Dengue is an arthropod-borne viral infection endemic to tropical areas. Dengue myocarditis is a rare cardiac manifestation of arrhythmic syncope; however, the pathophysiology behind it is not yet clearly elucidated. Severe electrolyte imbalance is among the common causes that aggravate cardiac dysrhythmia. One disease entity that presents with salt-losing tubulopathy is Gitelman syndrome.
A 27-year old female with gitelman syndrome in the convalescent phase of dengue infection, presents with recurrent loss of consciousness. Electrocardiogram showed sinus rhythm with prolonged corrected QT of 0.54 seconds which converted to Torsades de pointes. There was elevation of cardiac enzymes, and transthoracic echocardiogram (TTE) showed a reduction of ejection fraction (EF) from 61.6% to 41.9% with new onset LV global hypokinesia. She was given magnesium sulfate, but due to frequent recurrence of ventricular tachycardia (VT), an overdrive pacing was inserted and started on propranolol. She was started on heart failure regimen and monitored closely for recurrence of syncope that may warrant insertion of implantable cardioverter defibrillator (ICD).
Syncope, Ventricular arrhythmia, Pacemaker therapy, Implantable cardioverter defibrillator, Myocarditis
Dengue is an arthropod-borne viral infection caused by any of the four serotypes of dengue virus transmitted by the mosquito species Aedes aegypti and Aedes albopictus that are endemic to tropical countries [1]. Based on the Centers for Disease Control and Prevention (CDC), dengue classification is differentiated based on the presence of warning signs like abdominal pain, persistent vomiting, mucosal bleeding and others, the presence of a warning sign may predict severe dengue. The severity of dengue is based on severe plasma leakage, severe bleeding, or severe organ impairment. Dengue is divided into three phases, namely the febrile phase that lasts 27 days followed by critical phase typically lasts 24-48 hours wherein defervescence starts and plasma leakage subsides. Lastly, there will be the reabsorption of extravasated intravenous fluids and patients begin to improve and this is known as convalescent phase. Dengue causes multiorgan involvement specifically hematologic, cardiac, hepatic, and neurological. The cardiovascular manifestations of dengue are uncommon and almost exclusively associated with severe disease. The associated symptoms are bradyarrhythmia, left ventricular systolic dysfunction, pericardial effusion, myocarditis, and atrial fibrillation [2]. A retrospective observational study by Abdul showed that myocarditis has a prevalence of 4.2% in hospitalized dengue-infected patients wherein 59.5% had electrocardiography change and 24% had reduced ejection fraction. Electrocardiogram (ECG) abnormalities can be frequently encountered in the febrile phase of dengue that includes a variety of arrhythmias such as heart block, ectopic beats and incessant tachyarrhythmias [3]. The usual presentation of myocarditis is chest pain, fever, and dyspnea. Atypical presentation includes arrhythmia that may present as syncope, as seen in our patient’s case [4,5]. Arrhythmic syncope can be due to bradyarrhythmia or tachyarrhythmia associated with structural heart disease, channelopathies or inherited arrhythmia syndromes [6]. Severe electrolyte imbalance is among the common causes that aggravate cardiac dysrhythmia. One disease entity that presents with salt-losing tubulopathy is Gitelman syndrome [7].
A 27-year-old female with Gitelman syndrome was initially admitted due to a three-day history of high grade fever, abdominal pain and loose stool. The patient was managed as a case of dengue fever with warning signs. She had positive dengue nonstructural protein 1 (NS1) antigen, thrombocytopenia of 29,000, severe transaminitis of 85x the upper limit of normal (ULN), increasing creatinine trends with glomerular filtration rate (GFR) of 19 and persistent multiple electrolyte imbalance showing hypokalemia, hypomagnesemia and hypocalcemia. Supportive management with hydration, continuous electrolyte correction, and antipyretics were given. The patient was clinically improving, however, during the convalescent phase of dengue; she developed recurrent loss of consciousness lasting for less than 30 seconds which was preceded by nausea, vomiting and palpitation. There were no seizure-like symptoms or any abnormal tone elicited. She had a history of febrile seizures during childhood. There was no history of epilepsy, transient loss of consciousness, or recent head trauma. She had no history of previous dengue infection. There was no history of sudden cardiac death, arrhythmia, or congenital disease among family members. She is a non-smoker, non-alcoholic beverage drinker, and has no history of illicit drug use. There was complete spontaneous recovery after regaining consciousness. She was awake, oriented to three spheres. She was hemodynamically stable with a blood pressure of 140/80, heart rate of 80 beats per minute (bpm), and respiratory rate of 22 cycles per minute (cpm). There was no desaturation, pallor or cyanosis. There was no carotid bruit. The jugular venous pressure was 8 mmHg above the sternal angle. On the cardiac examination, there were distinct S1 and S2 sounds, no heaves, lifts, thrills, murmur, or S3 gallop. Pulses were full and equal in all extremities with no bipedal edema. The ECG showed normal sinus rhythm with a corrected QT interval (using the Bazett formula) was 0.54 seconds (Figure 1A), hence was monitored closely. On the next hospital day, there was recurrence of syncope and ECG showed an "R-on-T" phenomenon initiated by a "short-long-short" sequence as represented by a premature ventricular complex (PVC) with a long compensatory pause followed by a sinus beat with prolonged QT interval which was subsequently followed by a closely coupled PVC which triggered the TdP (Figure 1B). She was given magnesium sulfate intravenously, eventually converting the rhythm to sinus. Other cardiac workups showed evidence of cardiac injury with high sensitivity Troponin I (HsTropI) of 18.20. Transthoracic echocardiogram (TTE) showed decreased systolic function with an ejection fraction (EF) of 41.9% from 61.6% with grade II left ventricular diastolic dysfunction (Figure 2). There was new wall motion abnormality described as left ventricular global hypokinesia with the best contraction in the apical inferior interventricular septum and basal inferolateral segment. The cranial CT scan showed normal results. She had decreasing trends of creatinine and transaminases, but there were persistent hypokalemia (2.7 mEq/L) and hypomagnesemia (1.3 mEq/L) and elevated inflammatory markers. Arterial blood gas showed normal acid base balance with adequate oxygenation, and thyroid function tests, complete blood count (CBC) and coagulation studies were normal. She was transferred to the critical care unit wherein she presented with chest tightness. There was frequent non-sustained ventricular tachycardia (NSVT) and recurrence of loss of consciousness, there was no pulse or blood pressure appreciated. Hence code blue was activated and ACLS was initiated. She was subsequently intubated with return of spontaneous circulation (ROSC) after 6 minutes of resuscitation. However, there was a recurrence of pulselessness and ECG showed pulseless VT. Defibrillation using a biphasic shock of 200J was done with conversion to sinus rhythm and ROSC. A 24 hour holter was requested and showed frequent non-sustained ventricular tachycardia with longest beat of 13 seconds and episodes of torsades de pointes (Figure 3). To prevent recurrence of cardiac dysrhythmia, a temporary transvenous pacemaker was inserted to promote overdrive pacing, with a back up rate set to 100 bpm preventing “R-on-T” phenomenon (Figure 4). Eventually started on propranolol, a non selective blocker to reduce risk of recurrent arrhythmia. According to the latest ESC guidelines for the management of patients with ventricular arrhythmia and prevention of sudden cardiac death, there is Class I recommendation in giving non-selective beta-blockers (nadolol or propranolol) in long QT syndrome (LQTS) patients with documented QT interval prolongation to reduce risk of arrhythmic events and initiating transvenous pacing to increase heart rate in patients with recurrent torsades de pointes [8]. Decision to insert implantable cardioverter defibrillator (ICD) was still controversial since recommendations to insert ICD are indicated in patients with syncope with depressed cardiac function, and ventricular tachycardia without correctable cause [9]. In the patient’s case, syncope was attributed to dengue myocarditis aggravated by electrolyte wasting secondary to Gitelman syndrome. Hence, regular electrolyte monitoring and aggressive correction was maximized to prevent further risk of cardiac dysrhythmia.
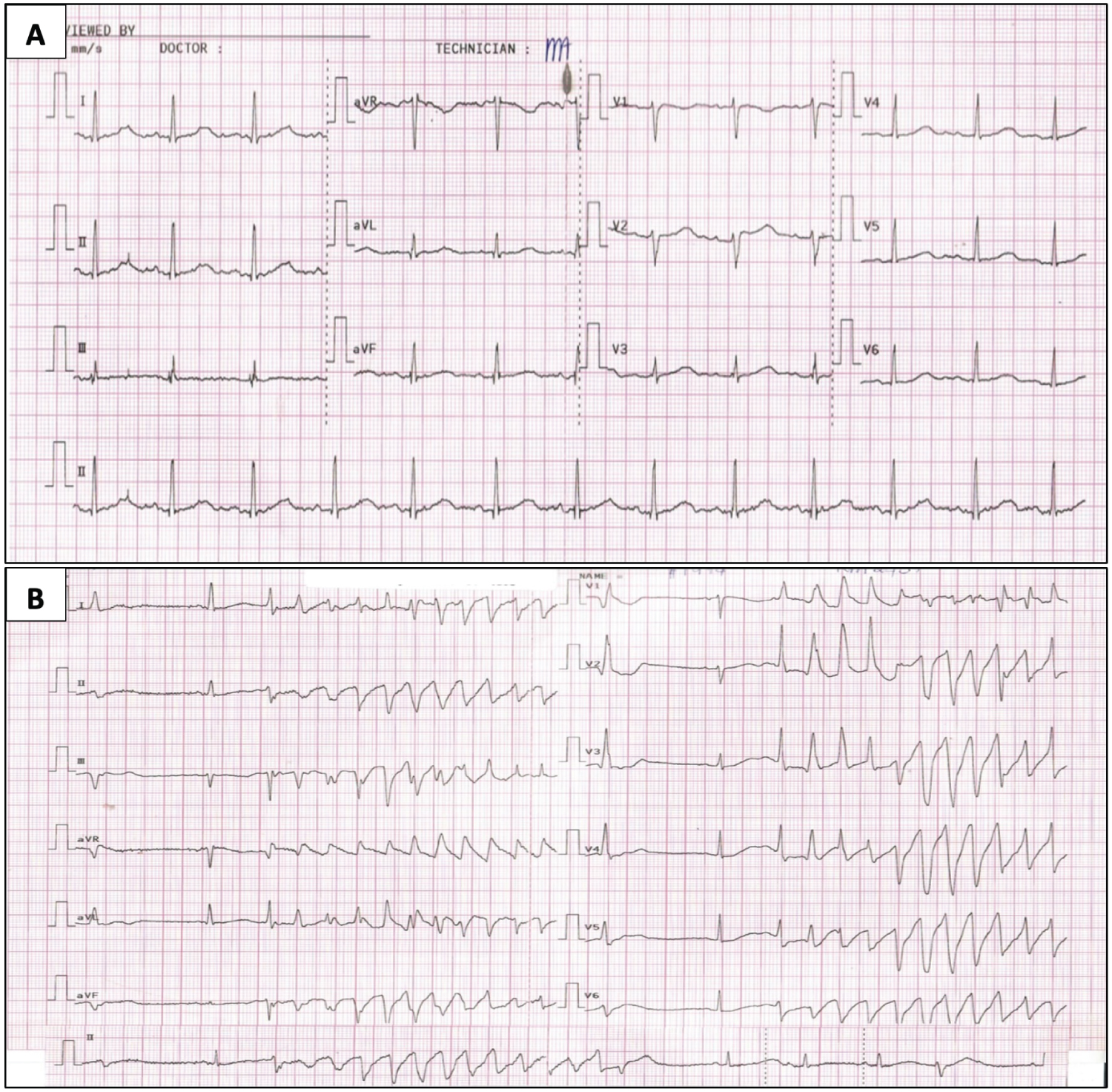 Figure 1: ECG after and during the loss of consciousness. A) ECG tracing showing prolonged corrected QTc (0.54 seconds) with U waves, taken after the patient regained consciousness; B) ECG tracing of torsades de pointes taken while the patient was unconscious.
View Figure 1
Figure 1: ECG after and during the loss of consciousness. A) ECG tracing showing prolonged corrected QTc (0.54 seconds) with U waves, taken after the patient regained consciousness; B) ECG tracing of torsades de pointes taken while the patient was unconscious.
View Figure 1
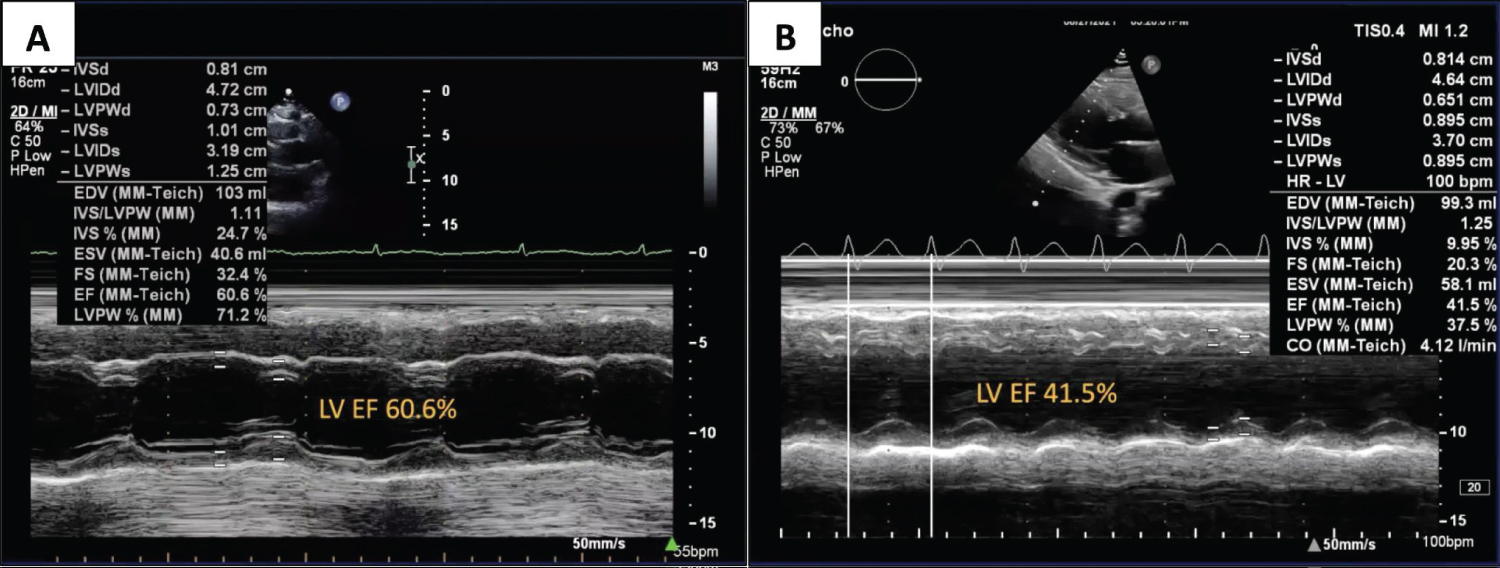 Figure 2: Transthoracic echocardiogram.
Figure 2: Transthoracic echocardiogram.
TTE in M-mode showing remarkable reduction in EF from 60.6% to 41.5% by Teichholz method.
View Figure 2
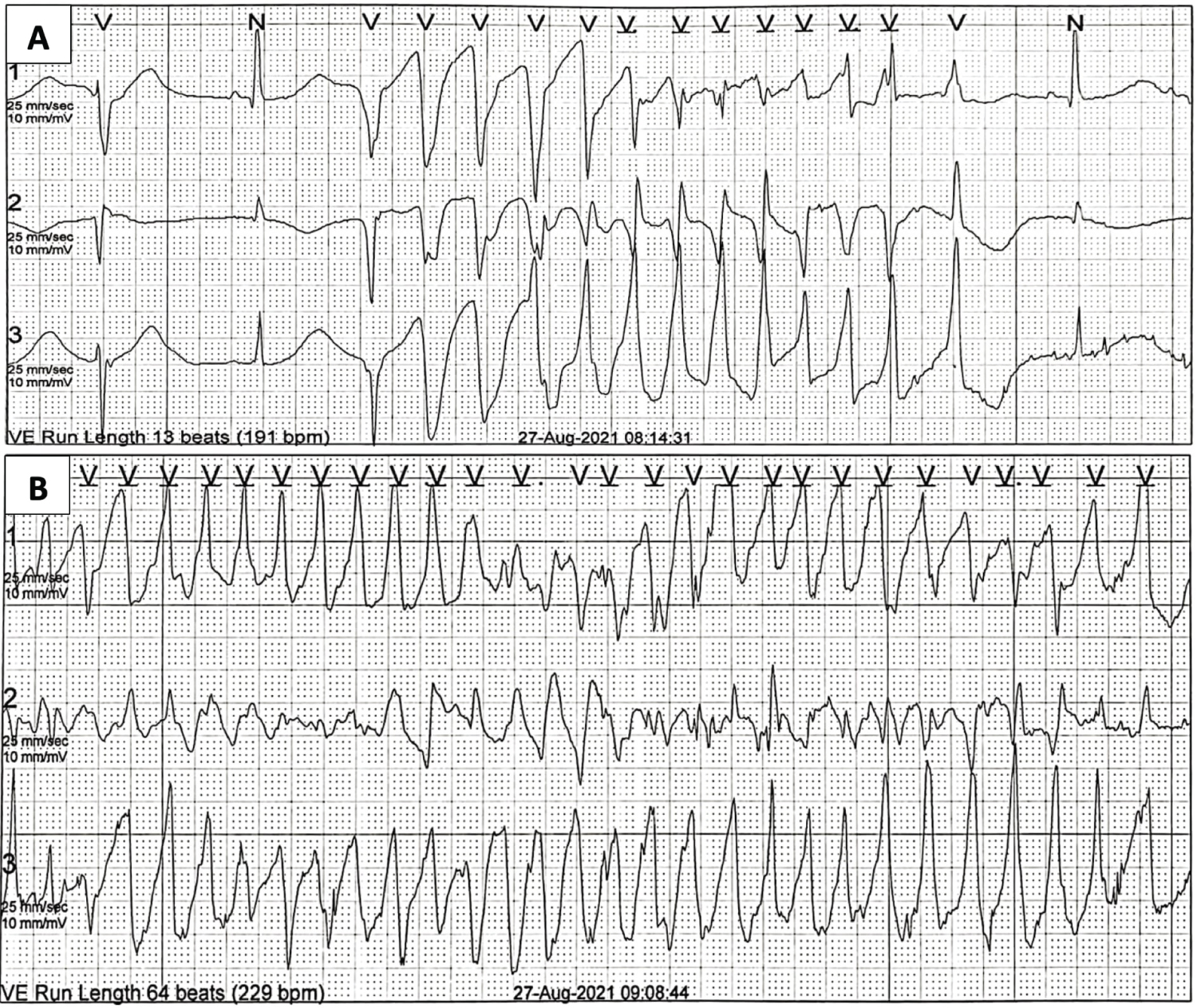 Figure 3: 24 Hour Holter at the Critical Care Unit. A) 13 beat run ventricular tachycardia at a rate of 191 beats per minute; B) Episode of torsades de pointes.
View Figure 3
Figure 3: 24 Hour Holter at the Critical Care Unit. A) 13 beat run ventricular tachycardia at a rate of 191 beats per minute; B) Episode of torsades de pointes.
View Figure 3
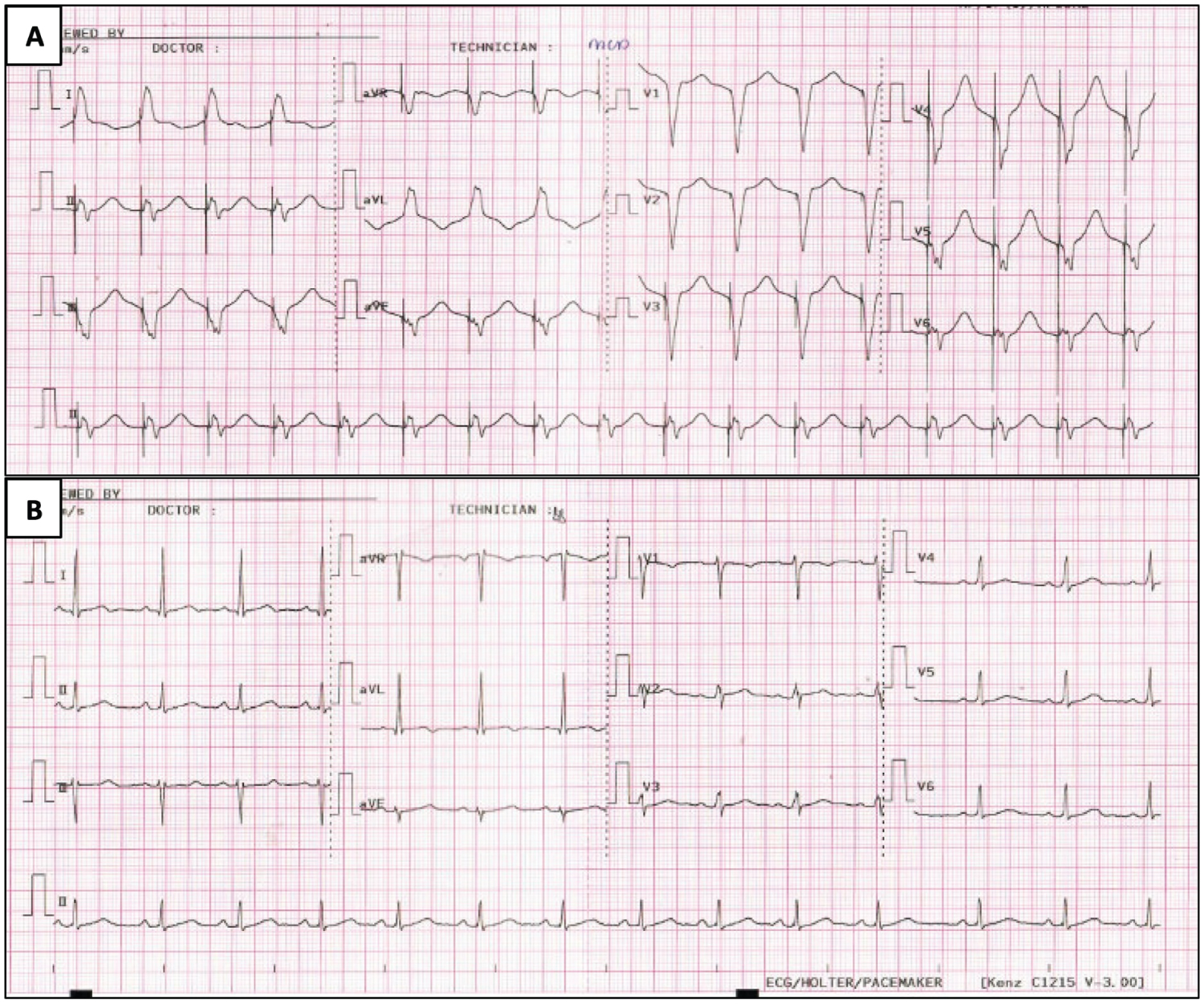 Figure 4: ECG prior removal of transvenous pacemaker and upon discharge. A) ECG showing ventricularly paced rhythm with good capture and sensing; B) ECG prior to discharge showing normal sinus rhythm with corrected QT of 0.44 seconds.
View Figure 4
Figure 4: ECG prior removal of transvenous pacemaker and upon discharge. A) ECG showing ventricularly paced rhythm with good capture and sensing; B) ECG prior to discharge showing normal sinus rhythm with corrected QT of 0.44 seconds.
View Figure 4
Since she presented with symptoms of heart failure with echocardiographic findings of ejection fraction of 41.9%, she was managed as heart failure with mildly reduced Ejection Fraction (HFmrEF) and started on guideline directed medical therapy (GDMT) specifically Sacubitril/Valsartan, Beta-blocker, Spironolactone and SGLT-2 inhibitor [10]. During the course of admission, she was weaned off from mechanical ventilator. Serial ECG monitoring showed a decreased QT interval (using the Bazett formula) was 0.44 seconds (Figure 4B) and the back-up rate of the transvenous catheter was gradually decreased and subsequently removed. There was also a decrease in HsTropI with a value of 1.5. Plans for ICD insertion were again raised but still wasn’t performed due to underlying risk. According to recent recommendations, in hemodynamically not-tolerated sustained VT in the acute phase of myocarditis, ICD insertion can be done prior to discharge (Class IIA) [9]. But since myocarditis may resolve without sequelae, risk of inserting an ICD is still a controversy. She was discharged with the pillars of heart failure regimen and oral correction of potassium, magnesium, and calcium. Beta blocker medication was continued as secondary prevention of ventricular arrhythmia, and to decrease incidence of sudden cardiac death. She was advised to follow up for reassessment if pacemaker or ICD insertion is still warranted and whether correctible factors have been resolved through medical management.
After two months, outpatient follow-up showed clinical improvement with no recurrence of syncope. Sacubitril/Valsartan, beta-blocker, SGLT-2 inhibitor and spironolactone were continued as part of the heart failure regimen. She followed up after one year with complete resolution of symptoms. TTE was repeated, showing improvement of EF to 65% from 41%. ECG showed normal sinus rhythm with a QT of 0.42 sec and Troponin T was < 50 ng/L. Repeat 24-h Holter likewise showed normal results. ARNI and MRA were discontinued, and she was maintained on beta-blocker and an SGLT-2 inhibitor.
In the recent consensus on myocarditis, there is 25% persistence of cardiac dysfunction and 21% may lead to dilated cardiomyopathy. Follow-up intervals of one to three months are recommended to monitor symptoms closely [11].
Dengue virus is a rapidly spreading mosquito endemic in tropical countries. Cardiac involvement has been reported in few studies. Dengue myocarditis is a rare manifestation of dengue fever, especially during the convalescent phase. Myocarditis presenting with acquired long QT syndrome leading to ventricular tachyarrhythmia is rare, and the proposed mechanism is due to altered intracellular calcium homeostasis resulting from cytokines and inflammatory processes affecting myocytes and the interstitium [12].
This can be aggravated by various causes, one of which is severe electrolyte imbalance that alters cardiac tissue excitability and conduction. A classic example is Gitelman syndrome - a rare, recessively inherited, salt-losing tubulopathy that causes hypokalemia and hypomagnesemia.
Fatal arrhythmia, such as torsades de pointes, a variant of PVTs, is a life threatening disease that can cause sudden cardiac death. Hence, immediate intervention is warranted. One of the well-established medical procedures to prevent fatal arrhythmia in patients with prolonged QT is the overdrive pacing preventing the “R-on-T” phenomenon. In addition, the initiation of a non-selective beta blocker like propranolol reduces the risk of arrhythmic events. There is a Level I recommendation in ICD insertion in LQTS with a history of cardiac arrest. Collaborative efforts of each subspecialty are needed to clearly weigh the benefit and the risk of inserting an ICD.
Myocarditis may also result in heart failure; hence, early initiation of the pillars of HF regimen is needed for better prognosis. In general, acute myocarditis usually resolves in the first 2-4 weeks of infection. However, there are some circumstances that may lead to persistent cardiac dysfunction. Close follow-up is warranted to determine whether continuation of HF regimen is needed and ICD insertion is necessary.
The authors have no conflicts of interest to declare.
The patient has provided written consent to the submission of the case report to the journal.
Data sharing is not applicable to these articles as no new data were created or analyzed in this study.
Institutional ethics approval was granted.