Background: Necrotizing fasciitis is a life-threatening rapidly progressing bacterial infection associated with necrotic changes of the superficial fascia, subcutaneous tissue, and the deep fascia. It is a surgical emergency requiring a high suspicion, early diagnosis, and early treatment. We aim to analyze the clinical and microbiological pattern of necrotizing fasciitis and to determine factors associated with mortality.
Methods: A prospective cohort study of the results of 476 patients who were diagnosed and treated for necrotizing fasciitis between January 2005 and December 2014in Al Ansar general public health hospital in Medina, Saudi Arabia was done.
Results: Age ≥ 40 years was found statistically to be associated with developing necrotizing fasciitis for both males and females. The majority of the patients had involvement of the lower limb; 31.7% in the right calf and 29.8% in the left calf. Soft tissue infection in 24.4% of the patients was the most common etiology. The most common organism isolated in the monomicrobial infection was Beta-hemolytic Streptococcus in 46.7% of the patients, and the most common organism isolated in the polymicrobial infection was Staphylococcus aureus in 72.8% of the patients. Surgical debridement was done to all patients (100%). The mortality rate was (7.1%). Acidosis (p < 0.05) and hypoalbuminemia (p < 0.05) were the significant factors for mortality.
Conclusion: The aggressive course of necrotizing fasciitis could lead to considerable morbidity and mortality. Acidosis and hypoalbuminemia were found to be factors strongly associated with high mortality. Other possible factors include truncal location (from the neck to the pelvic region) and leukocytosis.
Necrotizing fasciitis, Soft tissue, Infection, Surgical debridement, Antimicrobial therapy
Necrotizing fasciitis is a life-threatening rapidly progressing bacterial infection associated with necrotic changes of the superficial fascia, subcutaneous tissue, and the deep fascia. It is the most aggressive form of necrotizing soft tissue infection associated with a high mortality rate and can spread rapidly to the entire limb within hours [1,2]. The reported mortality rates of the disease have remained alarmingly high ranging from 6% to 76%. Multiple clinical studies have shown that the delay in the diagnosis and consequently delayed operative debridement had caused an increase in the mortality [2-4].
Initially, it does not show classical clinical signs and is hard to differentiate from cellulitis (edema, local anesthesia, and crepitations). Therefore, a high index of suspicion is needed to diagnose it. It first starts in the deep tissue plane, so superficial skin signs (redness, edema, and pain) may not be quite obvious initially. As a result, itusually leads to a delay in the diagnosis. Many patients may present with toxic features due to sepsis without any underlying signs. Later, they may develop edema, tenderness, vesicle, bullae, crepitus, patches of dusky blue discoloration lesions, and eventually gangrene of the affected tissues by the fourth or fifth day [4,5]. The systemic manifestations include altered mental status, tachycardia, tachypnea, oliguria, fever, chills, leukocytosis, hyperglycemia, and metabolic acidosis [5,6].
Based on the microbiological pathogens, necrotizing fasciitis is classified into type 1 (synergistic polymicrobial infections including anaerobes) and type 2 (monomicrobial infections), the former being more common [6,7]. The most common monomicrobial infection-causing organisms include Beta-hemolytic Streptococcus, Staphylococcus aureus, and Clostridial species. While the most common polymicrobial synergistic infection-causing organisms include Staphylococcusaureus, Streptococcus pyogenes, Enterococci species, Escherichia coli, Pseudomonas species, and anaerobic organisms such as Bacteroides [7,8].
The precise pathogenesis of necrotizing fasciitis is not entirely developed. Inoculation of microbes can occur through minor trauma, snake or insect bite, or surgical incisions. Under favorable environmental conditions like immunocompromised states, renal failure, liver failure, and diabetesmellitus, the organisms multiply to cause the disease process. Necrotizing fasciitis may occur in any part of the body, but more commonly reported in the extremities, abdomen, groin, and perineum [9,10].
The clinical features mostly diagnose necrotizing fasciitis, but other investigations could help to confirm it. A plain X-ray could show subcutaneous gas. Computed tomography scan and magnetic resonance images could show asymmetrical fascial thickening, fat stranding, and gas tracking along fascial planes. Tissue biopsy is the diagnostic test that reveals necrosis, nuclear polymorph infiltration, and thrombosis of vessels [11,12].
The keystone protocol of management necessitates early diagnosis, prompt debridement of involved tissues, and empiric antimicrobial therapy. It includes initial resuscitation measures (intravenous fluids), supportive care (analgesia), adequate control of risk factors such as blood sugar, intravenous antibiotics, extensive surgical debridement, and occasionally radical procedures such as amputations [13,14].
Unfortunately, despite its frequent presentation to the emergency departments, necrotizing fasciitis is still under-reported in Saudi Arabia. A thorough literature search showed a paucity of local articles studying the sociodemographic data, incidence rates, and the clinical, microbiological pattern of necrotizing fasciitis in the Saudi population. In addition, few international articles investigated the factors influencing the outcome or associated with mortality. Therefore, Inthis study, we aim to analyze the clinical pattern and microbiological characteristics of necrotizing fasciitis and to determine the factors associated with mortality.
A prospective cohort database analysis of the results of patients who were diagnosed and treated for necrotizing fasciitis between January 2005 and December 2014 in Al Ansar general public health hospital in Medina, Saudi Arabia was done. The treatment protocol, part of our quality care clinical pathway program, was designed on a digital database file. Ethical approval was granted from the Al-Ansar hospital ethical committee and the management guidelines and clinical pathway subcommittee of the quality care program at the same hospital.
All the patients who presented in the acute form were admitted from the emergency department, and those referred by other specialties and local hospitals (inpatients and outpatients) were admitted or shifted to the general surgical ward or the intensive care unit. Inclusion criteria included adult patients (above 12 years old as per the Saudi Arabian Ministry of age health guidelines) and a suspected or proved diagnosis of necrotizing fasciitis. No exclusion criteria.
All patients had the same workup (complete blood count, coagulation profile, blood chemistry, blood culture for bacteria, chest X-ray, electrocardiogram, an X-ray of the involved body part, ultrasound, and computed tomography scan). The medical management and surgical procedures were carefully discussed with the patients. All the patients were started on intravenous fluids, intravenous analgesia (Paracetamol 1g every 12 hours) and empiric intravenous antibiotics (after blood culture) (Penicillin 1-4 million U intravenous every 4 hours, Clindamycin 600 mg/kg intravenous every 8 hours, and ciprofloxacin 400 mg intravenous every 12 hours). All antibiotics were then changed according to the yield of the blood culture towards the causative organisms.
The surgical procedure was done as an emergency surgery within 2-6 hours of the diagnosis in all patients. The resected affected tissues were transferred to sterile plastic sample containers for culturing. The postoperative care was carried out in the intensive care unit for 384 (80.7%) cases while 92 (19.3%) mild cases were managed on the surgical floor.
As part of the clinical pathway of our quality care program, a digital database file was used to document all the patient's data. Statistical package for social science (SPSS) program (Release 22) was used for data analysis. Results were represented by absolute percentages, average, and mean.
The clinical and microbiological profiles of the patient's were analyzed for age, sex, clinical features, site of infection, risk factors, etiological factors, microbiological characteristics, and the treatment outcome. The standard deviation was calculated. The chi-square test was applied to all possible categorical variables ata 95% confidence interval and the p-value ≤ 0.05 was considered statistically significant.
476 patients who were diagnosed and treated for necrotizing fasciitis were included in the study. 318 (66.8%) were males and 158 (33.2%) were females. The male to female ratio was (2: 1). The mean age of the male patients was 42.6 ± 7 years, while it was 39.2 ± 9 years in female patients. 341 (71.6%) patients belonged to the age group 40-60 years. Age ≥ 40 years was found statistically to represent a significant difference in the association of necrotizing fasciitis for both males and females (odds ratio = 3.4, P < 0.05). The demographic distribution of the study population showed that 283 (59.5%) patients were from Medina city, 94 (19.7%) were from nearby towns and villages, 86 (18%) were pilgrims and Umrah visitors to Medina, and 13 (2.7%) were from rural areas.
The majority of the patients had involvement of the lower limb; 151 (31.7%) in the right calf region, 142 (29.8%) in the left calf area, 63 (13.2%) in the abdomen, 41 (8.6%) in the right arm, 39 (8.2%) in the left arm, 19 (4%) in the chest, 12 (2.5%) in the buttock and perineum, and 9 (1.9%) in the head and neck (Table 1).
Table 1: The location of necrotizing fasciitis in relation to gender. View Table 1
Regarding the risk factors, non-insulin dependent diabetes mellitus type 2 was recorded in 114 (23.9%) patients, insulin dependent diabetes mellitus type 1 in 69 (14.5%), hypertension in 37 (7.8%), ischemic heart disease in 17 (3.6%), renal impairment in 13 (2.7%), hematologic disease in 11 (2.3 %), bronchial asthma in 8 (1.7%), malignancy in 6 (1.3 %), and no risk factor could be identified in 201 (42.2%) patients.
The etiological factors included soft tissue infection in 116 (24.4%), skin lacerations in 102 (21.4%), blunt trauma in 84 (17.7%), penetrating trauma in 53 (11.1%), infected diabetic foot in 23 (4.8%), infected burn wound in 19 (4.1%), previous surgery in14 (2.9%), insects bite in 12 (2.5%), snake bite in 9 (1.9%), and unknown etiology in 44 (9.2%) (Table 2).
Table 2: The etiological factors of necrotizing fasciitis in relation to gender. View Table 2
Physical findings were recorded as follows: Tenderness was the most common in 428 patients (89.9%), edema in 351 (73.7%), erythema 318 (66.8%), skin vesicles/bullae 298 (62.6%), soft tissue crepitus 286 (60.1%), fever 264 (55.5%), hypotension 126 (26.5%), tachycardia 211 (44.3%), altered mental status 174 (36.6%), skin necrosis in 139 (29.2%), and skin ulceration in 121 (25.4%) (Figure 1).
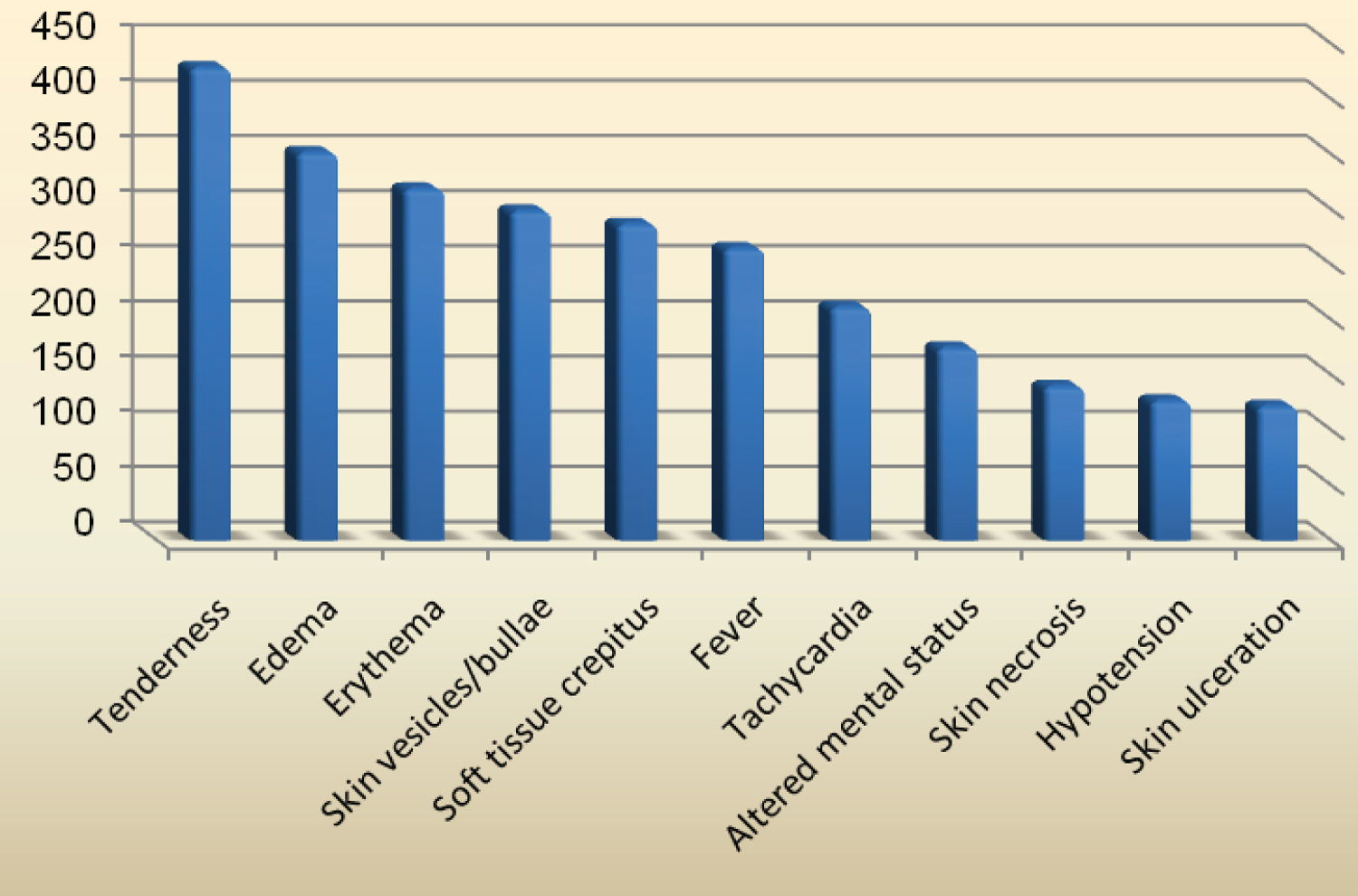 Figure 1: The physical findings of necrotizing fasciitis patients.
View Figure 1
Figure 1: The physical findings of necrotizing fasciitis patients.
View Figure 1
Blood culture was positive in all the patients (100%). It showed the growth of 25% monomicrobial (120 patients) and 75% polymicrobial (356 patients). The most common organisms isolated in the monomicrobial infection were Beta-hemolytic Streptococcus in 56 (46.7%) patients, Escherichia Coli in 41 (34.2%), and Clostridium difficile in 23 (19.2%). The most common organisms isolated in the polymicrobial infection were Staphylococcus aureus in 259 (72.8%) patients, Pseudomonas aeruginosa in 216 (60.7%), Staphylococcus Epidermis in 194 (54.5%), Enterococcus species in 172 (48.3%), Klebsiella pneumoniae in 136 (38.2%), Bacteroides in 113 (31.7%), Proteus Mirabilis in 107 (30.1%), Acinetobacter in 82 (23%), Citrobacter speciesin 64 (18%), and Alcaligenes Faecalis in 47 (13.2%) (Figure 2).
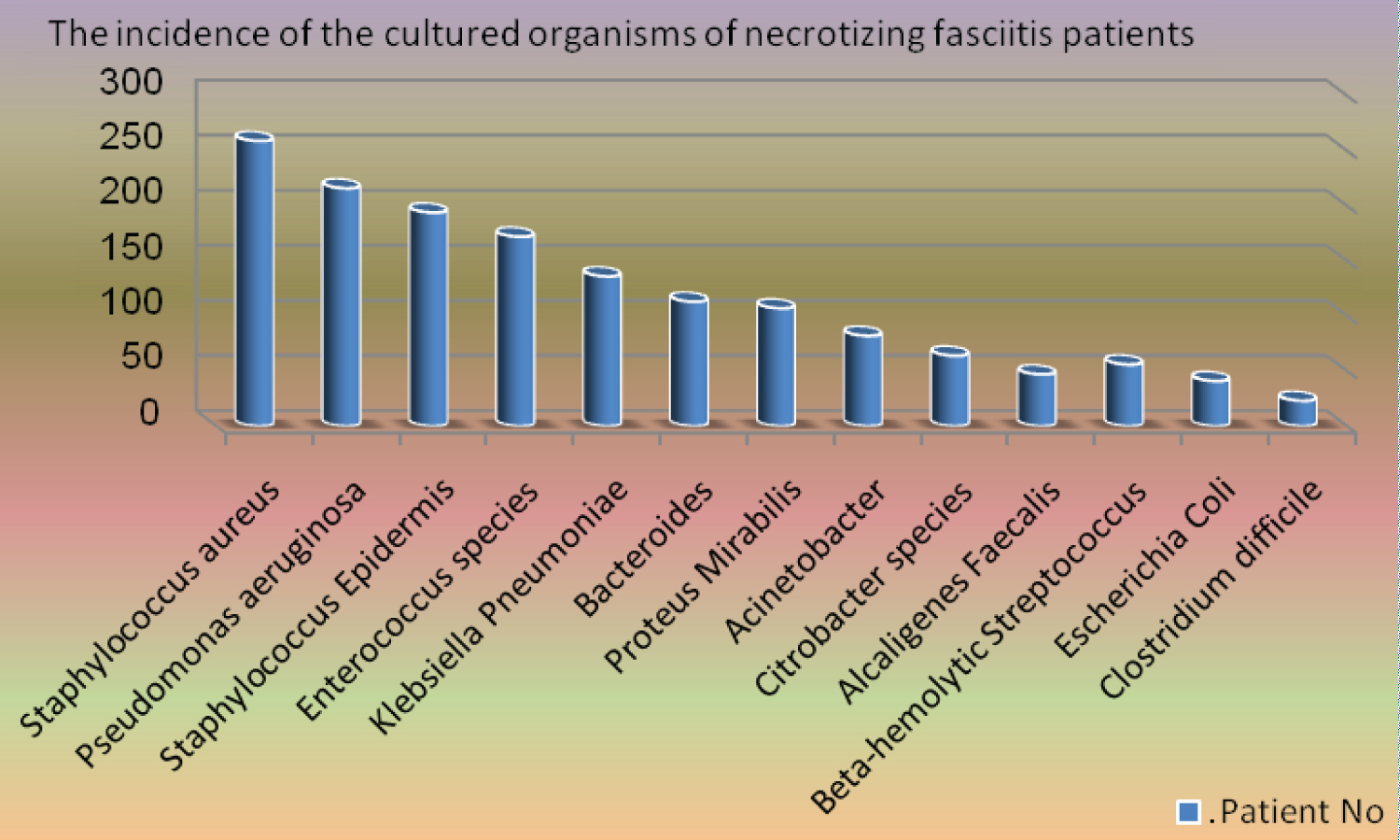 Figure 2: The incidence of the cultured organisms of necrotizing fasciitis patients.
View Figure 2
Figure 2: The incidence of the cultured organisms of necrotizing fasciitis patients.
View Figure 2
Tissue culture was positive in 348 (73.1%). The most common organisms isolated were Beta-hemolytic Streptococcus in 49 (14.1%) patients, Escherichia Coli in 37 (10.6%), Clostridium difficile in 19 (5.5%), Staphylococcus aureus in 107 (30.7%) patients, Pseudomonas aeruginosa in 53 (15.2%), and Staphylococcus Epidermis in 83 (23.9%).
Antimicrobial susceptibility for some of the isolated organisms is summarized in Table 3.
Table 3: Antimicrobial susceptibility of the isolated organisms of necrotizing fasciitis patients. View Table 3
Surgical debridement was done to all patients (100%). The mean time from diagnosis to surgical intervention was 4 hours, range 2-6. The mean operative time was 75 ± 10 minutes, range 40-110. Abscess formation was identified in 327 (68.7%) patients, soft tissue necrosis in 294 (61.8%), muscle necrosis in 138 (29%), multiple involvement in 129 (27.1%), and gangrene in 94 (19.7%). The mean number of surgical debridement performed on each patient was 3, range 1-5. A total of 386 (81.1%) patients required more than one debridement session. Amputation was done in 19 patients (4%), 12 (2.5%) below-knee amputation, and 7 (1.5%) above-knee amputation, all were diabetic patients.
Ten factors were assessed for correlation with mortality: age ≥ 40 years, hypotension, diabetes mellitus, truncal location (from the neck to the pelvic region), anemia (< 10 mg/dL), leukocytosis (> 12 × 109/L), thrombocytopenia (< 100 × 109/L), acidosis (pH < 7.35), hypoalbuminemia (< 30 g/L), and azotemia (blood urea nitrogen > 5.4 mmol/L) (Table 4).
Table 4: Risk factors for mortality in patients with necrotizing fasciitis. View Table 4
The results showed that truncal location (p = 0.036), leukocytosis (p = 0.039), acidosis (p = 0.002), and hypoalbuminemia (p = 0.004) were statistically found to be strongly associated with mortality. Independent predictors of mortality were analyzed, and the logistic regression analysis showed that acidosis (p < 0.05; 95% confidence interval: 1.8-44.8) and hypoalbuminemia (p < 0.05; 95% confidence interval: 1.6-64.5) were the significant factors for mortality. The mortality rate was (7.1%), 34 patients died while 442 (92.9%) completely recovered from the acute attack.
The mortality rate in our study was 7.1% which is lower when compared to other studies in the literature [3,4,9,10,14,15,24]. Because of the significant mortality associated with necrotizing fasciitis, a high index of suspicion is crucial. However, given the preponderance of initial nonspecific clinical manifestations, early diagnosis is often difficult. The disease outcome is adversely influenced by host immunity and bacterial isolates. Due to the spectrum of bacterial isolates involved, empiric antibiotics therapy must have an adequate wide spectrum against the typical bacteria involved. Particular clinical markers could be used to predict the possible adverse outcomes. Despite that, aggressive surgical debridement still the cornerstone treatment of this devastating soft tissue infection [23-25].
In our patient population, 341 (71.6%) patients belonged to the age group 40-60 years, which is consistent with the reported worldwide [9,10,19]. During 20 years of clinical practice, we have never encountered necrotizing fasciitis in the young age group (below 20 years of age), which could indicate that the disease is related to the middle and advanced age. A fact that is supported by our result that age ≥ 40 years was found statistically to represent a significant difference in the association of necrotizing fasciitis for both males and females (odds ratio = 3.4, P < 0.05). We could explain that by the fact that it commonly affects the working-age group who are frequently encountering trauma to the used body parts, especially the lower limbs.
Various risk factors predispose to this condition such as diabetes mellitus, hypertension, immunocompromised status, and chronic renal failure among which diabetes was the most common risk and predisposing medical condition that has been reported by others, with ranges from 32% to 62% [16,20]. Our result showed that 38.4% of our patients were diabetics, a finding that was consistent with many other studies. The increased blood sugar predisposes to the environment of low oxygen tension and acts as a good substitute for bacterial growth. The most common aerobes isolated from necrotizing fasciitis patients with diabetes were Escherichia Coli (21%) and enterococci (18%) [16].
The most-reported etiological factors included soft tissue infections, trauma, snake bite, postoperative status, with soft tissue infection being the prime cause in some studies, and trauma in other studies, though, many articles reported no prior skin lesion [17,18]. In our study, soft tissue infection was the cause in 116 (24.4%), which could be explained by the fact that most of the patients presented and were admitted as cellulitis but found later to have involved deeper tissues and diagnosed as necrotizing fasciitis.
The patients could present with a broad range of clinical features such as tenderness, edema, erythema, skin vesicle/bullae, soft tissue crepitus, hypotension, fever, tachycardia, and altered mental status [19-22]. In our study, tenderness (common in 428 patients (89.9%)), edema (in 351 (73.7%)), and erythema (in 318 (66.8%)) were the consistent physical which suggests that these may be the early signs of necrotizing fasciitis. Also, skin vesicles/bullae were present in 298 (62.6%) and soft tissue crepitus in 286 (60.1%). The infection can be present without a clear portal of entry such as a wound or ulceration. Therefore, clinical suspicion of its presence in a patient with severe pain and local signs of inflammation should be higher. Necrotizing fasciitis must be suspected if a patient present with a level of pain and tenderness that is out of proportion to the physical signs, present with dusky skin lesions with erythema, presence of crepitus, develops blisters in a short time, the affected body part develops necrotic changes, and the patient becomes very toxic.
In our study, 100% of the blood cultures yielded growth, out of which polymicrobial was the leading cause. The commonest organisms isolated were Staphylococcus aureus 259 (72.8%) and Pseudomonas aeruginosa 216 (60.7%). While the most common organisms isolated in the monomicrobial infection were Beta-hemolytic Streptococcus in 56 (46.7%) patients and Escherichia Coli in 41 (34.2%). Similar isolations were reported in many articles internationally [4,7,14,23,24]. Skin penetration was the most frequent cause of injury among trauma patients with necrotizing fasciitis in our study. Other patients had injuries that were sustained from blunt trauma. Among trauma patients who eventually developed necrotizing fasciitis, the most frequently isolated organisms were Escherichia Coli, Staphylococcus aureus, and Klebsiella pneumoniae.
Bacteroides were isolated in 113 (31.7%) patients in our series. Few articles reported the association of anaerobes with necrotizing fasciitis [19,25]. Variation in the isolates between studies is most probably explained by the use of different antibiotics and culture techniques. Since some bacteria can lead to necrotizing fasciitis, the disease must be considered as a clinical entity and not a specific bacterial infection [23-25].
Given the antimicrobial susceptibility patterns in our study, the suggested empiric antibiotic regimen should include broad-spectrum antibiotics with Gram-negative, Gram-positive, and anaerobic coverage. The following drugs have been shown to be highly effective against the organisms isolated in our study: Piperacillin-tazobactam, ampicillin-sulbactam, cefuroxime, ceftazidime, metronidazole, imipenem, and vancomycin. The empiric antibiotic regimen must be chosen by taking into consideration the suspicion of the underlying clinical setting of the necrotizing infection, with the most probable antimicrobial susceptibility of the organisms involved.
Most of the surgical debridement procedures done to our patients prioritized the evacuation of the abscess and the excision of necrotic tissues as much as possible to leave a healthy ground for healing. A total of 386 (81.1%) patients required more than one debridement session, which clearly indicates the aggressiveness of necrotizing fasciitis and how bad can it progress. In fact, in many patients, muscle necrosis was encountered and had to be debrided.
In our series, amputation was done in 19 patients (4%), 12 (2.5%) below-knee amputation, and 7 (1.5%) above-knee amputation, all were diabetic patients. This again indicates the severity of necrotizing fasciitis, especially in diabetic patients.
Worldwide, little is known about post-hospital outcomes for survivors of necrotizing fasciitis. Only a single published study analyzed long-term mortality rates for survivors of necrotizing fasciitis and found that they had markedly higher rates of death. For future specialized research studies, important aspects to be studied should include relationship stresses, physical functioning, depression, post-traumatic stress disorder, and employment concerns [17].
Our study identified acidosis and hypoalbuminemia as factors strongly associated with high mortality in necrotizing fasciitis patients. Other possible factors include truncal location (from the neck to the pelvic region) and leukocytosis. Other international studies showed that other factors are associated too. Liu, et al. found that more than one comorbidity, thrombocytopenia, anemia, more than 24 hours delay from onset of symptoms to surgery, and age greater than 60 were independently associated with mortality [27]. While Elliott, et al. found that Age > 60 years, female gender, high blood creatinine and lactate levels, the number of days from admission to the first debridement, (%) of body surface area and the number of organs failed on admission to significantly increase the risk of death [28].
We recommend conducting further research to evaluate the long-term outcomes for survivors of necrotizing fasciitis in our local community and internationally. We also recommend the development of a national public awareness program in our local Saudi Arabian community emphasizing the importance of good health care and attention to even the minor skin and soft tissue injuries, particularly in diabetics and immunocompromised patients. This aims to help avoid the progression of minor traumatic lesions to a severe catastrophic infection with all its medical, personal, and financial impacts.
The aggressive course of necrotizing fasciitis could lead to considerable morbidity and mortality. Early recognition, prompt treatment, and aggressive surgical debridement are crucial. Antimicrobial treatment is best administered according to blood culture yield. Though, empiric treatment should start immediately to help minimize the catastrophic progression of the disease. Acidosis and hypoalbuminemia were found to be factors strongly associated with high mortality. Other possible factors include truncal location (from the neck to the pelvic region) and leukocytosis.
The authors declare that they have no conflicts of interest.