-16 week sized uterus, prior to the start of the procedure, the patient became hypotensive with SBP of 40s, tachycardic to 180s, hypoxic, then pulseless for 5 minutes. The procedure was immediately terminated and a code was called. Anesthesia staff rushed into the room and the patient was immediately intubated, given phenylephrine, vasopressin, and epinephrine; central venous access and a femoral arterial line were placed. A bedside transesophageal echocardiogram (TEE) was performed, showing massive pulmonary embolism (PE) with clot traveling from the IVC to the right atrium and right ventricle through the pulmonary artery (Figure 2). The patient was taken to the ICU, and upon arrival, she started to have persistent vaginal bleeding and oozing from her IV sites. Lab work drawn in the ICU was consistent with severe DIC (fibrinogen < 50 mg/dL, PT > 140 sec, PTT > 240 sec, Platelets 114 E/nL, D-dimer > 100,000 ng/ml). With massive transfusion protocol started, she was quickly taken to interventional radiology where an IVC filter was placed (anti-coagulation for PE could not be started due to persistent bleeding) and bilateral uterine artery embolization performed (Figure 3). However, due to persistent vaginal bleeding, decision was made to proceed with emergent total abdominal hysterectomy with assistance from the gynecology oncology team. In the operating room, after evacuation of the molar pregnancy via hysterectomy, coagulation factors began to normalize with intra-op fibrinogen of 179 mg/dL and PTT of 28 sec. The patient received a total of 8 unit's pRBCs, 8 units FFP, 2 units cryoprecipitate, and 1 unit of platelets, with resolution of her DIC. The patient recovered appropriately during her hospital stay and was discharged on hospital day 6 in stable condition with IVC filter and anti-coagulation with Xarelto. Her IVC filter was removed 6 weeks after surgery and Xarelto anticoagulation was to continue for a total of 6 months. Partial molar pregnancy was confirmed by FISH testing, showing XXY karyotype. Pathology results showed a likely partial molar pregnancy due to the presence of hydropic chorionic villi, confirmed a placenta accreta, and confirmed the delivery of a severely macerated fetus of 8 weeks size with no evidence of external congenital anomalies. The beta HCG values normalized 2 months after surgery.
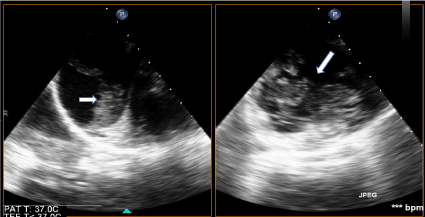 Figure 2: Intraoperative Transesophageal Echocardiogram with Emboli. View Figure 2
Figure 2: Intraoperative Transesophageal Echocardiogram with Emboli. View Figure 2
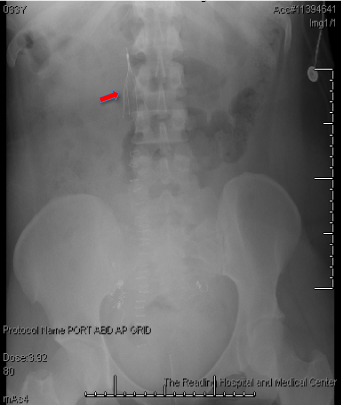 Figure 3: IVC filter placement. View Figure 3
Figure 3: IVC filter placement. View Figure 3
Co-existence of a fetus and a molar pregnancy is rare with isolated case reports showing partial moles and twin gestations. This case shows the rare instance of a partial molar pregnancy. This was not only suggested by the first trimester ultrasound findings showing an intrauterine pregnancy with a mixed echogenic focus, but also an enlarged uterine size (14-16 weeks) greater than gestational age (13w0d by LMP and 9w6d by ultrasound). Nonetheless, it is important to consider that the enlarged uterine size may have been due to the suspected twin pregnancy. Although the most common presenting symptom of a molar pregnancy is vaginal bleeding, our patient did not present with vaginal bleeding during any visit, including on the day of surgery. She only developed vaginal bleeding and oozing from IV sites in the ICU, at which time she had already progressed to DIC. It is important to be aware of the various presentations of molar pregnancies as well as their adverse sequela in order to treat patients immediately and appropriately to maximize their survival, especially in unexpected cases of DIC and AFES.
The triad of AFES is suggested to be hypotension, hypoxemia with respiratory failure, and DIC [3,12]. 80% of cases of AFES result in DIC [3]. DIC associated with AFES typically progresses faster than DIC associated with other medical comorbidities in pregnancy [14]. Perhaps it was the positioning of the patient in Trendelenburg and subsequent uterine manipulation with pelvic examination that caused the disruption between maternal and fetal compartments, leading to suspected amniotic fluid emboli in the maternal circulation, accounting for the massive PE evident on bedside TEE. This led to the initial hemodynamic instability with hypotension, tachycardia, hypoxemia, acute pulselessness, and thrombosis. This case illustrates the rare presentation of AFE that occurs in two clinical phases: Initially, our patient was in acute respiratory failure and pulseless, and, once surviving this initial phase, she progressed to the second phase of severe hemorrhagic shock with DIC. It is important to consider the patient's prior cesarean section for placenta previa, which resulted in her current placenta accreta (confirmed by pathology specimen). Placenta previa, African American race, and close proximity to 35 years of age increased this patient's risk for AFES. As mentioned previously, this patient did not show symptoms of DIC prior to the scheduled D&E, and only developed persistent bleeding after arrival to the ICU. This case demonstrates that obstetrician-gynecologists should be aware of both AFE and rapidly developing DIC in a patient with a first trimester molar pregnancy during the time of D&E. While AFES typically presents during labor and delivery or in the immediate postpartum period [8], our case shows its unique presentation in the antepartum period and its delayed occurrence after a missed abortion.
While, in retrospect, we suspect AFE as the culprit for this patient's instability, the differential should also include the possibility of the more common occurrence of thromboembolism during pregnancy. Pregnant women have a fourfold to fivefold increased risk of thromboembolism compared with nonpregnant women [15-17]. The increased risk of thromboembolism during pregnancy occurs due to the numerous physiologic changes in pregnancy including hypercoagulability, increased venous stasis, decreased venous outflow, compression of the inferior vena cava and pelvic veins by the enlarging uterus, increased coagulation factors favoring a thrombogenic state, and decreased mobility [17]. Perhaps the initial pelvic examination in the OR dislodged the enlarged, retroverted, impacted uterus, releasing potential pre-existing clots from the inferior vena cava (IVC). This could certainly have resulted in the observed emboli in the right ventricle and massive PE seen on TEE. In addition to the physiologic changes in pregnancy, thromboembolism could have also accounted for this patient's instability due to recent prolonged travel and surgery. While the most important individual risk factor for venous thromboembolism in pregnancy is a personal history of thrombosis, the second most important predisposing factor is a history of thrombophilia [17]. Our patient did not have a personal or family history of either of these individual risk factors.
Initial therapy for a first trimester molar pregnancy is to treat with D&E. Our case suggests that initial therapy may fail and the patient may require a hysterectomy. Perhaps the D&E could have successfully evacuated the mole had it been performed earlier (it was scheduled 4 days after initial evaluation), but, there is no evidence to suggest that the same complications would not have occurred with an earlier procedure. Perhaps a D&E should be performed at the time of diagnosis of a molar pregnancy, even when the patient is hemodynamically stable, to avoid potential adverse complications as was seen in this case. In managing a molar pregnancy, it is essential to collect specific labs and imaging, including a CBC, coagulation factors, liver and renal function studies, type and screen, beta HCG level and chest x-ray. This work up provides a baseline for comparison to post-treatment outcomes, and, it allows for monitoring of early signs of DIC and metastatic disease. All patients with molar pregnancies should be followed closely with beta HCG levels until reaching baseline values (< 5 mIU/mL) for at least 6 months to ensure no progression or development of post-molar gestational trophoblastic disease [9]. While it has been reported that the risk of post-molar gestational trophoblastic disease after hysterectomy remains approximately 3-5% [9], our patient's beta HCG levels normalized to less than five by two months after surgery, and they remain < 1 with no evidence of recurrence of disease.
The rare co-existence of a partial molar pregnancy with fetal parts has to be considered. It is important to recognize the diverse presentations of molar pregnancies and their adverse sequela, particularly because patients who are initially hemodynamically stable can quickly progress to instability and develop life-threatening complications. Specifically, 25% of patients with molar pregnancies and uterine enlargement more than 14-16 weeks size develop subsequent medical complications [9] such as AFE and DIC. AFE often leads to mortality and can occur in two distinct phases: Respiratory failure and cardiac arrest followed by severe hemorrhagic shock. AFE usually occurs intrapartum or postpartum, but this case illustrates it has to be recognized in early pregnancy and during the time of D&E for missed abortion. It is essential to have a high clinical suspicion of AFE, because, with early recognition, and an appropriate multidisciplinary team approach (as was done in our institution with anesthesia, obstetrics-gynecology, gynecology oncology, interventional radiology, and intensivist), it is possible to save the life of a patient.
This work and manuscript preparation was unfunded.
There are no conflicts of interest.