Malignant melanoma, Intussusception, Surgery, Metastases
Intussusception is the leading cause of intestinal obstruction and the second most common cause of an acute abdomen in children behind acute appendicitis but is considered a rare condition in adults with an incidence of only 1-5% [1-4]. First reported by Barbette of Amsterdam in 1674, intussusception can be defined as the telescoping of a proximal segment of the gastrointestinal tract (intussusceptum) into the adjacent distal segment of the gastrointestinal tract (intussuscipiens) [5]. However, it wasn't until 1871 that the first successful operation for intussusception in a child was performed by Sir Johnathan Hutchinson [6]. The aetiology of enteric intussusception in adults can be due to an intraluminal tumour in approx. 70-90% of cases and idiopathic in the other 10% [7]. Primary enteric malignancies are rare accounting for less than 2% of gastrointestinal tumours and so metastases from other primaries make the small bowel the second most common viscera to spread to after the liver [8,9]. Of these non-enteric primaries, malignant melanoma has an incidence of 35-70% to metastasising to the small bowel [10,11].
Here we describe an interesting case of duodenal intussusception secondary to histologically proven malignant melanoma of unknown primary origin.
A 66-year-old male with a background of hypertension; iron deficiency anaemia; and parotidectomy with radical neck dissection for malignant melanoma from his right parotid gland in 2016, presented to the acute surgical team at Derriford Hospital with central abdominal pain, severe nausea and lethargy. Prior to this acute admission, he had a follow-up PET CT for his unknown primary melanoma which showed no recurrence of his previously excised malignancy. The patient also had already been referred for outpatient investigation of intermittent epigastric pain, abdominal distension and iron deficiency anaemia for which he had an initial gastroscopy a few months earlier which showed a slightly irregular Z line, normal stomach and first part of the duodenum but was found to have a deep posterior ulcer in the second part of the duodenum with no sign of bleeding or visible vessel. A colonoscopy performed at the same sitting showed mild rectal angioectasia and severe sigmoid looping. As a result of these findings the patient was started on a proton pump inhibitor. A follow up endoscopy was performed 6 weeks later showing a malignant looking lesion in the third part of the duodenum rather than an ulcer, which felt firm at the time. The duodenal biopsy taken was thought not to be sufficient by the endoscopist as he felt that the mucosa was mainly sampled rather than all underlying layers. This subsequently suggested histological features of malignant melanoma but was reported as inconclusive and so was sent to an external lab for FISH (Fluorescence in-situ hybridisation) analysis. The final report of this was a normal result as almost all of the assessable cells in this section had a normal EWSR1 signal pattern and there was no evidence of a clone with a translocation involving the EWSR1 gene at 22q12.
During this acute admission, the patient continued to suffer with acute pancreatitis of unknown aetiology. A nasogastric tube was inserted to decompress his stomach as he could not tolerate keeping down semi-solid food as well as having a PICC line inserted to initiate TPN feeding. As a result of raised lipase his care was handed over to the HPB team.
His initial CT scan did not show any signs of pancreatitis, however, there was clear evidence of a jejunal tumour causing intussusception (Figure 1). It was not clear from the CT scan whether that was the cause of his symptoms, so he went on to have an ultrasound scan to exclude gallstones as a cause of pancreatitis which was reported to be negative for this. It was thought that the raised lipase is most likely due to a small bowel obstruction so the patient went on to have a further CT scan which showed small bowel obstruction and stranding around the jejunal tumour.
His case was discussed in the Hepatopancreaticobiliary Surgery Multi-Disciplinary Team meeting which concluded a likely Gastro-Intestinal Stromal Tumour (GIST) in his duodenum that needed resection simultaneously with the jejunal tumour that had been identified. We managed his symptoms of pancreatitis and then proceeded to a laparotomy for segmental small bowel resection.
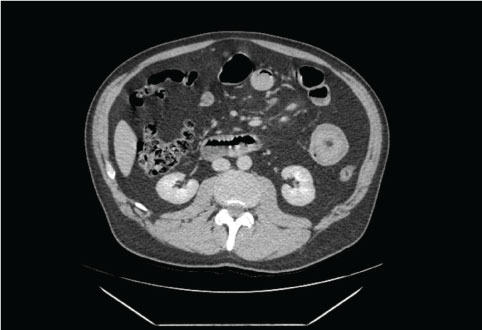 Figure 1: CT abdomen revealing jejunal intussusception - 'Donut' sign.
View Figure 1
Figure 1: CT abdomen revealing jejunal intussusception - 'Donut' sign.
View Figure 1
Intra-operatively, the patient was found to have three tumours: A large tumour in the proximal jejunum approximately 15-20 cm from the DJ flexure causing intussusception and obstruction of the proximal small bowel; and a second obstructing tumour in D2/D3 causing duodenal obstruction that was away from the Ampulla of Vater. After these two tumours were identified and resected, a third lesion in the jejunum further down from the proximal jejunal tumour was also found (Figure 2 and Figure 3). In total he had two jejunal resections and a segmental D2/D3 resection with three end-to-end anastomoses. There was no evidence of metastatic disease. At the end of the laparotomy, one of the specimens was incised and opened showing a polypoid tumour with black pigmentation suggesting that these are most like melanoma metastases (Figure 4).
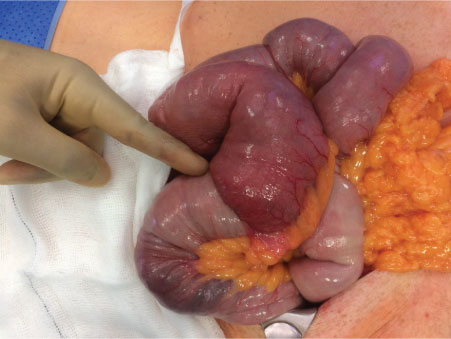 Figure 2: Operative findings of jejunal intussusception with clear lead point.
View Figure 2
Figure 2: Operative findings of jejunal intussusception with clear lead point.
View Figure 2
 Figure 3: Operative findings of separate jejunal tumour with dilated proximal bowel and collapsed distal bowel.
View Figure 3
Figure 3: Operative findings of separate jejunal tumour with dilated proximal bowel and collapsed distal bowel.
View Figure 3
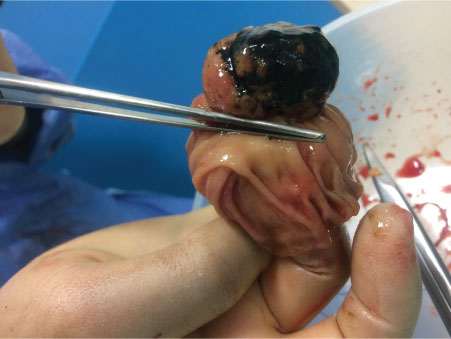 Figure 4: Specimen opened to show appearance of tumour in keeping with malignant melanoma.
View Figure 4
Figure 4: Specimen opened to show appearance of tumour in keeping with malignant melanoma.
View Figure 4
Histopathology all of all the small bowel lesions showed similar morphology where all of the lesions were composed of sheets of spindle cells with focal clearing of the cytoplasm with a more epithelioid appearance. In some areas the cells were arranged in short fascicles. The nuclei were plump, vesicular and showed focally conspicuous nucleoli. Numerous mitoses are seen, the tumour infiltrated the muscle wall and mucosa with surface ulceration. Lymphovascular permeation or perineural infiltration were not seen nor were there areas of necrosis, haemorrhage or evidence of metastatic spread. The most likely differential diagnosis for these findings was metastatic malignant melanoma.
The patient's post-operative recovery was uncomplicated other than a temporary ileus. The patient was discharged home and remained asymptomatic with no signs of recurrence at 3 months follow-up.
Malignant melanoma is the commonest tumour to metastasise to the small bowel and cause an enteric intussusception [12]. It has been reported in the literature in various case studies where the intussusception occurs many years after resection of the primary cutaneous, anal or retinal tumour. However, in this particular case, the original primary malignancy was never identified. The patient had previously had a right parotidectomy for a neck swelling which histologically confirmed malignant melanoma yet this was a deep seated tumour with no superficial extension to the skin. This calls into question whether there is still an undetected primary source in our patient however recent PET CT has not identified any further hotspots.
Although intussusception in adults is considered rare, accounting for only 5% of all intussusception cases, gastro-intestinal melanoma has a much higher incidence of occurrence, accounting for up to one third of abdominal metastases whereby the small bowel is the most common site for metastatic deposition [13,14]. So much so that Schuchter, et al. reported up to 60% of enteric metastases of malignant melanoma in an autopsy series [15]. It has been hypothesised by Mishima that melanoma of the small bowel might as a primary tumour arise from Schwannian neuroblast cells associated with the autonomic innervation of the gastrointestinal tract [16]. Whereas Gill, et al. have postulated that melanoma metastasises to the small bowel due to its rich vascular supply [8]. It has been reported in the literature by both Doyle and Gromet that cutaneous malignant melanoma may suddenly regress thus offering a possible explanation to the deficiency of extra-intestinal lesions in these cases [17,18].
In intussusception due to an intra-luminal lesion, the invagination of a bowel segment into its own lumen can be thought to be due to the lesion of the bowel wall affecting its peristalsis, and thus can act as a lead point for this pathological mechanism to occur [3]. However, in the cases without a lesion, the mechanism is not known. Although not typically obvious in our patient case, intussusception usually presents with features of acute, subacute or chronic bowel obstruction. The typical complex of abdominal pain, blood in the stools (often described as red-currant jelly), and a palpable abdominal mass with or without vomiting is rarely seen in adults and is often difficult to diagnose clinically. In approximately 82-90% of cases, it can be visualised on CT and ultrasound imaging as a target sign or doughnut perhaps with fat stranding and surrounding fluid build-up around the affected bowel segment [1,19]. This can also be eluded by the 'pseudokidney' sign on ultrasound where on an oblique or longitudinal view, the segment of the intussuscepted bowel can appear as a kidney, where the mesentery containing the vessels is pulled into the intussusception and so is reminiscent of the renal hilum, with the renal parenchyma formed by the oedematous bowel. However, when patients present with symptoms and signs related to an abdominal mass or gut motility such as intestinal obstruction, they are more likely to have a CT over an ultrasound scan as first line imaging thus reducing the risk of operator variability and poor views in patients with a large body habitus or distended gas filled bowel loops. Other investigative modalities can also be considered such as gastrograffin contrast studies (barium is contraindicated in intestinal obstruction as has risk of perforation) and endoscopy however depending on the institution and resources available may not be easily accessible and so may be considered in elective cases [20].
The management of intussusception can vary according to the aetiology and also patient population involved, as paediatric cases are often managed with air or intraluminal contrast insufflation in order to reduce the intussusception. Adult cases are rarer and usually associated with other pathologies thus may warrant surgical intervention, for example those due to malignancy would likely require an oncological resection. Non-surgical reduction is associated with the risk of worsening ischaemia, perforation and the seeding of malignant cells if the cause is due to a cancerous lesion [21]. Other considerations to bear in mind are whether to attempt reduction prior to surgical intervention in order to maximise the preservation of intestinal length. In an emergency presentation, our institution would opt to proceed to surgical intervention without delay rather than offer reduction pre-operatively again due to the aforementioned risks above. In cases where the aetiology is idiopathic or post-traumatic, it has been suggested that simple reduction is acceptable assuming that bowel ischaemia and perforation has been excluded [22,23]. The method of surgical intervention i,e. laparoscopy versus a laparotomy is dependent upon multiple factors: Patient's clinical condition; prognostic scores such as P-Possum; comorbidities and previous abdominal surgery; the extent and level of intussusception; resources available; and the skill set of the surgeon. Finally, once the intussusception has been surgically managed with the melanoma excised, the diagnosis of primary malignancy versus metastatic disease should be considered which will also determine the most appropriate follow-up pathway. A primary intestinal melanoma can be diagnosed based on three criteria proposed by Sachs, et al. (1) Histologically proven melanoma of the small intestine at a single focus; (2) No evidence of the disease in any other organs including skin; and (3) A disease free survival of at least 12 months after diagnosis [24]. However, the limitations of these criteria are that they do not take into account the detection of metastasised lesions which have been demonstrated in many cases within the literature [25,26].
Small bowel intussusception is a rare condition in adults however has been reported in the literature to be commonly associated with an enteric tumour, with metastases being the most common form encountered. This is usually poses clinical challenges in diagnosis and so a high index of suspicion, prompt symptom control resuscitation are necessary to prevent complications associated with intussusception such as perforation and bowel ischaemia. Malignant melanoma is known to metastasise to the small intestines, however is usually associated with other deposits such as cutaneous and retinal. Metastatic melanoma causing high grade intussusception in the duodenum and jejunum with no identification of a primary is rare but should involve an MDT approach where possible to offer the best prognosis for the patient.
No sources of funding.
None.
I declare the information stated in this article is accurate to the best of my knowledge.
Miss. Niroshini Rajaretnam - Primary author of case report.
Mr. Somaiah Aroori - Consultant surgeon who admitted and operated on the patient during this hospital admission. Involved in multidisciplinary team discussions and help edit manuscript.