Atrial arrhythmia (AA) and left ventricular systolic dysfunction (LVSD) frequently co-exist, and potentially represent growing public health problems. Determining whether tachycardia is an indicator or consequence of LVSD in patients with tachycardia and LVSD is difficult. Tachycardia-mediated cardiomyopathy (TCM) raises a "chicken or egg" question. Regardless of the etiology, patients with severe LVSD are considered candidates for the implantation of a cardioverter defibrillator as primary prevention. LVSD is well known to improve after successful treatment of AA in the TCM population, but enlarged left ventricular end-diastolic dimension (LVDd) is considered a parameter of irreversible LVSD.
Enlarged LVDd is not a predictor of irreversible LVSD in patients with AA.
We assessed patients with persistent AA and heart failure (ejection fraction (EF) < 45%) who received aggressive rhythm control therapy and maintained sinus rhythm with catheter ablation, electrical cardioversion and anti-arrhythmic drugs. Echocardiographic parameters and concentrations of brain natriuretic peptide (BNP) were evaluated before and after treatment.
Participants comprised 38 consecutive patients with persistent AA and LVSD (36 men; mean age, 64.3 ± 8.8 years). Thirty-three patients (86.8%) could be restored to and maintained sinus rhythm. After rhythm control therapy, EF, left ventricular end-systolic volume (ESV) and BNP were improved in all patients (EF: 30.7 ± 10.9% to 54.6 ± 7.4%; ESV: 80.8 ± 32.4 ml to 51.7 ± 22.9 ml; BNP: 382.0 ± 251.1 pg/ml to 50.5 ± 30.7 pg/ml; P < 0.001 each). Conversely, no significant improvements were seen in LVDd or left ventricular end-diastolic volume (EDV) (LVDd: 54.8 ± 7.3 mm to 54.1 ± 5.0 mm, P = 0.40; EDV: 118.2 ± 37.7 ml to 109.2 ± 34.9 ml, P = 0.17). Enlarged LVDd > 60 mm was observed in 7 patients (21%) before treatment. EF was normalized in patients with severe LVSD (EF < 35%, 16/38, 48%).
Unnecessary device implantation may be avoidable if aggressive rhythm control therapy is used in patients with AA and severe LVSD. Even in patients with severe LVSD and enlarged LVDd, LVSD is likely to prove reversible if AA is also present.
Atrial arrhythmia, Tachycardia-mediated cardiomyopathy, Catheter ablation, Left ventricular systolic dysfunction, Left ventricular end-diastolic dimension
Tachycardia-induced cardiomyopathy (TCM) is a reversible disease entity of dilated cardiomyopathy. Atrial arrhythmia (AA) plays a critical role in the lead-up to TCM, and the most common etiology is atrial fibrillation (AF) with rapid ventricular response [1]. The number of patients with AA is steadily increasing [2]. AA and left ventricular systolic dysfunction (LVSD) frequently co-exist, and appear to be looming as burgeoning public health problems. The presence of AF and heart failure (HF) is associated with increased morbidity, mortality, and healthcare costs [3,4]. The restoration and maintenance of sinus rhythm (SR) in patients with AA and LVSD are known to often improve symptoms [5]. Some recent clinical studies have shown that aggressive rhythm control therapy including catheter ablation leads to reductions in all-cause mortality and hospitalization for worsening (HF) and improvements in quality of life [6-9]. Furthermore, sub-analyses have shown that rhythm control therapy improved left ventricular systolic function [10]. However, in actual clinical practice, the relationship between AA and HF represents a common conundrum for cardiologists and general physicians alike, as a kind of "chicken or egg" situation as to whether AA begets HF or vice versa.
Although TCM is generally regarded as a reversible entity, LVSD is indicated for implantable cardioverter defibrillator (ICD) implantation regardless of etiology under the current guidelines. ICD therapy is recommended to prevent sudden cardiac death (SCD) in patients with symptomatic HF (New York Heart Association (NYHA) class II-III) and LVEF ≤ 35% after ≥ 3 months of optimal medical therapy who are expected to survive for ≥ 1 year with good functional status [11,12].
The only way to diagnose TCM is to treat arrhythmias [1]. Although LVSD in patients with TCM is generally considered reversible, some clinical data have shown that LVSD is irreversible when the left ventricle is severely enlarged [13]. Reversal of cardiomyopathy by eliminating arrhythmia not only confirms the diagnosis, but may significantly improve outcomes. We hypothesized that enlarged LVDd would not represent a predictor of irreversible LVSD among patients with AA. In the present study, we performed aggressive rhythm control therapy for patients with AA and LVSD. Cardiac parameters were observed after successful rhythm control therapy.
We assessed patients with persistent AA and HF (ejection fraction (EF) < 45%) who received rhythm control therapy. Patients who were hospitalized with persistent AA and LVSD between April 2016 and October 2021 receiving aggressive rhythm control therapy were enrolled in this study. Ischemic heart disease was excluded with coronary angiography or coronary computed tomography. Echocardiographic parameters and concentrations of brain natriuretic peptide (BNP) were evaluated for those patients in whom SR was maintained. The patients with severe valvular heart disease revealed with echocardiography at baseline were excluded from the study. This study was approved by the St. Marianna University School of Medicine Ethics Committee (approval no. 5020).
We performed the rhythm control therapy for patients with LVSD in AA, and then tried to maintain SR as long as possible. Catheter ablation (CA) was performed in all patients except those who declined to undergo the procedure. After the procedure, a class 1 or 3 antiarrhythmic drug (AAD) was administered. For patients who did not undergo CA, AAD was used in combination with external cardioversion.
Cases with left atrium (LA) or left atrial appendage thrombus were excluded by transesophageal echocardiography and/or contrast-enhanced computed tomography before the procedure in all patients. Written informed consent for the procedure was obtained from all patients.
Circumferential pulmonary vein isolation (PVI) was mandatory as the primary method. The procedure was performed with general anesthesia or deep sedation using midazolam and continuous infusion of propofol. Esophageal temperature was monitored during the whole procedure to avoid esophageal injury. A 5-Fr decapolar catheter (BeeAT; Japan Life Line, Tokyo, Japan) was positioned in the coronary sinus through the right jugular vein. Through the right femoral vein, a steerable 8.5-Fr long sheath (Agilis; Abbott Medical, Minneapolis, MN) and a non-steerable 8.5-Fr long sheath (SL0; Abbott Medical) were introduced into the LA using single transseptal puncture guided by fluoroscopy and intracardiac echocardiography. Directly after transseptal puncture, heparin was administered intravenously at a dose of 100 U/kg body weight. Thereafter, activated clotting time (ACT) was checked every 30 min and more heparin was administered as necessary to maintain ACT > 300s.
LA geometry was acquired using a multipolar mapping catheter (Pentaray; Biosense Webster, Diamond Bar, CA or HD Grid; Abbott Medical, Minneapolis, MN) in combination with a three-dimensional mapping system (CARTO3; Biosense Webster or EnSite; Abbott Medical) with computed tomography image integration. An open-irrigated radiofrequency (RF) ablation catheter with a contact force sensor (Surround Flow; Biosense Webster or Tacticath; Abbott Medical) was used in all cases. The ablation catheter was positioned in the LA through the Agilis sheath, and then PVI was performed. In one case, focal ablation was performed for atrial tachycardia (AT) arising from the lateral portion of the mitral annulus. PVI was not performed because no arrhythmia was inducible after the ablation of AT and SR was maintained.
Each pulse was delivered in a power-control mode. RF energy was limited to 35 W for all locations in the LA, ablation duration was limited to 60s. When esophageal temperature increased to > 39 ℃, energy delivery was stopped immediately and lower RF energy in the range of 20-25 W was applied.
The acute endpoint of AF ablation was the creation of conduction block in all PVs. Isolation of each pulmonary vein (PV) was confirmed by entrance block using the multipolar mapping catheter. When PVI was not achieved with circumferential ablation, earliest PV potential was targeted sequentially until complete isolation was achieved. Internal cardioversion using a catheter positioned in the coronary sinus was performed when SR was not achieved by ablation.
When AT remained after completion of PVI, mitral isthmus ablation, LA posterior wall isolation, and superior vena cava isolation, Cavo-tricuspid isthmus block line was added to PVI, depending on the mechanism of AT. Additional ablation lesions were made at the discretion of the operators.
After the procedure, AAD was administered, unless the patient had sinus node or atrioventricular conduction dysfunction. Efforts to maintain SR were continued even after CA.
Trans-thoracic echocardiography was performed during AA at baseline and SR after rhythm control therapy. Echocardiography was performed at 3 months after the restoration of SR. Echocardiography was performed in the left lateral decubitus position using a Vivid E95 system (GE Medical Systems, Milwaukee, WI). To avoid underestimation of left ventricular systolic function during AA at baseline, calculations of all parameters were performed at the preceding RR interval > 500 ms. Left ventricular end-diastolic volume (LVEDV) and left ventricular end-systolic volume (LVESV) were measured from standard apical four-chamber views in 2D mode using a modification of Simpson's rule, then LVEF was derived [14].
Left atrial volume (LAV) was measured in the same view. Left atrial volume index (LAVI) was then calculated as LAV divided by body surface area. Left atrial dimension (LAD), left ventricular end-diastolic dimension (LVDd), and left ventricular end-systolic dimension (LVDs) were measured from the standard long-axis view in M mode. Echocardiographic data were acquired by an investigator blinded to the study protocol.
All continuous variables are expressed as mean ± standard deviation. Student's t test was used for statistical analyses. Two-tailed probability values of P < 0.01 were considered statistically significant. All statistical analyses were performed using SPSS version 23 (SPSS, Chicago, IL, USA).
Aggressive rhythm control therapy was performed for 38 consecutive patients with persistent AA and LVSD (35 men; mean age, 64.3 ± 8.8 years). SR was maintained in 33 patients (86.8%) after rhythm control therapy during the observation period (mean, 5.1 ± 4.4 months). Patient characteristics are shown in Table 1. There were no cases with severe mitral regurgitation. No complications related to the procedure were encountered. An AAD was administered in 24 patients (73%). At baseline, enlarged LVDd > 60 mm was observed in 7 patients (21%). Sixteen patients (48%) displayed severe LVSD, with EF < 35%.
Table 1: Patient characteristics. View Table 1
Heart rate reduced from 98.7 ± 20.0 beats/min during AA to 58.7 ± 6.8 beats/min during SR (P < 0.001). EF and BNP were improved in all patients (EF: 30.7 ± 10.9% to 54.6 ± 7.4%, P < 0.001; BNP: 382.0 ± 251.1 pg/ml to 50.5 ± 30.7 pg/ml, P < 0.001) (Figure 1A). LA volume was reduced after maintenance of SR (45.0 ± 5.5 to 40.9 ± 4.8, P < 0.001). Significant reductions in LVDs and LVESV were also observed after maintenance of SR (LVDs: 46.5 ± 8.0 mm to 39.4 ± 6.5 mm, P < 0.001; LVESV: 80.8 ± 32.4 ml to 51.7 ± 22.9 ml, P < 0.001). On the other hand, no significant reductions were identified in LVDd and LVEDV (LVDd: 54.8 ± 7.3 mm to 54.1 ± 5.0 mm, P = 0.40; LVEDV: 118.2 ± 37.7 ml to 109.2 ± 34.9 ml, P = 0.17) (Table 2).
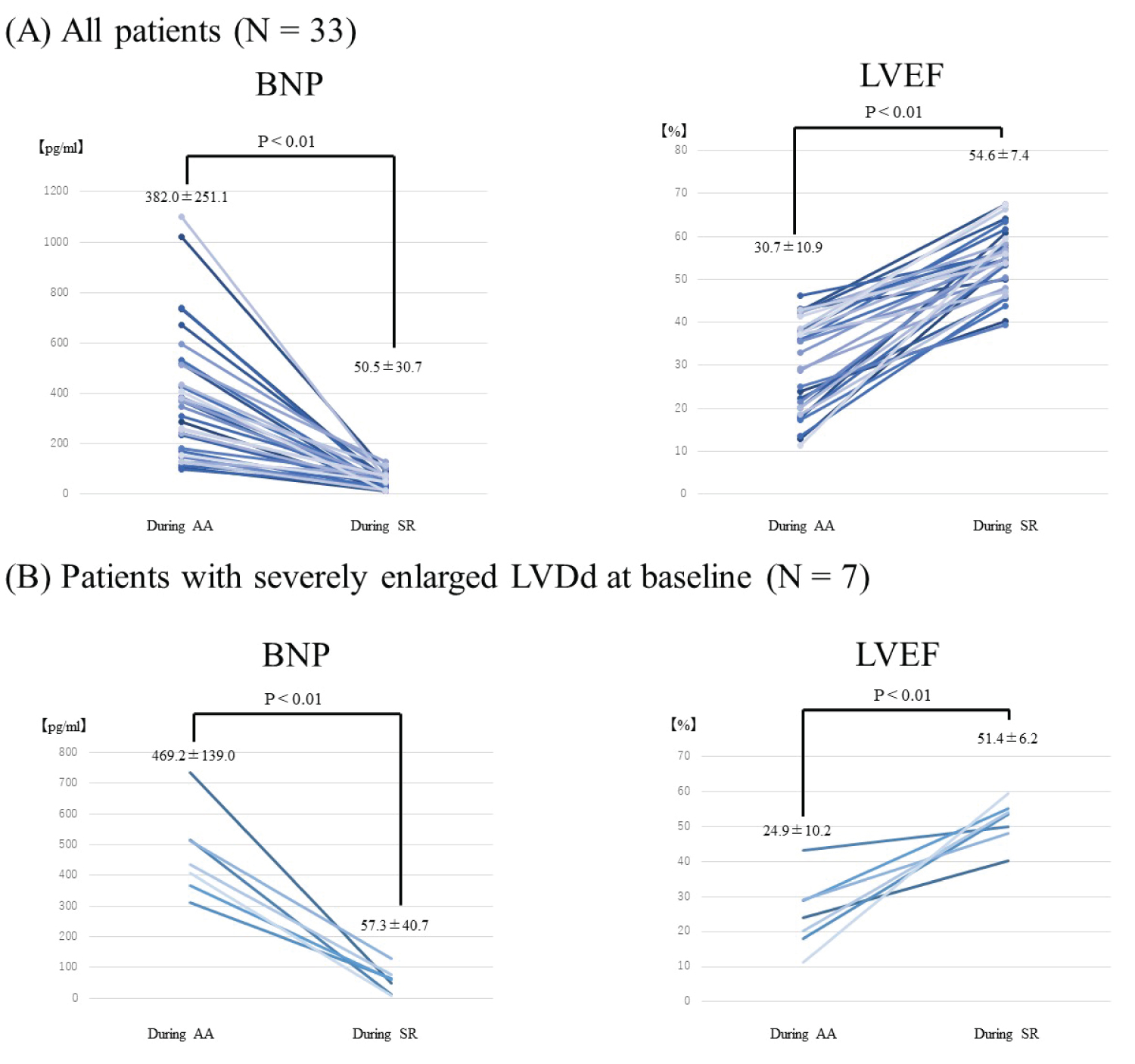 Figure 1: BNP and LVEF before and after rhythm control therapy. A) BNP and EF at baseline during AA and after rhythm control therapy during SR in all patients (N = 33). BNP and EF were improved after maintenance of SR in all patients; B) Among patients with severely enlarged LV (LVDd > 60 mm), LVEF and BNP were significantly improved.
Figure 1: BNP and LVEF before and after rhythm control therapy. A) BNP and EF at baseline during AA and after rhythm control therapy during SR in all patients (N = 33). BNP and EF were improved after maintenance of SR in all patients; B) Among patients with severely enlarged LV (LVDd > 60 mm), LVEF and BNP were significantly improved.
AA: Atrial Arrhythmia; SR: Sinus Rhythm; BNP: Brain Natriuretic Peptide; EF: Left Ventricular Ejection Fraction.
View Figure 1
Table 2: Echocardiographic parameters before and after rhythm control therapy. View Table 2
Among the 7 patients with severely enlarged LV (LVDd > 60 mm), LVEF and BNP were improved significantly to the same extent as patients without enlarged LV (Figure 1B). In comparisons between patients with enlarged LVDd and without enlarged LVDd, both ΔLVEF and ΔLVESV also improved (Figure 2).
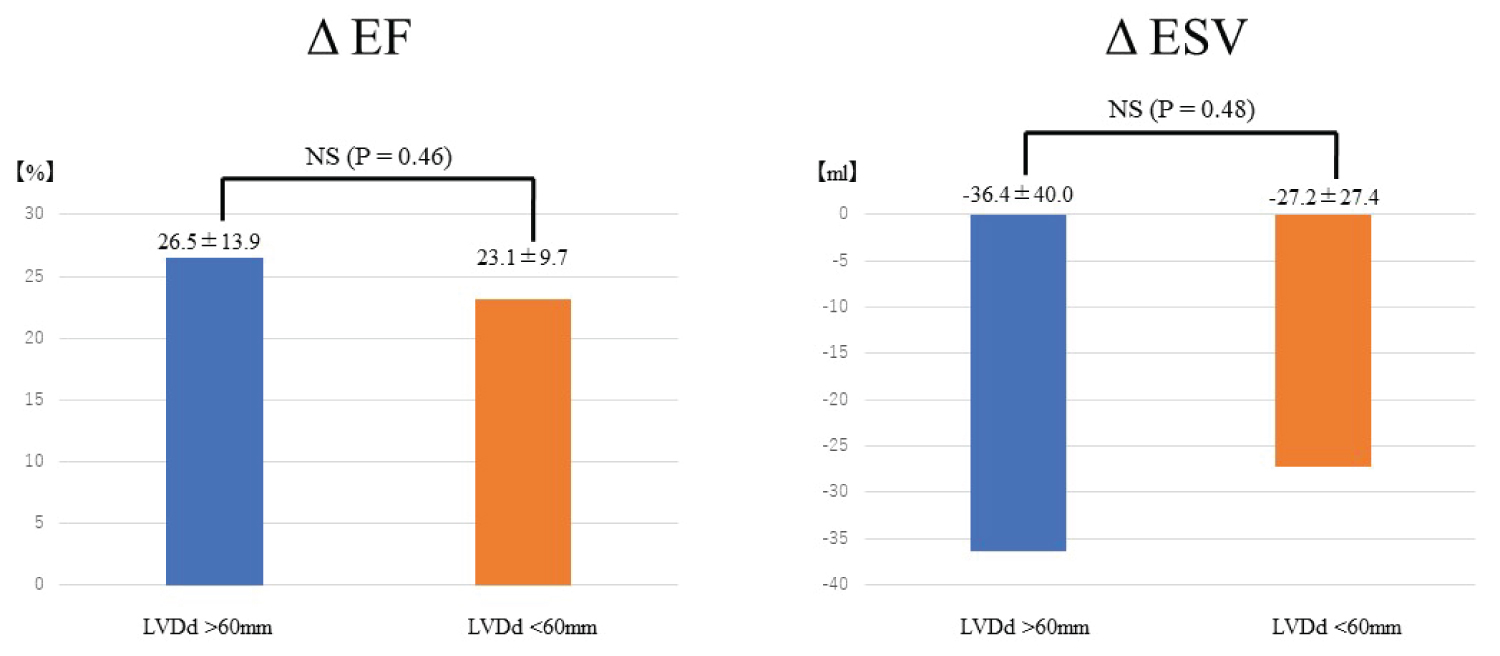 Figure 2: Comparison of ΔEF and ΔESV after rhythm control between patients with enlarged LVDd > 60 mm at baseline (blue bar, N = 26) and patients without enlarged LVDd at baseline (orange bar, N = 7).
Figure 2: Comparison of ΔEF and ΔESV after rhythm control between patients with enlarged LVDd > 60 mm at baseline (blue bar, N = 26) and patients without enlarged LVDd at baseline (orange bar, N = 7).
Left panel shows the comparison of ΔEF, and right panel shows the comparison of ΔESV. In comparison between patients with and without enlarged LVDd, both ΔLVEF and ΔLVESV are also improved. No significant differences in either parameter are seen.
AA: Atrial Arrhythmia; BMI: Body Mass Index; LVEF: Left Ventricular Ejection Fraction; LAD: Left Atrial Dimension; Lvdd: Left Ventricular End-Diastolic Dimension; Lvds: Left Ventricular End-Systolic Dimension; LVEDV: Left Ventricular End-Diastolic Volume; LVESV: Left Ventricular End-Systolic Volume; AAD: Anti-Arrhythmic Drug; ACEI: Angiotensin-Converting Enzyme Inhibitor; DOAC: Direct Oral Anticoagulant
View Figure 2
This clinical study revealed improvement of left ventricular systolic function after restoration of SR in the population of LVSD patients with AA. The main findings were as follows. First, aggressive rhythm control was able to maintain SR in 86.8% of patients with LVSD and long-lasting AA with a mean duration of 1.6 years. Second, 48% of patients had severe LVSD with EF 35%. Third, after SR had been restored and maintained, BNP and EF were significantly improved. Fourth, LV systolic function was improved even in those patients for whom LVDd was > 60 mm at baseline.
HF and tachyarrhythmia showed a strong association, particularly for AF. In general, tachyarrhythmia is a secondary consequence following advanced HF. This relationship may be partly attributable to the acute deterioration of LV function and rapid development of HF symptoms without structural remodeling. Tachyarrhythmias have long been considered part of the clinical presentation for HF. However, AA alone has been shown to result in reversible nonischemic cardiomyopathy, as so-called TCM. TCM is recognized as an entity of reversible LVSD solely due to the increase in ventricular rates, regardless of the etiology of tachycardia.
Despite AF being the most prevalent arrhythmia, clear data on the development of TCM in this population remain lacking. A single study reported TCM in 4% of patients referred for PVI [15]. AT can also be an etiological factor for TCM. TCM has been reported in 10% of patients with AT, particularly in incessant AT such as permanent junctional reciprocating tachycardia [16].
The mechanisms underlying TCM remain unclear. A previous study demonstrated that the irregular cycles of excitation and contraction with calcium mishandling induce an altered profile of gene and protein expression that are associated with functional changes at cellular and tissue levels [17]. Loss of atrial contraction, limited ventricular filling and increased filling pressure, functional mitral regurgitation, and diastolic dysfunction were considered as the mechanisms of TCM due to AA [18].
Although some mechanisms of TCM were revealed as mentioned above, no approach has been established to diagnose TCM before the treatment of tachyarrhythmia. During the persistence of AA, determining whether tachycardia leads to the LVSD or LVSD leads to the tachycardia is difficult. The coexistence of tachyarrhythmia and HF always raises a "chicken or egg" question.
The CAMERA-MRI study [19] reported the use of cardiac magnetic resonance imaging with late-gadolinium enhancement as a screening tool to predict LVSD reversibility before treatments. Absence of a ventricular scar or scar burden < 10% were predictors of the reversibility of LVSD.
TCM is a diagnosis of exclusion and should be primarily suspected in patients with nonischemic cardiomyopathy and persistent AA who do not improve after appropriate medical therapy and rate control.
Some clinical trials have shown that the CA strategy for AA resulted in early improvement of LVSD, evident at 1 month and sustained at 1 year [7,8]. This was accompanied by reversal of LV remodeling as evidenced by the reduction in ESV compared with the medical group. CA resulted in improved exercise capacity, BNP, NYHA class, HF symptoms, and quality of life compared with a medical rate control strategy [7,8,20,21]. Restoration of SR should be considered when TCM due to AA is suspected. Importantly, appropriate diagnosis and treatment of AA will improve not only LVSD with its associated morbidity and mortality, but also HF symptoms, quality of life and long-term prognosis in most patients [8,22,23].
We demonstrated that the restoration and maintenance of SR was achievable in up to 84.6% in patients with AA and LVSD. Previous studies have reported that AADs have an overall 30-50% success rate to maintain SR, with frequent discontinuation due to side effects [18]. In contrast, the CA strategy has been reported to achieve SR from 50-88% in AA patients with HF [8,18]. The achievement of SR in the present study was relatively high with treatment for persistent AA as compared to previous reports. In this study, we performed aggressive rhythm control therapy combined with CA and AADs. The results of our study were in accordance with findings from previous studies [7,8,24].
Current guidelines recommend ICD implantation for patients with severe LVSD (EF < 35%) to prevent SCD [11,12]. If guidelines were strictly complied with, 48% of patients in the present study would have been considered candidates for ICD. After rhythm control therapy, EF was improved to > 35% in all patients with severe LVSD, and no patients in this study suffered from SCD. As indications for cardiac device implantation must be determined with utmost care, careful observation after rhythm control therapy is necessary when the etiology of LVSD is suspected to be TCM.
One study identified enlargement of LVDd to > 60 mm as a predictor of irreversible LVSD [13]. Enlarged LVDd may reflect severe left ventricular structural remodeling. Recently, Stegmann, et al. reported that opposite LVEDV index and negative late gadolinium enhancement in cardiac magnetic resonance imaging were independent predictors of complete LVEF recovery [25]. The present study included 7 cases with LVDd > 60 mm (21%). Rhythm control therapy could restore SR and EF was then improved even in that population of cases with severely enlarged left ventricle. In this study, we assessed the left ventricular systolic function revealed with echocardiography. Compared to cardiac magnetic resonance imaging, echocardiograpy can be easily achievd in daily clinical practice. Among cases of LVSD and AA with severe enlargement of LVDd, that dimension alone is not a predictor of irreversible left ventricular systolic function. Restoration of SR should be considered when TCM is suspected.
Several limitations to this study should be acknowledged. First, this study was a retrospective, observational study of patients from a single institute and was not a randomized study. Second, we demonstrated that LVESV and EF were improved after aggressive rhythm control therapy, whereas no reduction in LVEDV was shown. One possibility is that the observation period was too short to observe reverse remodeling after restoration of SR. Although we did not refer to the clinical course such as total mortality or admission for worsening of HF, we could refer to statistical improvements in EF and BNP levels as important clinical parameters. Larger studies are required to validate these initial results.
Left ventricular systolic function improved after aggressive rhythm control therapy in patients with AA and LVSD. Unnecessary cardiac device implantation may be avoidable in this population. Even in cases with severely enlarged LVDd > 60 mm, LV systolic function appears reversible when AA is present.
The authors declare that there is no conflict of interest.