Cardiac involvement has been reported in children with SARS-CoV-2 inflammatory syndrome. The objectives herein were to assess changes in cardiac parameters and compare cardiac pathologies observed in SARS-CoV-2 to Kawasaki-(KW) and myocarditis-(MY).
Patients < 18 years of age, with symptoms of Kawasaki disease (KW group) and/or of acute myocarditis (MY group) from March 1 to May 31 2020 were included. Clinical, laboratory data, and cardiac parameters were recorded upon diagnosis and at several points over two weeks.
Sixteen patients (8 males) diagnosed in the context of SARS-CoV-2 pandemic, were included (8 KW, 8 MY); the mean age was 5.9 years (KW 2.7 years, MY 10.2 years; p = 0.0002). Fever was present in all. Conjunctivitis, buccal inflammation, lymphadenopathies, dermatological lesions were more frequent in KW, chest pain, and gastrointestinal symptoms more frequent in MY. C-reactive protein, procalcitonin, and fibrinogen levels were higher in MY. Platelets were higher, and hemoglobin and hematocrit lower in KW. Troponin and NT pro-BNP were higher in MY. Coronary anomalies were observed in KW, and mitral regurgitation and pericarditis in MY. LV systolic function impairment in MY was concomitant to the highest values of troponin, NT-pro-BNP, CRP, ferritin, D-dimers, and lowest value of lymphocytes and platelets. Mean length of intensive care stay was 7.8 days and that of hospital stay was 9.7 days (MY 5.1 days, KW 14.4 days, p = 0.0008). Systolic function was normalized within 15 days. Diastolic dysfunction appeared after normalization of systolic function.
Cardiac injury is parallel to biological changes, as evidenced by serial changes of systolic and diastolic myocardial ultrasound parameters.
SARS-CoV-2, Myocarditis, Kawasaki disease, Echocardiography, Inflammatory syndrome, Cardiac function
SARS-CoV-2 infection (COVID-19) has spread rapidly worldwide. While during the first wave of the pandemic children were not severely affected, a late wave unexpectedly occurred and pediatric patients presented with a new multisystem inflammatory disease mimicking Kawasaki disease (KW) and/or acute myocarditis (MY). Previous publications have reported the clinical presentation, laboratory data, and short-term outcomes of such cases to describe this novel pediatric syndrome triggered by SARS-CoV-2 [1-6]. To our knowledge, repeated measures of laboratory and of concomitant cardiac parameters have not been previously assessed in these pediatric cases, whereas cardiac data have been reported in adults [7-12].
We report herein the experience of the Lyon (France) medical center for children diagnosed from March to the end of May 2020, and compare data to that reported in the literature. The objectives of the study were to describe the clinical, laboratory, and cardiac data of pediatric patients suffering from SARS-CoV-2 multisystem inflammatory disease, to compare the myocarditis-like to Kawasaki-like cases, to evaluate the changes over time in the early phase the disease with repeated measurements of laboratory and cardiac parameters, and to evaluate short-term outcomes.
All patients under the age of 18, who were admitted during the SARS-CoV-2 pandemic at the Lyon medical center from March 1 to May 31, 2020, with fever and symptoms of KW and/or hemodynamic failure (MY), were included in the cohort. The SARS-CoV-2 was proven by PCR result or familial at-risk contact, or was suspected in the context of the epidemic period.
Hemodynamic failure was defined as compromised organ tissue perfusion, hypotension, tachycardia, signs of heart failure, low cardiac output, oliguria and inotrope support requirement, due to impaired myocardial function.
The following laboratory data were recorded at hospitalization and at different time points during hospital stay: C-reactive protein, procalcitonin, hemoglobin, hematocrit, leukocyte count, neutrophil count, lymphocyte count, platelet count, albumin, ferritin, D-dimers, fibrinogen, NT pro-BNP, and troponin; the time points were: at diagnosis: Day-0, day-1-2, day-3, day-4, day-5, day-6, day-8, day-10, day-12, and day-14.
The patients were divided into MY and KW groups and compared with regard to laboratory data and echocardiographic parameters. The MY group included cases with symptoms of heart failure and/or compromised hemodynamics and impaired myocardial function by ultrasound examination; the KW group included patients with clinical symptoms of KW and preserved myocardial function.
Cardiac function was evaluated by echocardiography and Doppler. Ultrasound assessment of left ventricle (LV) systolic function included measurements of ventricular end-diastolic ventricular diameter (LVEDD), ventricular end-systolic ventricular diameter (LVESD), left ventricular ejection fraction (LVEF) using the Simpson method, left ventricular shortening fraction calculated as (LVEDD-LVESD)/LVEDD and sub-aortic velocity integral (VTI), of right ventricular function by tricuspid annular plane systolic excursion (TAPSE), of LV diastolic function by E and A mitral waves, E/A ratio, and Doppler tissue imaging indices including Ea wave at mitral annulus and E/Ea ratio. Mitral regurgitation, pericardial effusion and coronary arteries anomalies were also assessed. Cardiac ultrasound data were recorded at admission and at each time-point defined above and compared to simultaneous laboratory data.
LVEF was considered as impaired if < 60% and LVSF if < 35%.
All data were entered into an Excel spreadsheet (Microsoft Excel 97-2003, Redmond, WA, USA), and Statview software version 5.0 (SAS Institute Inc., Cary, NC, USA) was used for statistical analysis. Continuous variables were expressed as mean ± standard deviation (SD), and categorical variables as counts and percentage. Comparisons were made using the two-tailed paired Student's t test or the Mann-Whitney
U test for the continuous variables, and the Chi2 test for nominal variables. The level of statistical significance was set at a p value of less than 0.05.
Sixteen patients were included, 8 males and 8 females, and the mean ± SD age was 5.9 ± 4.6 years (range: 2.5 months - 3.2 years) at diagnosis. Among these, 8 were included in the MY group, and 8 in the KW group. The mean age at diagnosis was significantly lower in KW group (mean ± standard deviation = 2.7 ± 2.0 years) than in the MY group (10.3 ± 3.3 years; p = 0.0002), as were weight and body mass index at diagnosis (Table 1).
Table 1: Patient characteristics. View Table 1
An at-risk contact with a family relative infected with COVID-19 was proved in 6 cases (37.5%): 2 in KW and 4 in MY group, and PCR swab was COVID-19 positive in 4 cases (25.0%): 0 in KW and 4 in MY group (Table 1). Onset of symptoms occurred at mean ± standard deviation = 4.7 ± 2.4 days prior to diagnosis. Fever was present in all cases. Dermatological lesions were significantly more frequent in the KW group and there was a trend towards more frequent conjunctivitis, buccal mucosal inflammation (cheilitis), and lymphadenopathies in this group, while there was a trend towards more frequent chest pain, gastrointestinal, and respiratory symptoms in the MY group; the frequency of neurological and ENT symptoms at onset did not differ significantly between both groups (Table 1). A total of 6/8 patients in the MY group presented with clinical symptoms of severe heart failure and were admitted to the intensive care unit at the time of diagnosis, whereas cardiac function deteriorated within 2 days after admission in the other 2 cases of MY group.
Lymphocyte counts were significantly lower in the MY group, while platelet counts were significantly higher and hemoglobin and hematocrit were significantly lower in the KW group. There was a trend towards higher C-reactive protein, procalcitonin, and fibrinogen levels in the MY group. D-dimers, ferritin, troponin, and NT-proBNP levels were elevated in MY patients (Table 2).
Table 2: Laboratory data at admission. View Table 2
Coronary artery abnormalities were observed in 5 cases (31.2%): 4 KW patients (50.0%), 1 MY patient (6.2%). No coronary aneurysm was diagnosed, coronary artery dilation was the only observed coronary artery lesion. One patient in MY group exhibited dilatation of right and left coronary arteries. Myocardial systolic function was impaired in the MY group (LVEF < 60%, LVSF < 35%). Mitral regurgitation was significantly more frequent, and there was a trend towards this for pericarditis in the MY group. Right ventricular systolic function was preserved (as assessed by TAPSE index) and did not differ significantly between both groups. Indexed left ventricular end-diastolic diameters were within the normal range in the MY group whereas end-systolic diameters were increased. LV diameters were significantly greater in the KW group than in the MY group. There was no significant difference between groups regarding the indices of diastolic function at diagnosis (E/A and E/Ea ratios) at presentation (Table 3).
Table 3: Echocardiographic data at admission. View Table 3
All patients received a 2g per kg dose of intravenous immunoglobulins infusion within 24 hours after admission (at day-0 or day-1-2): A single dose in the KW group and two doses of 1 g/kg/day each (for 2 days) in the MY group. Inotrope support was needed in 6/8 patients of the MY group (the duration of which ranged from 4 to 7 days, mean 5 days) and none of the KW group. No patient needed extracorporeal membrane oxygenator (ECMO) support. Other therapeutic management included: interleukin 1 antagonist monoclonal antibody in 2 cases (KW group), and corticosteroids in 2 cases. The mean ± standard deviation duration of intensive care stay was 7.8 ± 5.8 days in MY patients (no KW patient was admitted to an intensive care unit). The mean ± standard deviation duration of hospital stay was 9.7 ± 6.1 days; it was longer in the MY group (5.1 ± 3.5 days) than in the KW group (14.4 ± 4.2 days; p = 0.0008).
Mean ± standard deviation levels of D-dimers, C-reactive protein and ferritin (Figure 1A, Figure 1B and Figure 1C) were elevated from admission and peaked respectively at day 1-2, day 3 and day 4; they were higher in MY than KW patients. In both groups, the mean albumin levels were generally below normal values and lowest levels occurred at day 4 (Figure 1D). Lymphopenia and thrombopenia were observed in MY patients at admission (day 0) and normalized progressively during the first five days; lymphocyte count was within the normal range, and platelet count increased beyond fifth day in KW patients (Figure 1E and Figure 1F)).
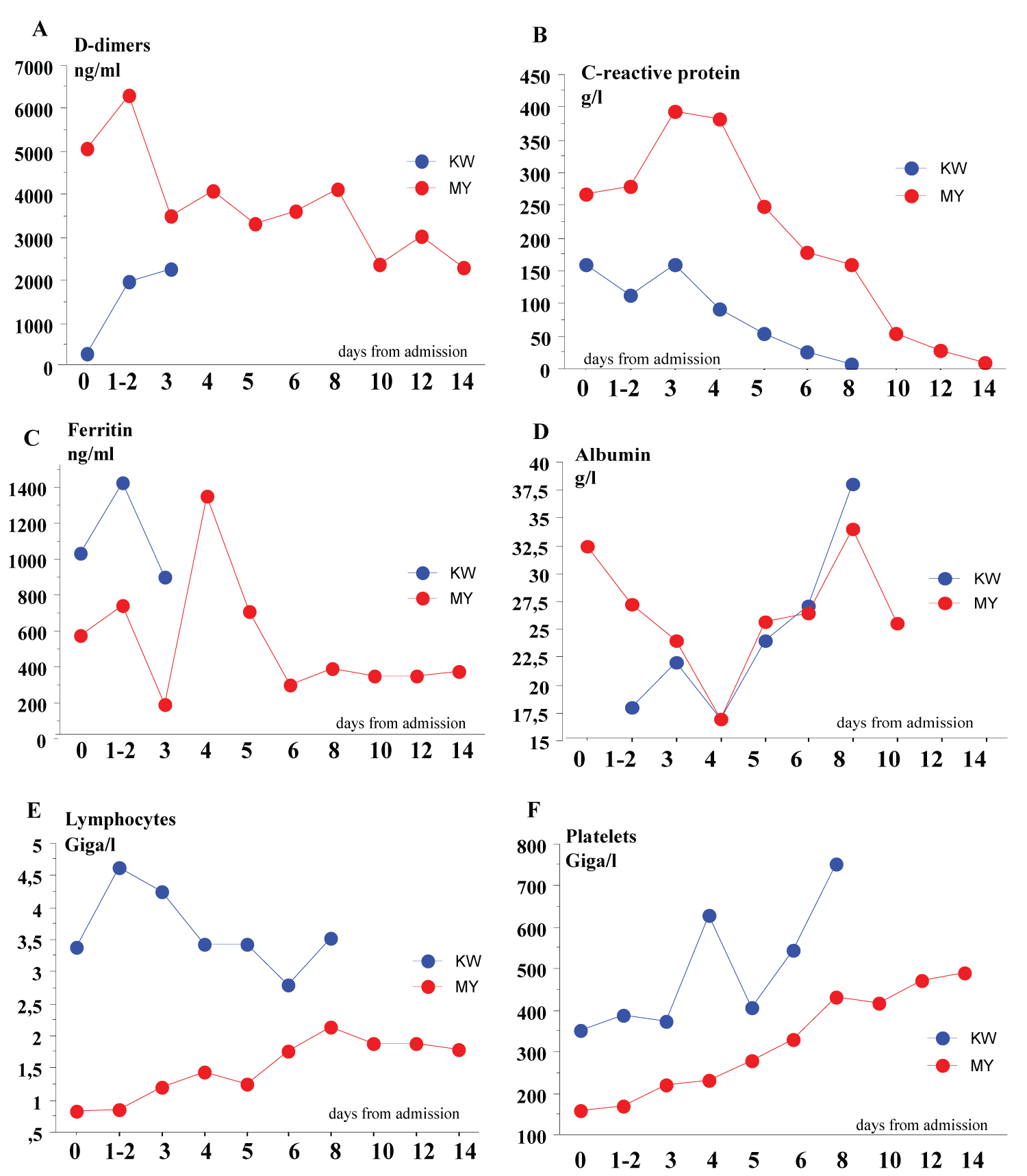 Figure 1: Laboratory parameters blood levels changes over the early phase of COVID 19. (A) D-dimers; (B) C-reactive protein; (C) Ferritin; (D) Albumin; (E) Lymphocytes counts; (F) Platelet counts. View Figure 1
Figure 1: Laboratory parameters blood levels changes over the early phase of COVID 19. (A) D-dimers; (B) C-reactive protein; (C) Ferritin; (D) Albumin; (E) Lymphocytes counts; (F) Platelet counts. View Figure 1
A qualitative serological test was performed in all patients. Anti-SARS-CoV-2 IgG antibodies were positive in 7/8 patients in the MY group (87.5%) and none in KW group.
LV systolic function (assessed by LVEF, LVSF) was impaired at onset and decreased within the first 4 days after admission, the lowest value was recorded at day 1-2 and day 3 (Figure 2A and Figure 2B), concomitant to the highest levels of troponin and NT-pro-BNP (Figure 2C and Figure 2D), and to peak levels of D-dimers, C-reactive protein, ferritin, and nadir levels of lymphocyte and platelets counts (Figure 1A to Figure 1C, Figure 1E and Figure 1F). Ultrasound parameters of systolic function returned to normal values within 10 days after admission. Diastolic dysfunction, identified by increased E/A and E/Ea ratios, was observed in the immediate course after normalization of systolic function, i.e. from T8 (day 14; Figure 2E and Figure 2F).
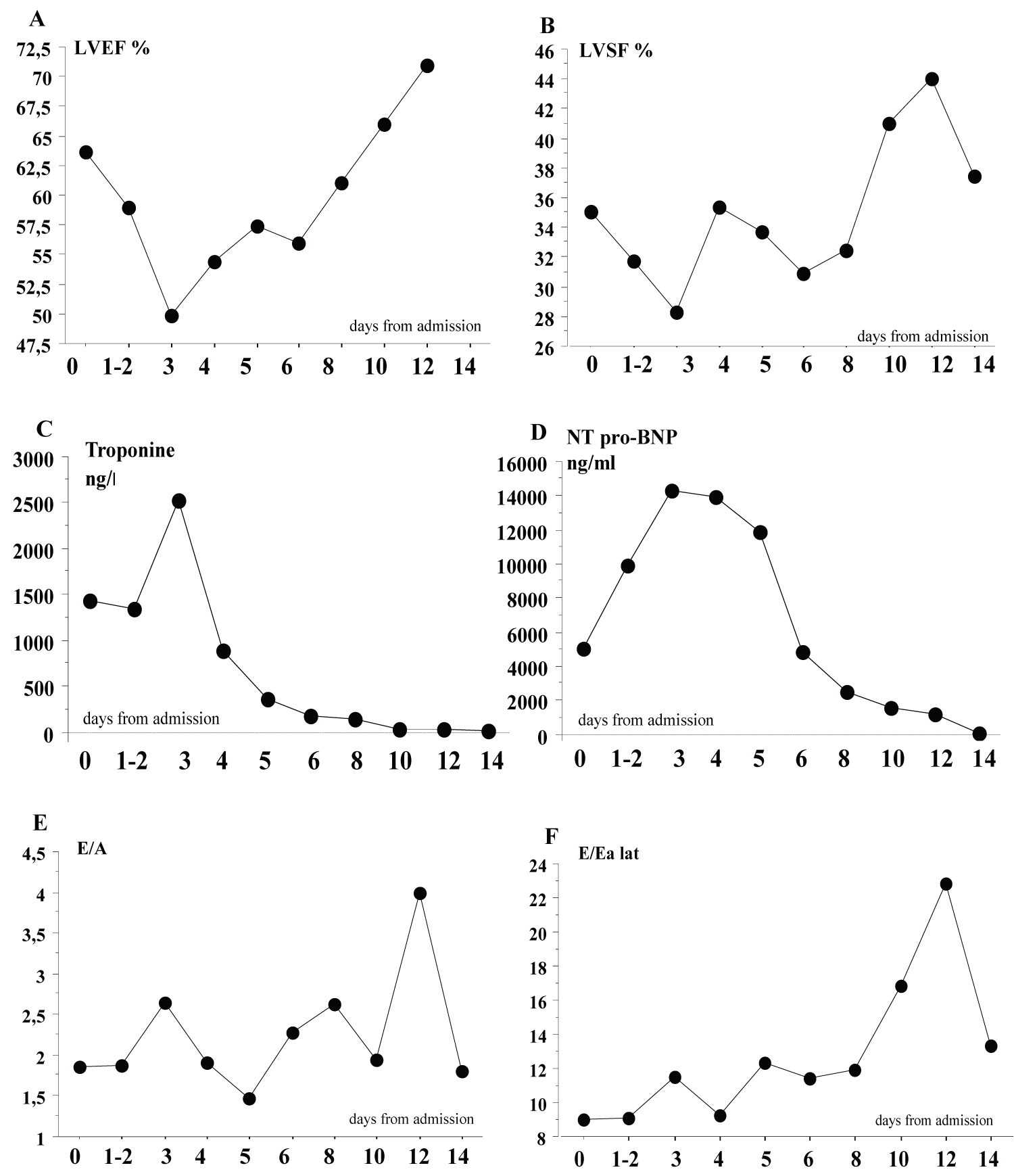 Figure 2: Myocardial ultrasound parameters of systolic function (LVSF, LVEF), diastolic function (E/A ratio, E/Ealat and cardiac biomarkers blood levels (troponin and NT pro-BNP) changes over the early phase of COVID 19. (A) LVSF (Left Ventricle Shortening Fraction); (B) LVEF (Left Ventricle Ejection Fraction); (C) Trponine; (D) NT pro-BNP; (E) Ratio E/A (mitral pulsed Doppler E wave and A wave); (F) Ratio E/Ea (mitral pulsed Doppler E wave and tissue Doppler imaging at mitral annulus Ea wave). View Figure 2
Figure 2: Myocardial ultrasound parameters of systolic function (LVSF, LVEF), diastolic function (E/A ratio, E/Ealat and cardiac biomarkers blood levels (troponin and NT pro-BNP) changes over the early phase of COVID 19. (A) LVSF (Left Ventricle Shortening Fraction); (B) LVEF (Left Ventricle Ejection Fraction); (C) Trponine; (D) NT pro-BNP; (E) Ratio E/A (mitral pulsed Doppler E wave and A wave); (F) Ratio E/Ea (mitral pulsed Doppler E wave and tissue Doppler imaging at mitral annulus Ea wave). View Figure 2
All patients in the MY group received angiotensin-convertor enzyme inhibitors and a first dose of beta blockers associated with aspirin antiplatelet therapy prior to discharge home.
The pediatric SARS-CoV-2 cardiovascular disease was characterized by severe inflammation (evidenced by CRP and fibrinogen) with decreased albumin levels, lymphopenia, and elevated D-dimers and ferritin blood levels, as has been reported elsewhere [1-6]. Some investigators have suggested that the coronavirus family might be one of the triggers of KW, SARS-CoV-2 being a particularly virulent strain able to elicit a powerful immune response in the host [13-18]. Acute MY causing severe left ventricle dysfunction has also been reported in the SARS-CoV-2 pandemic and might result from the same mechanisms [19-21]. The two clinical and laboratory test profiles, namely KW and MY, might be various expressions of the same disease. In the present study, all patients suffered from multisystem inflammatory syndrome which was the most severe in the MY group; the MY-type cases are characterized by a more severe laboratory test profile than the KW-type cases. One notable clinical feature is the frequency of gastrointestinal symptoms which can imitate severe acute peritonitis, so that some patients undergo laparoscopy for misdiagnosed peritonitis due to appendicitis; this was the case for one patient herein, and has been reported elsewhere [19].
The present study aimed to analyze the changes over time in myocardial function during the early phase of the disease and to assess the concomitant variations of laboratory test parameters and cardiac biomarkers. We found that myocardial systolic function is altered early during the inflammatory reaction; decreased ejection fraction was parallel to the peak of NTpro-BNP and occurred shortly after the troponin peak blood level. Thus, troponin, as a biomarker of myocardial injury, might help to predict cardiac dysfunction in such patients.
The prompt improvement of systolic function after intravenous immunoglobulin infusion, is in favor of reversible myocardial lesions. The mechanism of COVID-19 -induced MY is still a matter of debate and is supposed to be of multifactorial origin, i.e. direct viral injury, cytokine storm and myocardial hypoxemia [20-22]. In this model, SARS-CoV-2 leads to an inflammatory state, which results in a cytokine storm triggered by an uncontrolled and dysfunctional immune response [22-24]; cytokines then cause apoptosis and necrosis of myocardial cells, and viral infection may induce increased myocardial oxygen demand and create a state of oxygen supply-demand mismatch [22-25]. Aggression of the myocardium alters systolic function parallel to the biological inflammatory state. Direct viral attack on the myocardium is unlikely since the symptomatology is delayed by almost a month compared to the infectious contact and the positivity of nasal PCR is rare, which indicates low viral load at this stage. It is most likely that the inflammatory aggression of the myocardium is due to activation of the post-viral cytokine inflammatory cascade; this mechanism is comparable to KW. The major cytokine inflammation responsible for dramatic fall in systemic vascular resistance, might also lead to additional myocardial ischemia aggravating the process of acute myocardial aggression. This is supported by the requirement of norepinephrine to stabilize compromised hemodynamics in these patients, both in our experience and others' [26,27]. The rise in troponin occurs prior to the elevation of the natriuretic peptide and indicates myocytic damage, which would support significant myocardial injury. Surprisingly, herein, magnetic resonance imaging found no specific evidence of MY despite severe inflammatory syndrome and impaired myocardial function, except in one case who had gadolinium early enhancement in the lateral left ventricular wall, global left ventricle dysfunction by transthoracic ultrasound, ventricular early beats on ECG, and exhibited the highest troponin level at onset (12123 ng/L). One might argue that troponin levels need to reach some threshold for cardiac resonance magnetic imaging to detect myocardial inflammation. In Luetkens, et al. case report [28], the patient troponine levels were 18,800 ng/L and CMR imaging showed early gadolinium enhancement due to interstitial myocardial edema.CMR parameters have been defined in the revised Lake Louise criteria for diagnosis of acute myocarditis [29].
The end-diastolic diameter of the left ventricle was not significantly increased in the present study (end-diastolic indexed diameter was within the normal range, i.e. less than 31 mm/m2), which evidences the acute and sudden myocardial injury and absence of myocardial remodeling [9,30-33]. Intravenous immunoglobulins were administered to all patients, and laboratory and ultrasound parameters of systolic function returned to normal in the following days after infusion, i.e. within two weeks after onset of the disease. Inotrope support could be discontinued in less than one week after admission. The recovery of myocardial contractile function was observed very quickly after intravenous immunoglobulins infusion. This favorable and prompt outcome is unusual in pediatric acute MY, where the ability of injured myocardium to improve is uncertain and in any case occurs late and within months after onset of the disease [34]. Interestingly, the data presented herein show impairment of diastolic function in the immediate course after recovery of myocardial contractility and regression of the inflammatory process; one might argue that diastolic dysfunction was present earlier and only revealed after normalization of systolic function. These data encourage to monitor the myocardial cardiological outcome of these patients in the long-term in order to detect further myocardial fibrosis. No report exists to date about the mid and long-term outcomes of myocardium systolic and diastolic function. Further studies are needed to determine the potential of myocardium to evolve towards irreversible scars and fibrosis, by magnetic resonance imaging and/or echocardiographic and Doppler evaluation.
The present study has found that SARS-CoV-2 myocarditis-like cases are characterized by a more severe laboratory test profile than the Kawasaki-like cases, and has evidenced that cardiac injury is parallel to laboratory test changes. Serial echocardiographic monitoring of systolic and diastolic myocardial parameters should be also needed. Moreover, despite restoration of myocardial contractility, long-term evaluation of diastolic function disorders and detection of fibrosis should be recommended.
None.