Background/Objectives: Alopecia Areata (AA) is a chronic autoimmune disorder causing sudden, non-scarring hair loss due to hair follicle inflammation. The extent and severity of hair loss varies from smooth, localized patches on the scalp to recurrent episodes of total body hair loss. AA is associated with systemic and psychiatric comorbidities, disproportionately impacting children of color. Management guidelines for AA in pediatric patients of color remain limited.
Methods: We present a case series of five Black pediatric patients diagnosed with moderate to severe AA, treated between 2019-2025, using topical janus kinase inhibitors (JAKi), oral fexofenadine, and vitamin D supplementation. In two of these cases, additional treatment modalities including oral upadacitinib, platelet-rich plasma (PRP), red light therapy (RLT), and excimer UVB phototherapy, were incorporated alongside the aforementioned treatment regimen. Clinical outcomes were evaluated by calculating Alopecia Areata Investigator Global Assessment (AA-IGA) scores for each patient throughout their treatment.
Results: Significant reductions in disease severity were observed across all subjects. Adverse side effects were limited to site erythema and tenderness associated with excimer-308nm targeted UVB phototherapy. No other adverse side effects were reported. All five patients showed substantial hair regrowth and attained AA-IGA scores ≤ 1, indicating minimal or no disease activity.
Conclusion: This case series contributes to existing evidence on the efficacy of JAK inhibitors (JAKi) for the treatment of pediatric AA, and supports the use of topical JAKi as a viable alternative for pediatric patients who may have contraindications to corticosteroids, immunosuppressive agents, or oral JAKi. Combination therapy with topical JAKi, vitamin D supplementation, antihistamines, and other interventions may play a crucial role in reducing active disease progression and maintenance therapy is essential in preventing relapse.
Pediatric alopecia areata, Antihistamines, JAK inhibitors, Vitamin D, African American hair loss, Skin of color, Light therapy.
Alopecia areata (AA) is a chronic autoimmune disorder characterized by hair follicle inflammation, leading to non-scarring hair loss, typically appearing as well-demarcated bald patches on the scalp. In severe cases, it may advance to alopecia total is (AT), or alopecia universalis (AU). AA arises from the collapse of hair follicle immune privilege (HF-IP), which normally prevents inflammatory responses. Genetic factors and environmental factors such as emotional or physical stress, viral infections, and vaccinations can lead to an attack on the hair follicle’s natural immune privilege by inhibiting the production of two anti-inflammatory cytokines–transforming growth factor-β (TGF-β) and α-melanocyte–stimulating hormone (α-MSH)-and activating natural killer (NK) cells (namely CD8+ NKG2D+ T cells) [1,2]. Activation of NK cells prompts secretion of interferon-γ (IFN-γ) (via JAK1 and JAK3 pathways) which enhances the production of interleukin-15 (IL-15) in follicular epithelial cells (via JAK1 and JAK2 pathways) [1]. IFN-γ triggers the collapse of HF-IP, exposing autoantigens to CD8+ NKG2D+ T cells and initiating a positive feedback loop that drives the autoimmune attack on hair follicles [1,2].
AA has a lifetime risk of approximately 2%, yet it is the third most common dermatologic presentation in children with a higher disease prevalence among Black children compared to White children [3,4]. The onset of AA can be sudden and unpredictable, causing significant psychological distress. [2-6] Studies have shown that 13% of children and adolescents with AA are particularly susceptible to the psychosocial consequences related to fear of societal judgement due to their hair loss [7]. Flare ups can occur as stress causes an activation of the hypothalamic-pituitary-adrenal (HOA) axis leading to secretion of corticotropin-releasing hormone (CRH) and substance P (SP). These neuropeptides disrupt HF-IP [7,8].
Despite its prevalence and negative impact among Black children, there is limited literature and no FDA-approved treatment protocols on the management of AA in pediatric patients under the age of 12, particularly patients of color. Our findings present potential treatment regimens for AA in children, particularly the importance of maintenance therapy long after initial disease remission.
An electronic medical record review was conducted on pediatric patients aged 3-10 diagnosed with moderate to severe AA and treated between 2019 and 2025. Table 1 displays the notable baseline laboratory results. Primary interventions included topical JAK inhibitors, oral fexofenadine, and vitamin D supplementation. Details of each intervention and treatment response are detailed in Table 2. Photographs were taken using an iPad (9.7-inch, 5th Generation, 2017 model; Apple Inc., Cupertino, CA, USA). Diagnosis was determined via physical exam, HPI, and trichoscopy by the dermatologist. The Alopecia Areata Investigator Global Assessment (AA-IGA) scores [1] were calculated for each patient both at baseline and at the treatment endpoint.
Table 1: Notable baseline lab results. View Table 1
Table 2: Patient demographics, clinical presentations, treatment regimens, and therapeutic responses. View Table 2
1 The reference range column in this table accounts for age-specific differences to better reflect normal values for patients 1-5 based on their stage of development [9].
1 Intralesional Corticosteroids (ILCs) refers to the number of clinic visits where patients received triamcinolone acetonide injections for new or active patches of hair loss. Kenalog-10 was diluted to 2-5 mg/mL, with dosages tailored to individual needs. Topical lidocaine was applied an hour prior. The scalp was cleansed with alcohol, and sterile ice packs were provided to reduce discomfort. A 30-gauge needle was used to inject small aliquots of up to 1 mL solution diffusely within the affected areas.
2 Daily vitamin D supplementation was standardized, with all patients receiving the same brand of Vitamin D3 chewable dietary supplement, which indicates 500 IU per serving.
3 Treatment Duration refers to the time from the initial visit to disease remission, defined as the absence of recurrences for at least 3 months. Note that disease remission does not necessitate full regrowth, which may require additional time.
An 8-year-old Black girl presented with alopecia areata (Figure 1A) and joint pain that began one month after influenza infection and oseltamivir treatment. Baseline labs showed normal vitamin D (39 ng/mL), negative ANA, normal TSH (2.23 mlU/L), and normal ferritin (32 ng/mL) levels (Table 1). Initial treatment included topical tofacitinib, topical fluocinolone oil, oral fexofenadine, and vitamin D supplementation (Table 2). Partial hair regrowth was observed at 3 months and complete regrowth at 6 months. After one year, two new AA patches appeared on her posterior scalp. She was restarted on topical tofacitinib, fluocinolone oil, fexofenadine, and vitamin D. Significant hair regrowth occurred within 8 weeks, and she continued a consistent maintenance regimen (Table 2). Her AA remains in remission with this topical regimen (Figure 1B).
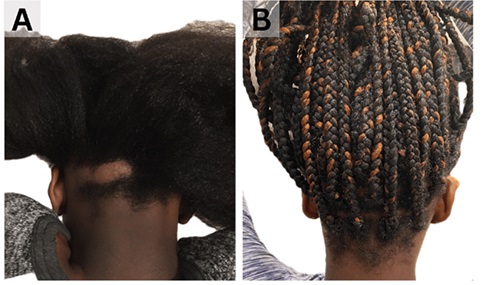 Figure 1: Patient 1 before and after completion of therapy.
Figure 1: Patient 1 before and after completion of therapy.
A. Before: AA-IGA=2, moderate
B. After: AA-IGA=0, None
View Figure 1
A 3-year-old Black girl with a 14-month history of alopecia universalis (Figures 2A, Figure 2B) presented after failure of several first-line therapies (Table 2). Examination revealed diffuse hair loss involving approximately 90% BSA, including total hair loss of the eyebrows and eyelashes. Baseline labs revealed vitamin D insufficiency (27 ng/mL) and negative ANA (Table 1). Initial treatment included topical tofacitinib, oral fexofenadine, and oral vitamin D supplementation. Weekly oral dexamethasone was added due to minimal regrowth at 8 weeks. Significant regrowth occurred over 4 months and tapered discontinuation of dexamethasone was initiated. Full regrowth was achieved at 10 months (Figure 2C) and she transitioned to maintenance therapy. She remained in remission for 3.5 years until discontinuation of all medications by the patient. Six months later, a single AA patch recurred. She was restarted on vitamin D, fexofenadine, and topical tofacitinib/fluocinolone ointment. Full regrowth was achieved within 8 weeks and has since sustained remission with maintenance therapy (Table 2).
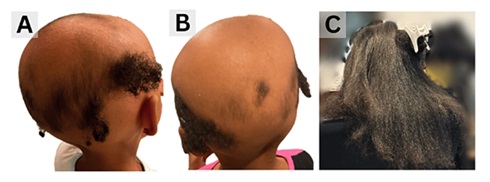 Figure 2: Patient 2 before and after completion of therapy.
Figure 2: Patient 2 before and after completion of therapy.
A. Before: AA-IGA=3, severe
B. Before: AA-IGA=3, severe
C. After: AA-IGA=0, None
View Figure 2
A 5-year-old Black girl presented with a 12-month history of treatment-resistant alopecia universalis with 100% BSA involvement (Figure 3A, 3C). Examination revealed complete eyebrow hair loss and severe, patchy AA with sparse, inconsistent scalp regrowth. Baseline labs showed low-normal vitamin D (32 ng/mL), negative ANA, normal TSH (0.84 mIU/L), and normal ferritin (18 ng/mL) levels (Table 1). She started oral vitamin D supplementation and fexofenadine. Topically, she used fluocinolone oil, a minoxidil/tofacitinib/fluocinolone ointment, and continued tofacitinib to her scalp (Table 2).
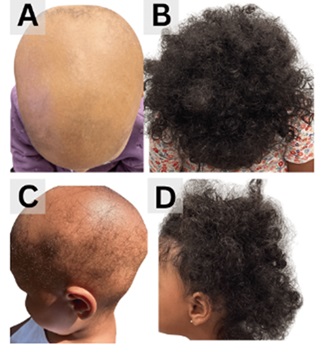 Figure 3: Patient 3 before and after completion of therapy.
Figure 3: Patient 3 before and after completion of therapy.
A. Before: AA-IGA=4, Very severe
B. After: AA-IGA=1, Limited
C. Before: AA-IGA=4, Very severe
D. After: AA-IGA=1, Limited
View Figure 3
After 6 months, scalp regrowth was observed in several areas, but there was no eyebrow regrowth, and new alopecic patches continued to develop on the central and frontal scalp. She underwent one ILCS treatment, which was discontinued due to pain. She also started oral dexamethasone and biweekly sessions of excimer-308nm targeted UVB phototherapy (Table 2). The patient noted erythema and slight tenderness on the scalp for less than 24 hours after a small number of sessions, but this is a common side effect of excimer treatment and this does not affect the course of treatment. The patient responded positively and dexamethasone is being tapered for discontinuation. Although she continues to develop new alopecic patches, disease progression has significantly slowed with continued weekly excimer treatment and topical use, and eyebrow hair growth was noted for the first time since the initial onset of disease (Figure 3B, Figure 3D).
A 7-year-old Black girl presented with a 5-month history of progressive patchy hair loss (Figure 4A, Figure 4C). Examination revealed multiple AA patches concentrated in the occipital and bilateral temporal scalp, consistent with an ophiasis pattern. Baseline labs were significant for vitamin D insufficiency (23 ng/mL), negative ANA, normal TSH (1.43 mIU/L), and normal ferritin (32.3 ng/mL) levels (Table 1). Initial treatment included topical tofacitinib, topical fluocinolone oil, oral fexofenadine, and vitamin D supplementation (Table 2). Significant hair regrowth was observed within 6 weeks, and complete hair regrowth was achieved within 6 months of treatment (Figure 4B, Figure 4D). She was started on a maintenance regimen (Table 2) and remains in remission.
 Figure 4: Patient 4 before and after completion of therapy.
Figure 4: Patient 4 before and after completion of therapy.
A. AA-IGA=2, moderate
B. AA-IGA=1, limited
C. AA-IGA=2, moderate
D. AA-IGA=1, limited
View Figure 4
A 10-year-old Black boy initially presented with superinfected atopic dermatitis (AD) which was treated with topical mupirocin and triamcinolone. Three months later, he returned with progressive hair loss patches on the posterior and bilateral temporal scalp (Figure 5A, Figure 5C) and joint pain. Baseline labs revealed positive ANA (Titer: 1:40, pattern: nuclear, speckled), normal vitamin D levels (38 ng/mL), normal TSH (3.29 mIU/L), and normal ferritin (34 ng/mL) levels (Table 1). He was initiated on topical tofacitinib, topical fluocinolone oil, oral fexofenadine, and oral vitamin D supplementation (Table 2). Triamcinolone acetonide was injected into alopecic patches at 6-week intervals for several months. The temporal scalp responded positively to this regimen, however, disease progression accelerated on the posterior scalp. Weekly pulse dexamethasone dosing was added, and ILCs were performed bi-monthly. This regimen resulted in complete hair regrowth. He transitioned to maintenance therapy after 7 months (Table 2).
 Figure 5: Patient 5 before treatment and after two years of continued therapy, preceding initiation of oral upacitinib, which ultimately resulted in sustained remission and a final AA-IGA score of 0.
Figure 5: Patient 5 before treatment and after two years of continued therapy, preceding initiation of oral upacitinib, which ultimately resulted in sustained remission and a final AA-IGA score of 0.
A. Before: AA-IGA=3, severe
B. After 2 years: AA-IGA=1, limited
C. Before: AA-IGA=3, severe
D. After 2 years: AA-IGA=1, limited
View Figure 5
After three months of remission, the patient returned with new patches on the posterior, occipital, and temporal scalp. ILCs was resumed, now on a bi-monthly basis, and his topical regimen was modified to include fluocinolone oil and a topical tofacitinib/minoxidill compound ointment (Table 2). After 5 months, nearly full hair regrowth was observed, except on the posterior scalp. Platelet Rich Plasma (PRP) treatment and LED low-level light phototherapy were initiated (Table 2).
Over the next 6 months, the patient completed 5 rounds of PRP, followed by 30 minutes of low-level light therapy by application of red light (RLT). The patient maintained a home regimen of vitamin D supplementation, oral fexofenadine, topical fluocinolone oil, topical ruxolitinib cream, and topical tofacitinib/minoxidil ointment (Table 2). During this period, disease progression stabilized, but the patient continued to experience perpetual relapses and remissions, although with reduced severity (Figure 5B, Figure 5D). Once he met the minimum age requirement for oral JAK inhibitor treatment, he began daily oral upadacitinib in addition to his topical regimen. Complete regrowth of hair was achieved within 4 months. He remains in remission with maintenance topical therapy and oral upadacitinib. ILCs is performed as needed at eight-week intervals.
Conventional therapies for alopecia areata in pediatric patients include intralesional and topical corticosteroids, topical immunotherapy, topical minoxidil, and systemic corticotherapy [3]. However, due to AA’s non life threatening nature and adverse side effects from these treatments, especially in children, alternative therapies warrant evaluation. These include JAKi, UVB phototherapy, excimer laser, and PRP- each with the potential to reduce this disease burden and promote standardized treatment guidelines.
While oral JAK inhibitors have shown high efficacy in open-label clinical trials, they can be linked to growth related systemic adverse effects in children [1,10-13]. With the exception of recently approved oral ritlecitinib for ages 12 years and older, systemic JAKi for pediatric AA are currently limited to off-label use and may be cost prohibitive. Topical JAKi therapies show promising results for hair regrowth and should be considered as a first-line option for patients with contraindications to oral JAKi. A systematic review on the effectiveness and safety outcomes of topical JAKi therapies for AA reported trends in Severity of Alopecia Tool (SALT) score measurements. Tofacitinib resulted in 32.2% of cases achieving ≥50% (SALT50) reductions from baseline SALT [10]. In our case series, all patients treated with topical tofacitinib 2% ointment experienced hair regrowth with no adverse events reported.
Vitamin D supplementation may play a role in treating AA by inhibiting proinflammatory processes, primarily through the inhibition of Th17 cells [14]. AA patients with higher Fitzpatrick skin types have a significantly higher prevalence of 25-hydroxyvitamin D insufficiency and deficiency compared to healthy controls in addition to decreased Vitamin D levels overall [6,14,15]. Our patients presented with low-normal or insufficient vitamin D levels, and they all received oral vitamin D supplementation as part of their treatment regimen.
Antihistamines, such as fexofenadine, may also benefit pediatric AA patients. Lee, et al., found that patients receiving adjunct antihistamines showed greater hair regrowth than those treated with corticosteroids and cryotherapy alone [16]. Antihistamines suppress Th1/CD8+ and Th2/IL-4 pathways, reducing perifollicular mast cells [17]. All our patients received fexofenadine and showed hair regrowth. H1-antagonists should be considered, especially in patients with comorbid atopic dermatitis [1,16-19].
Photobiomodulation therapy (PBMT) may serve as an alternative treatment option for AA due to its anti-inflammatory properties. One case study showed significant hair regrowth with continuous wave-red laser (660nm) in a 31-year-old patient with mild AA [20]. Limited data exist on red light therapy in the pediatric population or in patients of color. Recent studies have reported 308 nm excimer light (EL) as an effective therapy against patchy AA. EL has cutaneous immunosuppressive properties via UVB triggered T-cell apoptosis, resulting in reduced perifollicular inflammation and hair follicle damage [21]. When compared to intralesional corticosteroids (ILCS), 380 nm EL is equally effective at promoting hair regrowth but may have delayed effects compared to ILCS [22,23]. It provides an alternative for pediatric patients with needle phobia, pain, and other contraindications to ILCS.
This case series details the successful multimodal treatment approach of five Black pediatric patients with AA using a combination of therapies, including topical JAK inhibitors, vitamin D, antihistamines, low-potency topical steroid oil, and alternative approaches to intralesional corticosteroids such as low-level red light and excimer 308 nm light therapy. Additionally, a maintenance protocol is imperative to prevent or minimize disease recurrence. All patients followed a maintenance regimen of vitamin D supplementation, fexofenadine, fluocinolone oil, and preventative application of topical JAK inhibitors as needed.
Given that children of color often present with more severe disease and poor outcomes compared to their white counterparts, further research is imperative to better understand the nuances of AA in this demographic. Moreover, the inclusion of vitamin D, antihistamines, and topical JAK inhibitors in treatment protocols should be strongly considered for managing AA in pediatric patients of color.