In the past few years, a link between periodontitis and Alzheimer's disease was established by a growing number of clinical studies. But one question remains to be answered: How can the periodontitis be implicated in the Alzheimer's disease?
The aim of this work is to make a systematic integrative review of published literature to assess a possible association between Porphyromonas gingivalis (P. gingivalis) and Alzheimer's disease.
A research was realized on PUBMED and PUBMED CENTRAL. Of 79 articles found, only 12 were of interest between 2008 and 2020.
Part of (?) P. gingivalis has been identified in the brain of patients with Alzheimer's disease: Its toxic components (as gingipains and lipopolysaccharides) have been identified and proposed as the cause of production of amyloid plaque and abnormal protein tau, source of the neuroinflammation and neurodegeneration.
P. gingivalis could be a therapeutic target for patients with Alzheimer's disease and simultaneously reduce the incidence and severity of the pathology. Prevention (by dental health care) of P. gingivalis dissemination from site of periodontitis to the brain and inhibition (recent therapeutic approach) of its neurotoxicity could be beneficial for patients with Alzheimer's disease.
Porphyromonas gingivalis, Alzheimer's disease, Vascular dementia, Periodontitis
P. Gingivalis: Porphyromonas Gingivalis; LPS: Lipopolysaccharide; Aβ: Amyloid β; NTF: Intraneural Neurofibrillary Tangle; CSF: Cerebrospinal Fluid; IgG: Immunoglobulin G; BBB: Blood-Brain Barrier; CNS: Central Nervous System; Kgp: Lysine Gingipain; Rgp: Arginine Gingipain; CatB: Cathepsin B Protease; TLR: Tool-Like Receptor; PAR2: Protease Activated Receptor 2; PAMPs: Pathogen-Associated Molecular Pattern
Alzheimer's disease is the most common cause of dementia in elder population [1,2]. Due to ever-increasing life expectancy, Alzheimer's disease is of great public concern. This disease is characterized by a brain disorder, which can alter the memory and cognitive function [3]. It may appear as an early form or as sporadic form, which is the case in 95% of Alzheimer's disease patients [4,5]. Peripheral inflammation is largely considered to contribute to pathogenesis of Alzheimer's disease, as it can increase brain inflammation [6]. Inflammation is detected in disease's brain throughout the presence of pro-inflammatory cytokines such as Interleukin 1-Beta (IL 1-β), Tumor necrosis factor alfa (TNF-α) and Interleukin 6 (IL-6). Two hallmark proteins are representative of the disease and present in the brain of Alzheimer's patients: Amyloid-beta (Aβ) plaque and hyperphosphorylated Tau [2,7,8]. The exact onset of Alzheimer disease is not entirely known, except for the age-dependent risk, but in recent years, the theory of bacterial infection has been placed as a likely responsible for the progression of the disease.
In the past few years, a link between periodontitis and Alzheimer's disease was established by a growing number of clinical studies [9-11]. But one question remains to be answered: How can the periodontitis be implicated in the Alzheimer's disease?
Periodontitis, an oral multi-bacterial disease, is a periodontal low-grade chronic inflammation [11]. This periodontal disease is presumed to aggravate systemic inflammation [5] and is caused by a dysbiosis of oral bacteria, where Porphyromonas gingivalis (P. gingivalis) has a predominante role. Therefore, this review is focused on a major periodontal pathogen P. gingivalis which has been proposed, as risk factor, to contribute to the development of Alzheimer's disease [5,12-14]. The dissemination of P gingivalis from the area of periodontal inflammation to systemic circulation can be induced by common tasks, such as brushing, flossing and mastication, as well as dental procedures like scaling or teeth extraction [9,13,15]. P. gingivalis can lead to important systemic alterations and is implicated in the onset of different systemic pathologies, such as cardiovascular, neurodegenerative pathologies, and rheumatoid arthritis.
The search for an association between P. gingivalis and Alzheimer's disease is the main aim of this review, by understanding the possible ways by which P. gingivalis acts on the brain, and how can a dentists shed new lights on oral management of Alzheimer's patients.
PICO model was used: Patient (with Alzheimer's disease), Intervention (positive for periodontal bacteria, P. gingivalis), Control (patient without Alzheimer disease) and Outcome (association of P. gingivalis in patients with Alzheimer's disease). The built question is the follow: Is P. gingivalis, a periodontal bacterium, associated with Alzheimer's disease? Methods has been inspired from PRISMA protocol by its use of a flow diagram.
A bibliographic search was performed on electronic platform via PUBMED. Different combinations of Keys words were used: "Porphyromonas gingivalis" AND ("Alzheimer's disease" OR "vascular dementia") AND ("periodontitis" OR "periodontitis").
Several kinds of publications were taken in consideration: Meta-analyses, experimental studies, study cases, clinical trials, in vitro and in vivo studies and integrative/systematic reviews. Only articles dealing with the role of P. gingivalis in association with Alzheimer's disease and published, between January 2008 and April 2020, in English or Portuguese were selected. Election of articles was focused on the last 20 years, because there was no researcher interested in this topic before. Only the ones published in full and in digital form were elected. Title, resume and abstract were analyzed in order to assess relevance of each article. Then potentially eligible articles were exanimated by a full text analysis to check if they were of interest for our study.
Inclusion and exclusion criteria were the following:
• Studies explaining association of P. gingivalis in the Alzheimer's diseases were included.
• Studies describing influence of P. gingivalis on the onset of the Alzheimer's disease were included.
• Studies conducted on human and mice were included.
• Article not written in English or Portuguese, before January 2008 were excluded.
• Finally, literature reviews were excluded for results.
After identification of eligible articles through several databases and platforms, selection of relevant articles was performed in order to investigate influence of P. gingivalis on Alzheimer's disease.
Total of articles found (all article selected were compiled in Mendeley citation manager), where duplicates were removed. Full Evaluation of each publication was realized to elect accurate publication, which will meet the purpose of the study. Screening included analyses of title and abstract in order to remove non-relevant articles for this review. After the screening phase, all articles were read in full-length, to assess eligibility for qualitative analysis. Different factors were chosen to evaluate articles selected: Name, authors, years, country, type of study, sample size, part tested, methodologies used, result and conclusion. Elected articles needed to respond to the outcome of this study, reflecting the association between P. gingivalis and Alzheimer's disease.
A total of 79 articles were identified on the PubMed database, between 2008 and 2020. With application of study eligibility criteria (studies about relation of P. gingivalis, major pathogen of periodontitis with Alzheimer's disease), 32 articles were taken into consideration. After screening and application of inclusion and exclusion criteria, 12 articles were included for qualitative analysis. Studies identified but not selected were used later to deepen and extend knowledge on this review subject matter and to enrich discussion. All the procedure is explained in a flow chart procedure (Figure 1).
 Figure 1: Flow diagram-Methodology of the search strategy.
View Figure 1
Figure 1: Flow diagram-Methodology of the search strategy.
View Figure 1
On 12 studies selected, 4 were conducted on humans, 7 were conducted on mice and 1 on both. Extracted data was compiled in tables: Authors, years, ways of P. gingivalis inoculation, sample size, technic used for evaluation, result and conclusion in Table 1 and authors, years, study design, sample size and tested anatomic structure, used technic, result and conclusion for Table 2.
Table 1: Relevant data gathered from the selected studies on mice. View Table 1
Results were collected and organized in following section:
In 50% of surveys, P. gingivalis or its products, Lipopolysaccharide-pg (LPS-pg) and gingipains (trypsin-like cysteine proteinases produced by P. gingivalis) were found in the brain tissue of mice after oral inoculation of P. gingivalis or induction of periodontitis [3,13,15,16]. Three studies demonstrated that expression levels of pro-inflammatory cytokines TNFα, IL-6 and IL-1β in the brain tissues of mice infected orally with P. gingivalis were increased in comparison with experimental groups [3,16,17]. A study conducted on the liver of mice also proved that infection with P. gingivalis promoted the rise of IL-1β laud in liver macrophages of P. gingivalis contaminated mice [18].
In 62.5% of the researches on mice, infection by P. gingivalis also induced increase of amyloid β in the brain [1,3,13,16,18]. In one study, Dominy, et al. colocalized hyperphosphorylated Tau protein with gingipain and showed that Tau was a target of gingipain proteolysis capacity [13].
Mice's behavior was evaluated in four trials: 3 of the studies pointed a cognitive function impaired in the P. gingivalis infected mice [3,10,17]. One study showed that continuous brain exposure of Pg-LPS didn't enhance cognitive impairment in Alzheimer's disease model mice [14].
As shown on Table 2, five studies with human subjects were compared in this survey. In a prospective pilot study performed by Dominy, et al. [13], authors took samples of Cerebrospinal fluid (CSF) in patients with clinical Alzheimer's disease and collected matching saliva of each patient. All of the 10-saliva samples were positive to P. gingivalis and 7 of the 10 samples of CSF were significantly positive with P. gingivalis. In the most recent study performed on humans, authors found a possible association between P. gingivalis and Alzheimer's disease: 5 of 7 patients with Alzheimer's disease and periodontitis stage III or IV presented a strain of periodontal bacteria P. gingivalis. These positives to P. gingivalis patients, manifested lower score (with p < 0.05) in cognitive test [5]. Three studies (75% of researches conducted on human) had concerned themselves with levels of P. gingivalis in serum IgG antibody [19-21]. They found that periodontitis pathogen, P. gingivalis, is linked to poor cognitive performance. Subjects with a higher load of P. gingivalis IgG were more likely to have a bad delayed verbal memory and impaired subtraction, with apparent dose-response (the more P. gingivalis in IgG, the poorer cognitive performance) [19]. Stein, et al. [20] found out that antibodies level to P. gingivalis were elevated in Alzheimer's disease patients (same level before and after onset of Alzheimer's disease): 39 μg/ml against 22 in control patients (without cognitive disease). Antibodies high level in cohort of patients with Alzheimer's disease were similar to serum antibodies level in population of patients with clinical periodontitis: 43.1 μg/ml ± 6.4 in comparison with healthy patients: 8.2 μg/ml ± 0.9.
Table 2: Relevant data gathered from retrieved studies on humans. View Table 2
P. gingivalis is a key bacterium of periodontitis, a-saccharolytic gram-negative anaerobe. It survives in periodontal pockets, a major sign of this periodontal pathology [22-24]. P. gingivalis is part of the red Socransky complex of biofilm's bacteria [11,22]. Some systemic pathologies might be associated with P. gingivalis, such as cardiovascular disease, neuronal degeneration, diabetes, arthritis [14,23,24]. P. gingivalis was found in brain tissue and in CSF [13], providing definitive evidence of its migration from oral cavity to the brain [15,25]. The mechanism of P. gingivalis from a peripheral infection (periodontitis) until the brain is a complex process, which involves several pathways: Stimulation of peripheral nerves and blood dissemination or by action of LPS (lipopolysaccharide) in site lacking Blood-brain barrier (BBB) as in circumventricular organs, either direct action of LPS from systemic circulation on vascular cell of BBB [1,6,14]. Once past the BBB [5], P. gingivalis is going to induce neuroinflammation through different means, which is going to participate to pathogenesis of neurodegenerative disease as Alzheimer's disease [1,14].
P. gingivalis have major toxic virulence factors localized in its outer membrane, such as gingipain, LPS and fimbria [14,15,22]. These toxins induce inflammation by promoting production of pro-inflammatory cytokines [3]. Gingipains, which are cysteine proteases, are highly potent modulators of host colonization, inactivation of host defenses and tissues destruction [13]. Gingipain have been found in brain of young mice after oral infection at P. gingivalis [16] and in brain of individuals with Alzheimer's disease [13].
Two types of gingipains are of interest: Lysine gingipain (or Kgp) and arginine gingipain (or Rgp). These gingipains contribute, after infiltration in the brain, to cell migration of microglia towards infected site and lead to neuroinflammation. Gingipains activates PAR2, a cell-receptor activated by protease, which plays a role in inflammation. Kgp and Rgp may increase vascular permeability by degrading component of basal membrane and endothelial cell tight junction proteins and so contribute to loss of blood-brain barrier functional integrity [12,26,27].
LPS, components of virulence on P. gingivalis outer membrane, also induce increase of proinflammatory cytokines, activating CNS innate immune system and therefore contributing to neuroinflammation and neurodegeneration in Alzheimer's disease [3,4] (Figure 2). LPS, using Pathogens associated molecular patterns (PAMPs), can activate Toll-like receptor (TLR) present in surface of glia cells, such as microglia. This activation LPS/TLR lead to an antibacterial response [1,4]. In a post-mortem study, p.g-LPS was identified in Alzheimer's disease brain but not in control case without cognitive troubles [25]. P.g-LPS were also found in mouse brains and serum after inoculation of P. gingivalis in order to create experimental periodontal disease [3], supporting the theory that P. gingivalis may play a role in Alzheimer's disease.
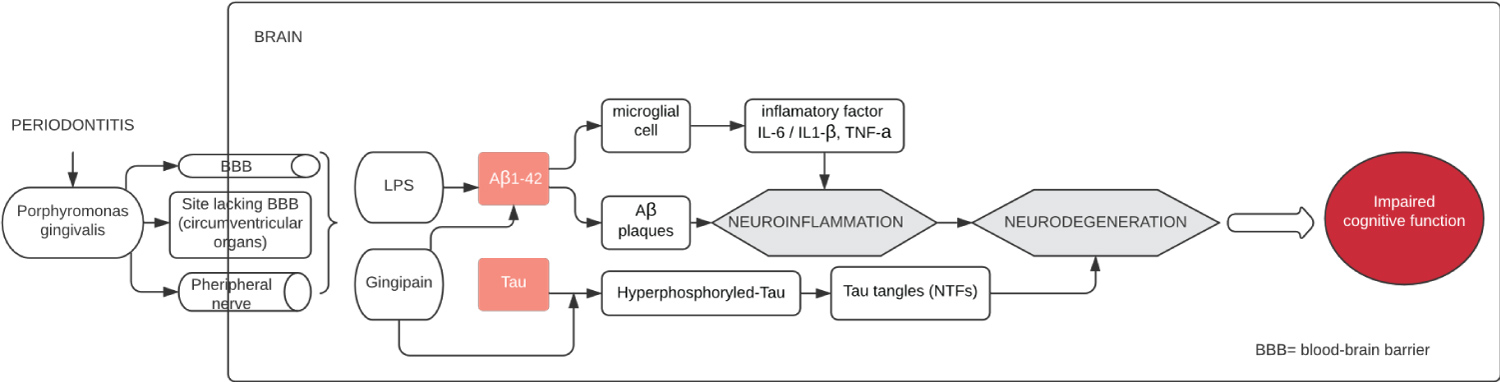 Figure 2: Model for P. gingivalis contribution to Alzheimer's disease.
View Figure 2
Figure 2: Model for P. gingivalis contribution to Alzheimer's disease.
View Figure 2
Moreover, p.g- LPS and gingipains can suppress deposition of opsonons (IgG, C3b, C5b9) on the bacterial cell surface. Blocking C3, directly implicated in elimination of pathogens, and on which all complement pathway converges, could allow for infection to take hold [8]. However, one study did not show a positive association between an injection of LPS and onset of Alzheimer's disease in healthy individual. This outcome could be due to too short exposure of p.g-LPS. It was concluded, however, that a continuous injection of LPS may worsen the prognosis of a patient already suffering from Alzheimer's disease [14].
In one of the first studies conducted in 2009, authors concluded that patients with a concentration of IgG's P. gingivalis in serum had poorer verbal memory and subtraction performance test scores than individuals without P. gingivalis [19]. A longitudinal study confirmed the previous one by identifying an association between serological markers of periodontitis (of which P. gingivalis is a part) and Alzheimer's disease, via the study of IgG antibody serum [21].
The patients studied by Sparks Spein, et al. expressed, before the clinical diagnosis of Alzheimer's disease, a significant level of P. gingivalis antibodies, supporting hypothesis of an association between P. gingivalis from periodontitis and onset of Alzheimer's disease [20].
In a study conducted in 2020 on 20 patients with possible Alzheimer's disease, a significant association was observed between P. gingivalis and low Mini mental state examination (MMSE) score and Clock drawing test (CDT) score, meaning that P. gingivalis may play a role in cognitive function impairment. Although this study is limited by the small number of subjects, it pointed like the previous studies to an association between P. gingivalis and Alzheimer's disease [5].
Two types of lesion are characteristic of Alzheimer's disease on a histologic plane: One lesion extracellular, known as amyloid plaque or dense deposits of beta-amyloid peptide (Aβ 1-42) and hyperphosphorylated protein Tau, intracellular, which aggregated in intraneural Neurofibrillary tangle (NTF). Ultimately these two typical features lead to cerebral dysfunction and loss of neurons [2]. Gingipains, which happens to fragment Tau were colocalized with Tau tangles (NTF) and ubiquitin. It turns out that fragmentation of Tau may play a role in formation of hyperphosphorylated Tau, participating in neurodegeneration [13] (Figure 2). In the same study, conducted by Dominy, et al. P. gingivalis oral exposure in mice conducted to induction of the stereotypical Alzheimer's disease marker: Aβ 1-42. Aβ, which has antibacterial effect, accumulated itself in reaction to gingipain leading to an Aβ 1-42 deposition in the brain [13].
Following induction of experimental chronic periodontitis by oral chronic application of P. gingivalis, extracellular plaques of Aβ 42 were detected in non-transgenic C57BL/6 mice. In this study authors also show that oral P. gingivalis infection lead to neuroinflammation, NTF production and neurodegeneration in wild-type mice [16].
Ishida, et al. used transgenic mouse model of Alzheimer's disease (APP-Tg mice) to find out whether periodontitis, via P. gingivalis, exacerbates or not features of Alzheimer's disease [3]. Cognitive functions were significantly altered in mice with periodontitis compared with control mice. Amyloid β and pro-inflammatory cytokines IL-β and TNF-α load were higher in APP-Tg mice than in control mice: p.g-LPS could therefore exacerbate Aβ accumulation, promoting neuroinflammation [3]. In the same study, an in vitro neuron cell culture experimentation showed that LPS induce an increase in Aβ42 deposition and promotes production of TNF-α and IL-1β [3]. After exposition at p.g-LPS, wild type middle age mice suffered from Aβ accumulation, augmentation of Cathepin B protease (CatB) in microglia and neuron and increase IL-1β production in comparison with young mice and CatB-/- middle aged mice. Wu, et al. concluded that chronic exposure to pg-LPS induce microglia-mediated neuroinflammation, Aβ accumulation and impairment of cognitive function in middle -aged mice, in a CatB dependent manner [1]. Cat B was also showed to have a critical role in study conducted by Ran Nie, et al. The authors found out that chronic systemic P. gingivalis infection induces accumulation of Aβ in inflammatory macrophage via the activation of CatB and increases of IL-1β and inflammatory response in liver macrophages of middle-age mice. This suggested that P. gingivalis infection induces production of Aβ in inflammatory macrophages which may serve as vehicle for Aβ entrance in the brain. Even if this study was conduct in the liver, it exposes induction of Aβ in inflammatory macrophages via CatB after P. gingivalis infection [18].
In another survey on middle age mice, oral infection with P. gingivalis induced an increase of TNF-α, IL-6 and IL-1β laud, cause of brain inflammation. P. gingivalis can also impair spatial learning and memory abilities in these mice population. However, no significant impact of P. gingivalis was demonstrate on the young mice in this study [17]. In a resume, different studies as much on mice as on humans support association of P. gingivalis, and its components on the two hallmark proteins that can be found in the brain of Alzheimer's disease: Amyloid-beta (Aβ) plaques and hyperphosphorylated Tau [8] leading to neuroinflammation and neurodegeneration.
Alzheimer's patients, due to their impaired cognitive functions, have more difficulties to adhere to daily dental hygiene, thus creating a favorable environment for P. gingivalis [28]. Removing a bacterial focus by stabilizing the periodontitis and thus decreasing possible systemic exposure to P. gingivalis, can help to reduce progression of disease [11,28]. Beside daily oral hygiene, Alzheimer's disease patient should be advised of importance of dental healthcare. Indeed, periodontal health will prevent induction of inflammation and gum diseases such as periodontitis, ambient in which bacteria P. gingivalis is prosperous.
Oral health decreases as cognitive functions deteriorate [29]. In a study conducted by Ide, et al. poor oral hygiene and particularly periodontitis are associated with increase in cognitive decline [30]. Failure to adhere to rules of daily oral hygiene could increase risk of developing dementia by 22% to 65% compared to individuals with normal hygiene [31].
Thus, poor oral hygiene can lead to oral commensal microbial dysbiosis and trigger periodontitis, leading to virulence of pathogen P. gingivalis. Dental care should therefore not be neglected, and maintenance of oral hygiene should be acquired in order to decrease severity of the disease.
As shown in different investigations, P. gingivalis plays a fundamental role in pathogenesis of Alzheimer's disease. Nie, et al. propose CatB as a potential Alzheimer's disease therapeutic target, given its implication in Aβ accumulation [18]. Another study (Dominy, et al.) implicates gingipains from P. gingivalis. Indeed, if gingipains participate to progression of sporadic dementia, its inhibition should participate to an amelioration of Alzheimer's disease patient's state. A small molecule, COR388, targeting lysine gingipains of the P. gingivalis was tested successfully in decreasing the P. gingivalis bacterial load. In other words, this study demonstrates that a treatment with this gingipains inhibitors will reduce accumulation of amyloid β, P. gingivalis brain infection, and slow and even prevent neurodegeneration in the brain [13], providing new lead for possible Alzheimer's disease treatment.
A major part of studies is in favor of a relation between Alzheimer's disease and P. gingivalis confirming the role of bacterial infection in this pathology. P. gingivalis virulence factors such as LPS and gingipain appear to have a major role in induction of Aβ and phosphorylated Tau archetypal markers of Alzheimer's disease, thus causing increase in pro-inflammatory cytokines, source of neuroinflammation leading to neurodegeneration. But even if data collected through this review point to a consistent causative role of P. gingivalis in Alzheimer's disease, it will be relevant to effectuate a prospective study with a larger population to completely and definitively confirm pathogenesis of P. gingivalis in Alzheimer's disease.
Despite the fact that periodontitis varies according to individual susceptibilities, it seems primordial to prevent this infection in order to avoid dissemination of periodontal pathogens to the brain. It's therefore vital to involve in dental care as much the patient as its healthcare staff and family. Therefore, further studies are necessary to validate efficiency, utility, innocuity and then possibility to diffuse this treatment at public scale.
Statement of clinical relevance:
• P. gingivalis could be a therapeutic target for patients with Alzheimer's disease and could thus reduce the incidence and severity of the pathology.
• Prevention (by dental health care) of P. gingivalis dissemination from the site of periodontitis to the brain and inhibition (recent therapeutic approach) of its neurotoxicity could be beneficial for patients with Alzheimer's disease.
Not applicable.
The authors have consented to publish this paper.
Data used for this study can be made available to anyone whom requests it, by contacting Dra. Marta Relvas, Instituto Universitário de Ciências da Saúde, Gandra, Paredes, Portugal Rua Central de Gandra, 1317 PC 4585-116 Gandra, Paredes, Fax/Tel: 00351934190223 E-mail: marta.relvas@iucs.cespu.pt
Not applicable.
This study did not require funding.
Not applicable.
All authors gave their final approval and agree to be accountable for all aspects of the work.
Author B.C. 1: Contributed to conception, design, data acquisition and interpretation, drafted and critically revised the manuscript; Author L-J.P. 2: Contributed to critically revised the manuscript; Author A.B. 3: Contributed to conception, design, and critically revised the manuscript and Author R.M. 4 Contributed to conception, design, and critically revised the manuscript.