This review article explores the association between atrial flutter and systemic lupus erythematosus (SLE). Atrial flutter, a common cardiac arrhythmia, has been recognized as a potential cardiovascular manifestation of SLE, a chronic autoimmune disease. The abstract summarizes the current literature on the epidemiology, pathophysiology, clinical presentation, diagnosis, and management of atrial flutter in patients with SLE. Furthermore, it highlights the challenges in diagnosing and treating this specific arrhythmia in the context of SLE and discusses potential underlying mechanisms contributing to its occurrence. By shedding light on this relationship, the review aims to enhance the understanding of atrial flutters in SLE and facilitate improved patient care and outcomes.
Systemic Lupus Erythematosus (SLE or Lupus) is a heterogeneous autoimmune disease with many clinical manifestations, such as rash, arthritis, and fatigue. SLE can affect the cardiovascular system in more than 50% of patients [1]. SLE can affect the cardiac vasculature, valves, myocardium, pericardium, and conduction system, leading to many cardiovascular manifestations that can remain clinically silent or lead to substantial cardiovascular morbidity and mortality. The conduction system is affected in around 10% of patients [2]. Conduction disturbances reported include atrioventricular block (first, second, or third degree), bundle branch block, sinus tachycardia, atrial premature contractions, and atrial fibrillation [1]. One meta-analysis suggested a significantly increased atrial fibrillation risk in SLE patients [3]. Pathophysiology of cardiac involvement in patients with SLE includes immune-mediated damage, atherosclerotic complications, or even the adverse effect of the treatment (chloroquine-induced cardiotoxicity). Some antibodies, such as anti-Ro/SS-A, target cardiomyocytes and can cause myocarditis and conduction abnormalities [4]. The prevalence of atrial flutter has not been mentioned much in the literature. Diagnosis of SLE requires a high index of suspicion because patients can present with isolated complaints or infrequent disease characteristics like our patient and can represent more of a diagnostic challenge.
A 65-year-old female presented to our hospital in 2021 with a few days of intermittent chest pain and shortness of breath. She had a past medical history of SLE. Her last lupus flare-up was ten years back.
She had been stable on Mycophenolate and Prednisolone. She had never had cardiac or pulmonary symptoms before. Physical examination did not show any significant findings. Upon presentation, she was found to be in atrial flutter. She was started on continuous Diltiazem infusion, which controlled her heart rate slightly. She was admitted to our cardiovascular unit for further management (Figure 1).
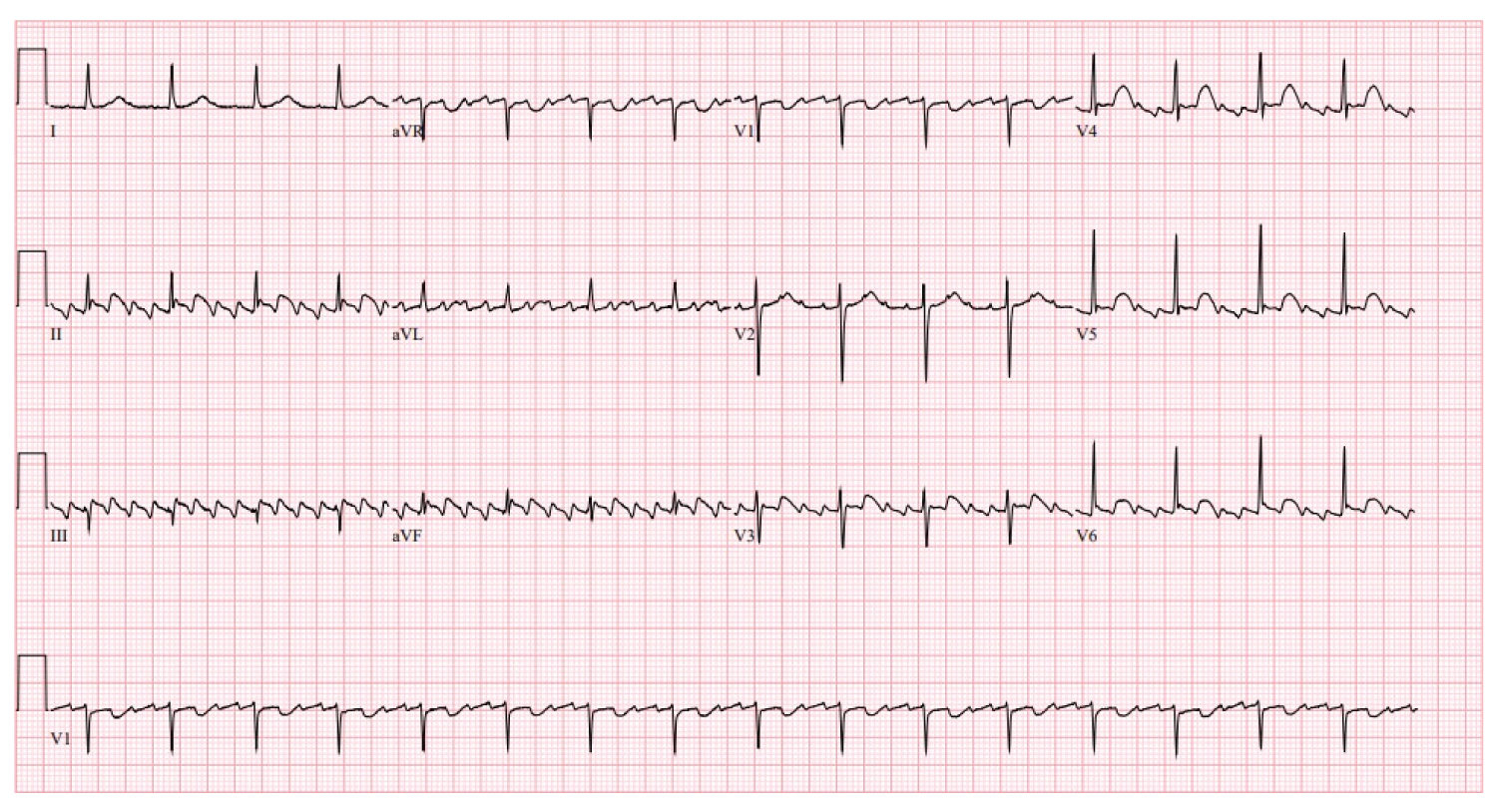 Figure 1: Initial EKG shows atrial flutter with 4:1 conduction and a heart rate of around 100/min.
View Figure 1
Figure 1: Initial EKG shows atrial flutter with 4:1 conduction and a heart rate of around 100/min.
View Figure 1
Initial labs (blood count, chemistries, thyroid function, urinalysis) showed no abnormality. Transthoracic echo showed no structural or valvular abnormalities other than AFib, with normal RV/LV size/function with normal right/Left atrium size. CT angiography of the chest showed no evidence of pulmonary embolism or focal lung consolidation.
She remained hemodynamically stable. Despite trials of rate control medications, she remained in persistent A-flutter. The cardiologist made plans for a transoesophageal echocardiogram with electrical cardioversion. However, the CT abdomen on day 3 showed pericardial effusion and increasing bilateral pleural effusions, raising concerns for lupus flare as the etiology. Lab work showed high dsDNA and low C3/C4 levels supporting lupus flare. She was started on high-dose steroids (Solu-Medrol 125 mg IV Q6 hr).
She converted to sinus rhythm the next day, and the planned cardioversion was canceled. Figure 2 shows EKG after IV steroid administration.
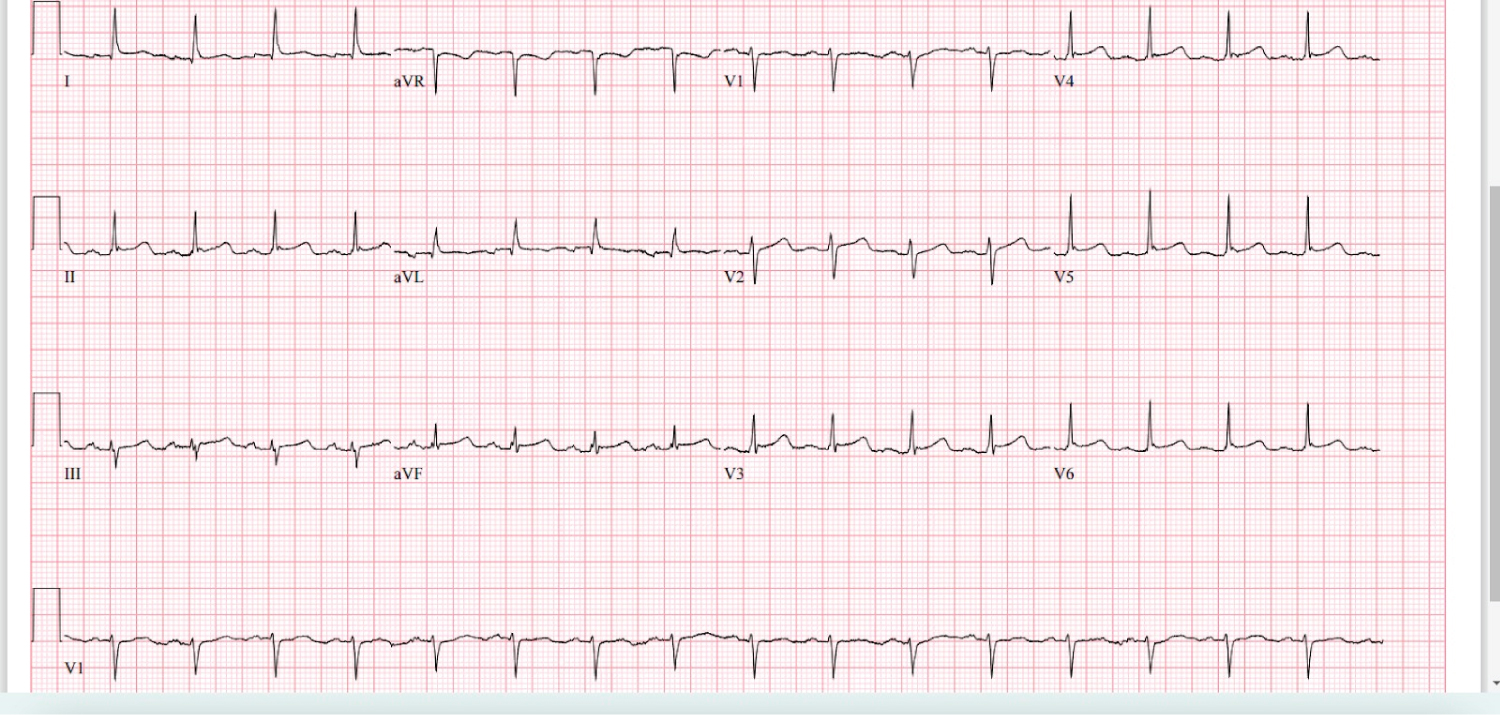 Figure 2: EKG after IV steroid administration.
View Figure 2
Figure 2: EKG after IV steroid administration.
View Figure 2
She received three days of high-dose IV steroids with improvement in symptoms and maintenance of sinus rhythm. She was discharged on hospital day 6 with oral steroids, direct-acting oral anticoagulation, oral beta-blockade, and follow-up with cardiology and rheumatology.
SLE is a multisystem autoimmune inflammatory disorder. It predominantly affects women of reproductive age. Around 50% of patients with SLE have a risk of cardiac diseases during their lifetime [1]. Multiple factors play a role in these patients' pathogenesis of cardiac involvement. SLE may contribute to cardiac remodeling via chronic inflammation. High levels of inflammatory markers in SLE can lead to structural and electrical remodeling. As a result of this remodeling, arrhythmias like Atrial Fibrillation or Atrial Flutter may develop [3]. According to PM Seferović, et al. malignant ventricular arrhythmias are rarely reported in systemic lupus erythematosus (SLE). Sinus tachycardia, atrial fibrillation/Atrial flutter, and atrial ectopic beats are most frequent. Supraventricular arrhythmias are often transient and may be related to myocarditis and pericarditis. Sinus tachycardia occurs in 50% of patients and may be the only cardiac manifestation of SLE [5,6].
The development of arrhythmias in SLE could be secondary to pericarditis, myocarditis, or ischemia caused by coronary vasculitis. Pericarditis is the most common cardiac manifestation of systemic lupus erythematosus. Myocarditis may also be suspected in patients presenting with unexplained tachycardia, conduction disturbances, unexplained systolic dysfunction with or without heart failure, or arrhythmias [7]. Doria A, et al. reported that it is still controversial whether the arrhythmias in acute pericarditis are of myocardial or pericardial origin [8,9]. An endomyocardial biopsy is needed to make an accurate diagnosis. Thus, our patient who presented with acute SLE flare-up might have had myocarditis in addition to the pericarditis, which could be the possible cause of the Aflutter/Afib.
Intravenous calcium channel-blocking agents (verapamil or diltiazem) or intravenous beta-blocking agents (esmolol or metoprolol) are effective in rapidly controlling ventricular response [10]. Antiarrhythmic drugs such as ibutilide, flecainide, and propafenone can be used to convert atrial flutter to sinus rhythm [10]. Radiofrequency catheter ablation has a 95% cure rate in patients with atrial flutter, which makes this treatment of choice for most patients [10].
According to Dein Eric, et al. mild pericarditis in an SLE flare can be treated with intramuscular triamcinolone injection or oral methylprednisolone. Severe pericarditis or pericardial tamponade should be treated with an intravenous bolus of methylprednisolone (initial dose usually 1 gram for three days) [11]. Our patient was treated with IV Solu-Medrol 125 mg with significant improvement of her clinical condition and conversion to normal sinus rhythm after no spontaneous conversion while on rate control medications.
Studies regarding the use of systemic anticoagulation in a case of Aflutter/AFib secondary to an autoimmune process are limited. The A-flutter in our patient appeared to be caused solely by the SLE flare (a reversible process infrequently occurring in her case). After a discussion with the patient concerning her CHA2DS2 VASC score of 2 and uncertain risk of recurrence, we opted to continue oral anticoagulation. At the same time, she is being monitored outpatient by cardiology and rheumatology.
In this exciting case of A-flutter due to a lupus flare-up, we successfully treated the arrhythmia with high-dose steroids. But we cannot recommend how long the patient should be on a rate control medication and anticoagulant once the lupus flare-up has resolved.
Further research is required on chronic anticoagulant and rate control medication use in patients with SLE-induced arrhythmia or arrhythmia caused by reversible conditions. In addition, it also gives an insight into the prompt recognition of cardiovascular abnormalities needed for timely and appropriate management in an SLE flare. It emphasizes that therapies directed toward the inflammatory process are crucial in reducing cardiovascular disease morbidity and mortality.
Informed consent was obtained from the patient according to the established instructions of the University of Maryland Capital Region Health for research studies.
None of the authors have any conflict of interest to declare in any form.