The incidence of and death rate from cutaneous melanoma (CMM) continue to increase. Prior data suggests that more than half of CMM are detected by patients or their close contacts and that these patient-detected melanomas (PDM) present at higher stage than those found by physicians. As outcome is directly related to stage at presentation we undertook this study to identify strategies to improve timely patient detection of early CMM.
We reviewed prospective questionnaire data obtained at surgical oncology consultation along with demographic and pathology information on 488 CMMs in 435 patients. Data analysis with performed with SAS statistical software.
248 CMM (51%) were PDM, 34% were diagnosed by non-dermatologists and 15% by dermatologists. Factors associated with patient detection included female sex, younger age, non-Caucasian race, no prior skin cancer, non-truncal site, anterior location and nodular or acral lentiginous histology, but not family history or CMM diameter. The main symptoms prompting patients to seek care were change in size (32%), color (31%) and bleeding (10%). PDM was associated with higher tumor level and thickness (p < 0.005). Median tumor thickness was 1.07 mm for PDM versus 0.61 mm for physician-detected melanoma (MDM), p < 0.0001. Two-thirds of CMM > 2 mm in thickness and 85% of T4 melanomas were PDM. Patient-detected disease was more frequently ulcerated and lymph node-positive (p < 0.05). Nodular, acral lentiginous and amelanotic lesions were more often PDM. Of MDM, most were identified by non-dermatologists.
We identified several modifiable barriers that hinder early detection of melanoma. Our data suggest that public health efforts such as targeted screening programs, along with skin examinations during routine primary care visits and novel educational tools might improve awareness among high-risk patients who are unlikely to detect their own CMM, resulting in a meaningful improvement in melanoma outcomes.
Melanoma, Early detection, Screening, Diagnosis, Treatment
Cutaneous melanoma (CMM) is the most lethal form of skin cancer. The incidence of and death rate from CMM continue to rise globally, making it a major public health concern [1-3]. In 2016 in the USA alone, an estimated 76,380 new cases will be diagnosed and more than 10,130 deaths will be attributable to melanoma [3]. Prognosis is directly related to tumor thickness and stage at presentation. Tumor thickness is the dominant predictor of outcome for clinically node-negative melanoma patients along with sentinel lymph node status [4-7]. Ten-year survival rates of greater than 90% are reported for tumors < 1.00 mm in thickness whereas ten-year survival drops to 50% for tumors > 4.00 mm in thickness [4].
Patient-detected melanomas (PDM) are reported to present at a more advanced stage than those found by physicians (MDM). Most prior reports show that early stage CMM is more likely to be detected by physicians than by patients [8-13]. Evidence suggests that detection of early-stage CMM increases in direct correlation with the number of dermatologists or primary care providers per population unit [14]. However, the majority of CMM still appear to be detected by patients or their close contacts. The proportion of cutaneous melanomas reported to be patient-detected ranges from 40 to 80% [9,13,15-17].
Since early stage melanoma is easily treated and has a favorable prognosis, efforts to improve patient detection of these early lesions and focus physician screening to those at greatest risk of harboring an undetected melanoma are desirable. Thus, we undertook this study with the aim of investigating how melanomas are detected to identify factors associated with early versus later stage diagnosis. Such information can identify high-risk groups and strategies to improve timely detection of melanoma, which has implications for both individual patient care and public health.
With IRB approval, we reviewed data prospectively recorded in our Melanoma Patient Registry from 2002 to 2011 on 488 newly diagnosed melanomas in 435 patients. At the time of the initial surgical oncology consultation, each patient completed a questionnaire regarding melanoma risk factors, symptoms (if any), medical history, family history and how their melanoma was identified. The questionnaire was reviewed with the patient by the surgical oncology team and data clarified, if necessary. Histopathology and treatment data were confirmed by review of pathology reports and operative notes.
Patient and tumor characteristics were assessed using univariate logistic regression, the Pearson's chi-squared test and the Student's t-test. Data are presented as proportions (percentages), medians with Interquartile Range (IQR) or means ± standard error of the mean unless otherwise stated. A SAS statistical package (JMP 10.0, Cary, N.C.) was used for data analysis. P values of < 0.05 were considered significant.
In our patient cohort, 51% of CMM (n = 248) were discovered by the patient or their close contacts. Of the remainder, non-dermatologists diagnosed 34% of cases (n = 167) while dermatologists diagnosed 15% (n = 73).
As shown in Table 1, patient factors associated with self-detection of melanoma included female sex, younger patient age and non-Caucasian race. Individuals with a prior history of non-melanoma skin cancer were more likely to be diagnosed by physicians. There was no effect of family history of melanoma on mode of detection.
Table 1: Summary of patient characteristics by method of detection. View Table 1
The proportion of PDM increased linearly with melanoma T-stage such that 62% of in situ lesions were diagnosed by physicians whereas 85% of T4 lesions were detected by patients, p < 0.0001 (Figure 1). Of all melanomas > 2 mm in thickness, 67% (97/145) were PDM. The median tumor thickness of invasive PDM in males was 2.5 mm versus 1.38 mm in females, p = 0.06. Tumor features associated with self-detection of melanoma included anatomic site and anterior versus posterior location; nodular and acral lentiginous melanomas also were more likely to be patient-detected, as were ulcerated, amelanotic and node-positive melanomas (Table 2). Mode of detection did not correlate with lesion diameter.
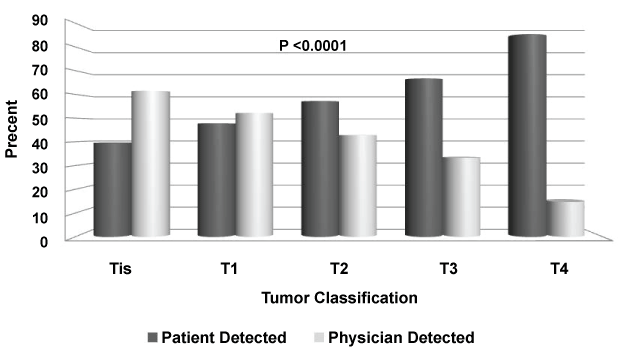 Figure 1: Mode of melanoma detection by T-stage classification at diagnosis. View Figure 1
Figure 1: Mode of melanoma detection by T-stage classification at diagnosis. View Figure 1
Table 2: Summary of tumor characteristics by method of detection. View Table 2
Among 248 PDMs, the most common primary symptom which prompted medical consultation was a change in lesion size in 80 (32.2%), followed by a color change in 76 (30.6%) and bleeding in 24 (9.7%). Less frequent presentations were notation of a new pigmented lesion in 20 (8.1%), change in thickness in 19 (7.7%), change in shape in 18 (7.3%), itching in 8 (3.2%), non-healing skin ulceration in 2 (0.8%) and pain in 1 (0.4%).
In our cohort, just over half of cutaneous melanomas were detected by patients. These PDMs were thicker and more often ulcerated and node-positive, i.e. of more advanced stage at diagnosis, than MDMs. Furthermore, PDMs in males were thicker than PDMs in females and the majority of PDMs (74%) were in patients aged 50 years and older. Other contemporary reports also show a persistently high proportion of self-detected melanomas, especially among older men, and an association between PDM, advanced stage and poorer prognosis despite decades of public health efforts aimed at both primary prevention and early detection [9,10,15,17,18]. These data suggest that greater melanoma awareness among primary healthcare providers and improved primary physician screening is likely to be the most effective method for earlier detection of melanoma, as physicians detect significantly thinner lesions. A recommendation for routine total skin examination at the time of primary care physician visits, especially for males, should be considered. Evidence of a benefit to such routine skin cancer screening is suggested by population-based studies [19].
Among physician-detected melanomas, non-dermatologist physicians identified the majority (68%) of lesions, while dermatologists detected thinner and otherwise favorable melanomas in accordance with several prior reports [9,11,12,20-21]. Roetzheim, et al. found that for each additional dermatologist per 10,000 population, the odds of early melanoma diagnosis increased by 39%. Similarly, for each additional family practitioner, the odds increased by 21% while they decreased by 10% per additional general internist [14]. Grange, et al reported that although general practitioners played a pivotal role in melanoma detection, the melanomas they found were significantly thicker than those identified by other physicians (on average 2.05 mm versus 1.32 mm, p < 0.01) [22]. Collectively, these data suggest that primary care providers may benefit from additional training to facilitate earlier diagnosis of melanoma, especially among high-risk patients.
Several studies have shown that melanomas diagnosed at the time of scheduled routine total skin examination are thinner, less often ulcerated and less mitotically active than those diagnosed otherwise [17,23]. Utilizing non-physicians as screening authorities has also been suggested. For example, using hairdressers for skin cancer screening outreach has been proposed; however, the efficacy and logistics of this approach remains to be fully defined [24-26]. Use of smartphone applications, including patient-initiated screening tools along with teledermatology and teledermoscopy, have been studied; however, thus far the outcomes of these approaches have been quite variable [27-29]. Simplifying and standardizing self-screening skin examination to improve compliance and enhance earlier self-diagnosis also has been proposed [30].
Anterior and head or neck or extremity (versus posterior and truncal) tumor location have been previously described as factors associated with patient detection and confirmed in our cohort [12]. Anatomically, these areas are more visible to patients than posterior and truncal lesions. We also found that nodular and acral lentiginous melanoma was more often found by patients than lentigo maligna or superficial spreading subtypes. To the best of our knowledge, this has not been previously reported. We also observed that amelanotic lesions were more often discovered by patients than physicians. These findings emphasize the importance of thorough skin exams and underscore the importance of physician evaluation of anatomic areas less frequently visualized or more challenging to inspect by patients themselves. Finally, physician education on detection of the less common histologic subtypes of melanoma is also critical.
Limitations of our study include the use of data from a specialist referral practice. Further studies using national-level data may provide more generalizable findings that corroborate with our results. Strengths of this approach are the level of detail provided on presentation which is difficult to ascertain from large databases. Additionally, some element of recall bias may be present; however, this is likely of little consequence due to the prospective nature of the study questionnaire which was reviewed by the healthcare team at the time of presentation to the surgical oncologist. Despite these limitations, we believe our study adds valuable information on the mode of melanoma detection on stage at diagnosis.
Our study demonstrates that the majority of cutaneous melanomas continue to be detected by patients or their close contacts and that despite great effort to increase awareness of melanoma among the general public, these PDMs still present at a significantly later stage than physician-detected tumors. We report for the first time that amelanotic lesions and nodular and acral lentiginous histology subtypes are more often patient-detected. We also found that dermatologists identified the most favorable lesions, underscoring the benefit of regular skin examinations for, at the least, high-risk patients. However, recognizing that access to specialist care may be limited, education of primary care providers to improve detection of earlier stage disease along with investigation of novel technologies appears warranted.
Here we identified patient and tumor characteristics that can be viewed as barriers to early detection of CMM. Our data support a recommendation for routine screening skin examinations as a component of general healthcare practice. Further, targeted outreach and enhanced screening for those at highest risk of harboring undetected thick melanomas, particularly males aged 50 and older, could improve stage at diagnosis and subsequent mortality from melanoma.
The authors have no conflicts of interest to declare.
None.