A 38-year-old male with previous surgeries for an incomplete atrioventricular septal defect successfully underwent percutaneous transcatheter tricuspid balloon valvuloplasty for a deteriorated bioprosthetic valve. Our case highlights the imperative of considering catheter-based minimally invasive therapy for the increasing number of adults with congenital heart disease who may present multiple risks factors for surgery.
Because of advancements in medical and surgical treatment, there has been a decline in mortality from congenital heart defects over the past several decades [1]. As a result, the number of patients with adult congenital heart disease has increased over time [2,3]. Most patients with adult congenital heart disease underwent several surgeries during childhood. In some cases, surgical or catheter interventional treatment in adulthood is recommended because of sequelae or remote-term complications [4,5]. In some cases, catheter intervention is preferable to surgery because the patient is considered a high-risk candidate. In our report, we describe the first case of percutaneous transcatheter bioprosthetic tricuspid balloon valvuloplasty for a patient with adult congenital heart disease.
Adult congenital heart disease, Structural valve deterioration, Percutaneous transcatheter tricuspid balloon valvuloplasty
A 38-year-old male presented with right-side heart failure, with symptoms that included dyspnea on exertion, leg edema, and pleural effusion. Soon after birth, he had been diagnosed with incomplete atrioventricular septal defect. At two years of age, a surgical primum atrial septal defect closure, mitral valve plasty, and tricuspid valve plasty were performed. Because he developed severe tricuspid regurgitation at the age of five, a tricuspid valve replacement with a mechanical prosthetic valve (SJM27 mm) was performed. At the age of 20, stuck-valve and severe tricuspid stenosis occurred, and he underwent a second tricuspid valve replacement with a bioprosthetic valve (CEP27 mm). During surgery, an expansive pannus was observed. At the age of 29, the patient ceased medical follow-up. On admission, his physical examination revealed moderate edema in both legs. A Levine Grade III/VI, diastolic rumble also was auscultated at the lower left sternal border. A transthoracic echocardiogram revealed severely calcified, thickened, and immobile leaflets of the bioprosthetic tricuspid valve (Figure 1A, Movie 1). The peak velocity was 2.5 m/sec with a mean diastolic gradient across the tricuspid valve of 14 mmHg (Figure 1B). Tricuspid regurgitation was mild. An invasive hemodynamic study by cardiac catheterization revealed 9.8 mmHg of mean transvalvular pressure gradient and mean right atrial pressure that had elevated to 17 mmHg. The tricuspid valve area was measured as 1.0 cm2 by Gorlin's equation (Figure 2A). He also had two major comorbidities. The first was hepatic cirrhosis due to the hepatitis B virus and a congestive liver; the second was a progressive post-mediastinum tumor, the size of which was 50 × 55 mm. His fourth sternotomy was considered a high-risk procedure. His hepatic function also was thought to be insufficient for highly-invasive surgery. Therefore, the patient was offered a percutaneous transcatheter tricuspid balloon valvuloplasty, a less-invasive procedure.
Movie 1: A transthoracic echocardiogram revealed severely calcified, thickened, and immobile leaflets of the bioprosthetic tricuspid valve.
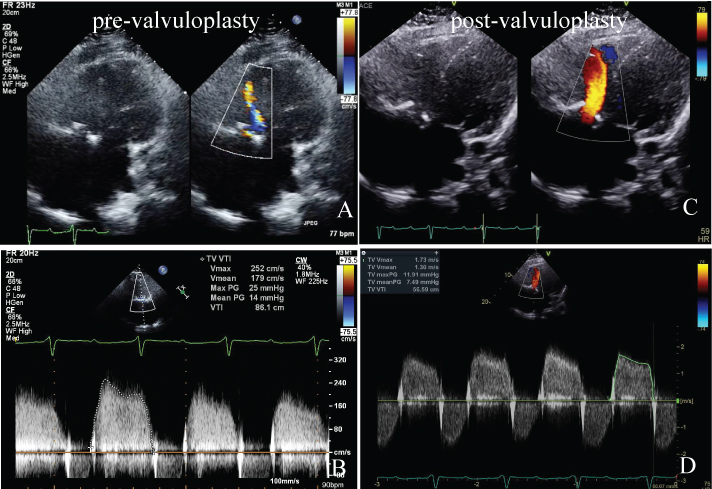 Figure 1: A) Before balloon valvuloplasty, a color Doppler image in the apical four-chamber view shows the narrow tricuspid inflow jet during diastole; B) Before the procedure, peak velocity was 2.5 m/sec and the mean pressure gradient across the tricuspid valve was 14 mmHg, suggesting tricuspid stenosis; C) After the procedure, tricuspid stenosis improved. The color jet of the tricuspid inflow widened. No significant tricuspid regurgitation occurred; D) After the procedure, peak velocity decreased from 2.5 m/sec to 1.7 m/sec and the mean pressure gradient across the tricuspid valve decreased from 14 mmHg to 7.5 mmHg.
View Figure 1
Figure 1: A) Before balloon valvuloplasty, a color Doppler image in the apical four-chamber view shows the narrow tricuspid inflow jet during diastole; B) Before the procedure, peak velocity was 2.5 m/sec and the mean pressure gradient across the tricuspid valve was 14 mmHg, suggesting tricuspid stenosis; C) After the procedure, tricuspid stenosis improved. The color jet of the tricuspid inflow widened. No significant tricuspid regurgitation occurred; D) After the procedure, peak velocity decreased from 2.5 m/sec to 1.7 m/sec and the mean pressure gradient across the tricuspid valve decreased from 14 mmHg to 7.5 mmHg.
View Figure 1
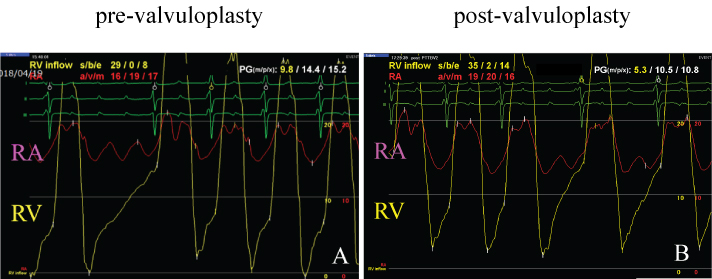 Figure 2: A) Cardiac catheterization revealed 9.8 mmHg of mean transvalvular pressure gradient and mean right atrial pressure that had elevated to 17 mmHg. The tricuspid valve area was measured as 1.0 cm2 by Gorlin's equation; B) After the procedure, mean transvalvular pressure gradient decreased to 5.3 mmHg, mean right atrial pressure was then reduced to 15 mmHg and the tricuspid valve area increased to 1.3 cm2.
View Figure 2
Figure 2: A) Cardiac catheterization revealed 9.8 mmHg of mean transvalvular pressure gradient and mean right atrial pressure that had elevated to 17 mmHg. The tricuspid valve area was measured as 1.0 cm2 by Gorlin's equation; B) After the procedure, mean transvalvular pressure gradient decreased to 5.3 mmHg, mean right atrial pressure was then reduced to 15 mmHg and the tricuspid valve area increased to 1.3 cm2.
View Figure 2
A 14-Fr long sheath was inserted via the right femoral vein and the guide wire passed through the bioprosthesis to the right pulmonary artery. A 26 mm Inoue balloon (Toray Medical Ltd., Japan) was inflated to a maximum of 26 mm, equivalent to 96% of the bioprosthetic valve (Figure 3A and Figure 3B). After the procedure, mean transvalvular pressure gradient decreased to 5.3 mmHg, mean right atrial pressure was then reduced to 15 mmHg and the tricuspid valve area increased to 1.3 cm2 (Figure 2B). The echocardiogram revealed that peak velocity had decreased to 1.7 m/sec, with a mean diastolic gradient of 7.5 mmHg (Figures 1C and Figure 1D, Movie 2). Deterioration of tricuspid regurgitation did not occur.
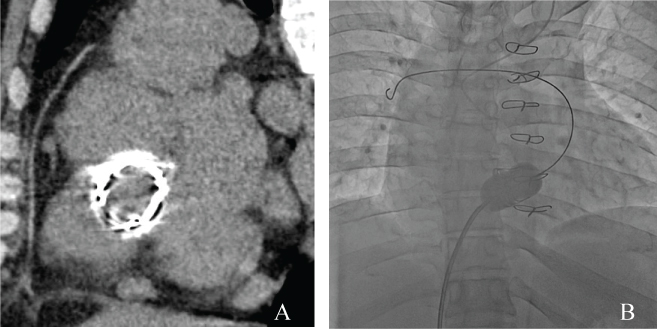 Figure 3: A) CT image before the valvuloplasty shows a highly calcified bioprosthetic valve in the tricuspid position; B) Fluoroscopic image in the anterior-posterior view shows the stiff guide wire passing through the center of the deteriorated bioprosthetic tricuspid valve, with the wire tip in the right pulmonary artery. A balloon valvuloplasty was performed using an Inoue balloon (maximum diameter 26 mm).
View Figure 3
Figure 3: A) CT image before the valvuloplasty shows a highly calcified bioprosthetic valve in the tricuspid position; B) Fluoroscopic image in the anterior-posterior view shows the stiff guide wire passing through the center of the deteriorated bioprosthetic tricuspid valve, with the wire tip in the right pulmonary artery. A balloon valvuloplasty was performed using an Inoue balloon (maximum diameter 26 mm).
View Figure 3
Movie 2: The echocardiogram revealed that peak velocity had decreased to 1.7 m/sec, with a mean diastolic gradient of 7.5 mmHg.
The patient's symptoms immediately improved. At six months postoperative, no recurrence of bioprosthetic tricuspid stenosis was reported. Thereafter, resection surgery on the large, post-mediastinum tumor was successfully performed via a left thoracotomy without any complications.
Severe tricuspid valve stenosis is associated with a mean gradient across the tricuspid valve of at least 5 mmHg, a calculated TV area of less than 1.0 cm2 or both [6,7]. Because of insufficient data, a percutaneous transcatheter tricuspid balloon valvuloplasty is less preferable to a tricuspid valve replacement. However in some situations, a tricuspid balloon valvuloplasty can be a useful treatment option, especially in patients considered to be a higher surgical risk. In general, a history of prior cardiac surgeries in an adult with congenital heart disease makes repeat surgery difficult because of adhesions, excessive bleeding, and other factors [8]. In our case, a fourth sternotomy and on-pump surgery seemed too invasive.
Feit and colleagues first reported on percutaneous transcatheter tricuspid balloon valvuloplasty in 1986 [9]. Rana and colleagues reviewed case reports of this type of transcatheter intervention, excluding the native tricuspid valve, that were published in journals from 1980 through 2017 [10]. In their review of 15 articles, 19 cases were identified and in 18 of these 19 cases (95%), the authors reported a successful reduction of the transvalvular pressure gradient. The mean pre-procedural pressure gradient was 12 mmHg and the post-procedural gradient improved to an average mean residual of 7 mmHg. There were no major procedure-related adverse events reported. In most cases, underlying cardiac disease was involved, such as rheumatic fever or infectious endocarditis. Wren and colleagues reported on a congenital heart disease case involving a minor-age patient [11]. No one reported on a case of adult congenital heart disease.
It remains unclear how long a bioprosthetic tricuspid valvuloplasty is effective. In general, the durability of a prosthetic valve in the aortic or mitral position is thought to be 10 or 20 years [12]. The durability of a prosthesis in the tricuspid position is less-known. A mechanical valve in the tricuspid position is known to be at a 20-times higher risk for valve thrombosis than in the aortic or mitral position [13]. When our patient undergoes a third bioprosthetic valve replacement, he will still need more valve replacement surgeries in the future. A balloon valvuloplasty is less invasive and can be repeatedly used as a palliative option, which may reduce the total number of valve replacement and sternotomy procedures. As a final point, the transcatheter implantation of a valve-in-valve for a failed tricuspid bioprosthesis might prove to be an appropriate option for highly select patients [14]. However durability remains unknown. To the best of our knowledge, this is the first case report of a percutaneous transacatheter balloon valvuloplasty for bioprosthetic tricuspid valve stenosis in a patient with adult congenital heart disease.
A percutaneous transcatheter tricuspid balloon valvuloplasty can be an alternative option to surgical tricuspid valve replacement, especially in high-risk patients. Certain patients with adult congenital heart disease will be suitable for this procedure.
None.
This report received no specific grant from any funding agency, commercial or not-for-profit entity.
The authors have no conflict of interest to disclose.
The patient provided informed consent for publication.
The authors have no conflicts of interest to disclose.