Temporal lobe necrosis (TLN) is often seen in nasopharyngeal carcinoma secondary to less frequent other extracranial malignancies. Radiotherapy is one of the most frequently used methods in the treatment of nasopharyngeal carcinoma. This method can have many side effects. One of the side effects associated with high-dose radiotherapy is cerebral necrosis. Cerebral radiation is the sum of the dose of radiotherapy given as the most important risk factor for necrosis. It is often seen within the first 5 years after the completion of treatment. At present, the development of temporal lobe necrosis, radiotherapy planning, and current techniques are less common than their predecessors. The combination of radiotherapy and chemotherapy treatment also increases the risk of cerebral necrosis.
TLN symptoms are a highly variable spectrum. For this reason, the lesion can mimic many diseases. Differential diagnosis of TLN includes intracranial nasopharyngeal carcinoma (NPC). Expansion, second primer intracranial neoplasm, brain metastasis, meningeal spread and brain abscess. On the way to diagnosis, imaging methods can help, but no method is specific. However, magnetic resonance (MR) imaging features commonly seen in radiation necrosis are a soap-bubble-like core and a Swedish cheese-like appearance. In the MR spectroscopy; there is no increase in the colonic peak in the lesion, in the MR perfusion; perfusion increase in perilesional edema and the lack of diffusion restriction is a finding that supports the diagnosis of radiation necrosis. Imaging features can be diagnosed by evaluating with history and clinical findings. Accurate diagnosis and early detection is very important, as it will avoid unnecessary interventions.
We will talk about the diagnosis of nasopharynx carcinoma and bilateral temporal lobe necrosis secondary to radiation at different times in a patient who has completed radiotherapy treatment.
Nasopharyngeal cancer is a remarkable geographic and racial distribution worldwide. It is a rare malignancy with less than 1 incidence per 100,000 population per year [1]. Treatment of nasopharyngeal carcinoma can be quite challenging due to the invasive nature of the tumor and its anatomical proximity to critical structures. In addition to macroscopic dissemination of the tumor, extensive submucosal extension can also be seen. Even with the help of cross-sectional imaging methods, it is often not possible to accurately determine the microscopic size of the tumor. Radiotherapy (RT) is one of the most preferred methods in the treatment of nasopharyngeal carcinoma [2]. All potential involvement foci in radiotherapy should be kept within the target volume. The lower parts of both temporal lobes should be included in the RT field to cover the sphenoid sinuses and the middle cranial fossa, which are the local spread routes [3] Providing maximum tumor control with minimum complications in RT dose and volume adjustment is the most important goal. Possible side effects of RT therapy include xerostomia, sensorineural hearing loss, optic neuropathy and cerebral necrosis.
Radiation-induced temporal lobe necrosis (TLN) was first described by Fisher and Holfelder in 1930 after treatment of the basal cell carcinoma of the temporal region [3]. Generally, NPC is a rare side effect associated with high dose radiation for pituitary adenoma and skull base tumors. The incidence of TLN is 0-24% with conventional fractionation and up to 35% with accelerated fractionation [4,5]. However, due to developments in RT planning and modern techniques, TLN has become more rare in daily practice [5]. Symptoms range from asymptomatic to severe morbidity and often to mortality. TLN is a lesion that mimics primary tumor recurrence, brain metastasis or high grade primary brain tumors. This case report emphasizes the importance of accurate and timely diagnosis of temporal lobe necrosis and the clinical and radiological changes seen during the follow-up period.
A 34-year-old male patient who was diagnosed with NPC three years ago was brought to the emergency room due to syncope. It was learned that he completed his chemotherapy and radiotherapy treatment 2 years ago. On physical examination, glaskow coma score (GCS): 15, pupils isochoric, neurological examination was found to be normal. Cranial CT of the emergency department revealed extensive vasogenic edema and midline shift in the left temporal lobe. The cranial MRI revealed a 43 × 30 × 40 mm T2A hyperintense, heterogeneous, cystic-necrotic area with peripheral contrast enhancement, a classic 'Swedish cheese' mass lesion and wide vasogenic edema around the left temporal lobe. In the midline structures of the brain, 7 mm right hemisphere displacement was observed. High ADC values in diffusion weighted MRI and decreased rCBV values in perfusion MRI were observed in the edema area. Reduced NAA levels and very clear lactate peak were observed in multivoxel 1H MR spectroscopy examination, but there was no significant increase in choline (Figure 1). With these findings, radiation necrosis was diagnosed and radiologic follow-up was performed. In control examinations, it was observed that the lesion observed in the left temporal lobe was markedly regressed and gliosis developed. A one-year follow-up MRI revealed a similar lesion with a similar character that could be consistent with radiation necrosis of the T2A images with large vasogenic edema around the right temporal lobe anterior (T2A images) (Figure 2). Because the heterogeneous contrast enhancement of the lesion and other imaging features were similar to the lesion in the left temporal lobe, this lesion was also diagnosed with radiation necrosis. The cystic encephalomalacia areas and gliosis in both temporal lobes in the cranial MRI of the patient who did not receive any treatment other than the support treatment (Figure 3) proved the diagnosis of radiation necrosis occurring at different times in both temporal lobes.
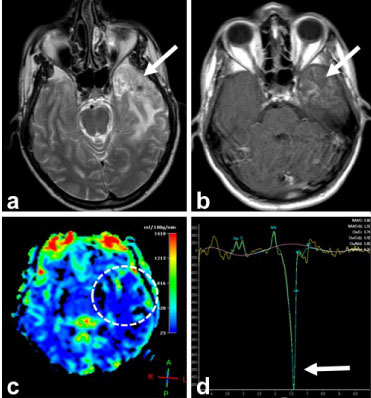 Figure 1: MRI examination performed at the first visit of the patient. Axial T2A image shows (a) heterogeneous signal increase and edema in the left temporal lobe with the 'Swedish cheese' appearance. In the post-contrast axial T1W image (b), heterogeneous contrast enhancement was observed in this region, but perfusion MRI showed low perfusion in rCBF map (c). The very apparent lactate peak observed in MR spectroscopy obtained at the mid TE supports the diagnosis of radiation necrosis. View Figure 1
Figure 1: MRI examination performed at the first visit of the patient. Axial T2A image shows (a) heterogeneous signal increase and edema in the left temporal lobe with the 'Swedish cheese' appearance. In the post-contrast axial T1W image (b), heterogeneous contrast enhancement was observed in this region, but perfusion MRI showed low perfusion in rCBF map (c). The very apparent lactate peak observed in MR spectroscopy obtained at the mid TE supports the diagnosis of radiation necrosis. View Figure 1
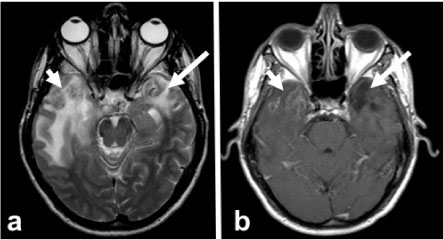 Figure 2: First year control MRI. Axial T2A image (a) in the left temporal lobe edema decreases, gliosis is observed to develop (arrow). In addition, another T2 hyperintense lesion with a similar character is present in the right temporal lobe (short arrow). Post-contrast axial T1W image (b) shows no contrast enhancement in the left temporal lobe (arrow) and heterogeneous enhancement in the right temporal lobe (short arrow). View Figure 2
Figure 2: First year control MRI. Axial T2A image (a) in the left temporal lobe edema decreases, gliosis is observed to develop (arrow). In addition, another T2 hyperintense lesion with a similar character is present in the right temporal lobe (short arrow). Post-contrast axial T1W image (b) shows no contrast enhancement in the left temporal lobe (arrow) and heterogeneous enhancement in the right temporal lobe (short arrow). View Figure 2
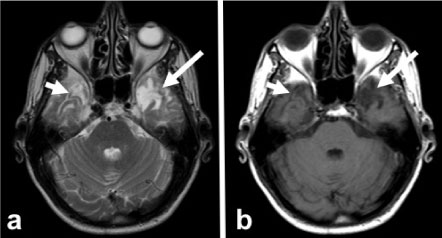 Figure 3: 18 months control MRI. Axial T2A (a) and T1A (b) images show the disappearance of edema and mass in both temporal lobes and encephalomalacia and gliosis (arrows). View Figure 3
Figure 3: 18 months control MRI. Axial T2A (a) and T1A (b) images show the disappearance of edema and mass in both temporal lobes and encephalomalacia and gliosis (arrows). View Figure 3
The necrosis secondary to radiation from the temporal lobe is most commonly seen in nasopharyngeal carcinomas and a less frequency in other extracranial malignancies. In one or both temporal lobe damage, near past memory loss, selective attention, recognition of faces, difficulties in finding spoken words or finding words occur. Symptoms of mass effect such as vasogenic edema, headache, nausea, vomiting, photophobia, irritability and diplopia can also be seen. Necrosis and edema may result in hypercoagulability, resulting in thrombosis and stroke. TLN has a variable delay time from three months to 13 years; however, most are seen within the first 5 years after the completion of radiation therapy [5]. TLN occurred at different times in our patient and it was seen in the 2nd and 3rd years after the completion of RT treatment.
The differential diagnosis of TLN includes intracranial NPC expansion, second primary intracranial neoplasm, cerebral metastasis, meningeal extension and brain abscess [6]. CT, MR and PET-CT are useful tools for the diagnosis of TLN, but none are specific. However, MR imaging features commonly seen in radiation necrosis are a soap-bubble-like inner part and a Swedish cheese-like appearance [7]. To our patient; A history of RT for the neck region, heterogeneous appearance of the lesion in the 'Swedish cheese' character, no increase in choline peak in the lesion, increased perfusion in the perilesional edema area or no diffusion restriction was diagnosed as radiation necrosis. Lesions are not simultaneous and occur with a one-year interval make the diagnosis difficult. The presence of gliosis in these regions in the control examinations also confirmed the diagnosis.
The most important risk factor for radiation necrosis is the sum of the radiotherapy dose given that the section size is also important. In a study by Ruben, et al. Chemotherapy after radiation has been shown to increase the risk of cerebral necrosis approximately fivefold [8]. Dosage recommendations are made regardless of this situation [5].
In the case presented above, TLN was diagnosed. Detailed radiological imaging with a well-taken anamnesis can help in early detection. It should be kept in mind that TLN may occur at lower doses than those suggested and associated with concurrent chemotherapy. TLN should always be kept in mind in the differential diagnosis of temporal lobe lesions after radiation therapy in the neck region, although the incidence has been decreasing due to recent technological advances. Because of the similarities in clinical findings and routine imaging methods, metastatic masses and primary brain malignancies may not always be clearly differentiated from TLN. In this case, advanced imaging methods such as spectroscopy and perfusion MR can be helpful in making a diagnosis with a good history.
As a result; In patients with a history of RT and in patients with a head mass, differentiating radiation necrosis from metastases and other malignancies will prevent unnecessary operation and treatment interventions.