The majority of coronary artery fistulas are congenital and discovered incidentally on coronary angiography. Coronary artery fistulas may also develop after cardiac transplantation as a complication of endomyocardial biopsies. The natural history and optimal management of post-transplant coronary artery fistulas are uncertain. We report the case of a patient who developed symptoms from an early post-transplant coronary fistula, which was treated successfully with a covered stent. We review the literature, discussing current knowledge on incidence, natural history and management.
Coronary Artery Fistulas (CAF) have been reported after orthotopic heart transplantation as a possible complication of Right Ventricular (RV) endomyocardial biopsies. Their true incidence, natural history and management have not been well described in the literature. We present a unique case of early detection of CAF at 1-month post-cardiac transplant and serial evaluation by intracoronary hemodynamics to guide management. Post-transplant CAFs are likely a more common complication of endomyocardial biopsies than recognized and routine screening should be considered. We review available data on CAFs in order to better understand the natural history and treatment outcomes.
A 53-year-old male with an ischemic cardiomyopathy underwent orthotopic heart transplantation with biatrial anastomosis. He received basilixumab induction therapy and subsequent maintenance immunotherapy of tacrolimus, mycophenolate mofetil and prednisone along with aspirin and a statin. His immediate post-operative course was uncomplicated and he was discharged 12 days after transplant. As part of routine post-transplant rejection surveillance, RV endomyocardial biopsies were performed weekly with a total of 21 samples obtained in the first month. As per current major society guidelines, an average of 4 to 5 endomyocardial samples was obtained at each biopsy. There were no reported complications, however, on the 4th procedure, 4 of 5 biopsy samples contained fibroadipose tissue that was suspicious for epicardial fat sampling. An echocardiogram was performed as the patient complained of shortness of breath. This showed abnormal diastolic flow into the RV apex/apical septum, suggestive of a CAF (Figure 1). Both ventricles were normal in size and systolic function. Coronary angiography confirmed the presence of a mid Left Anterior Descending Artery (LAD) to RV fistula (Figure 2). Concurrent intracoronary hemodynamic evaluation showed reduced LAD coronary flow reserve of 0.5 distal to the fistula compared to a coronary flow reserve of 1.2 proximal to the fistula, suggesting significant coronary steal (Table 1). A right heart hemodynamic study was also performed, which did not demonstrate a step up in oxygen saturation or left to right shunting (Qp:Qs 0.99). As the patient's symptoms resolved following diuresis, conservative management was pursued with close hemodynamic and imaging monitoring.
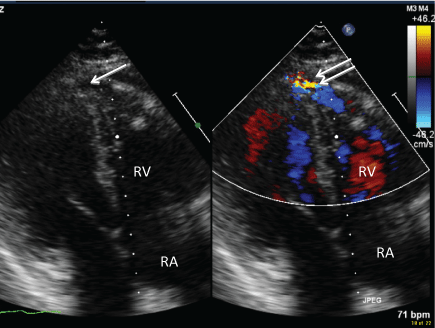 Figure 1: Reverse apical 4-chamber view 2D image (left) and color doppler (right) demonstrating fistula (single arrow) and diastolic flow (double arrow) into the Right Ventricle (RV).View Figure 1
Figure 1: Reverse apical 4-chamber view 2D image (left) and color doppler (right) demonstrating fistula (single arrow) and diastolic flow (double arrow) into the Right Ventricle (RV).View Figure 1
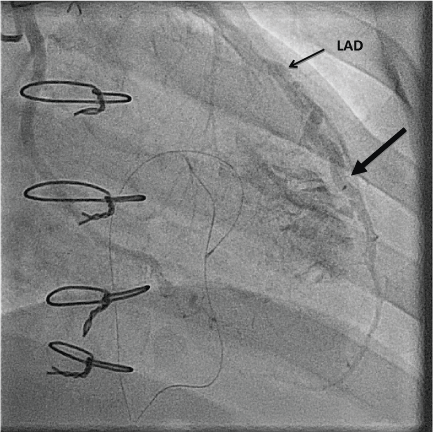 Figure 2: Angiogram demonstrating the Left Anterior Descending Artery (LAD) to right ventricle fistula (large arrow). View Figure 2
Figure 2: Angiogram demonstrating the Left Anterior Descending Artery (LAD) to right ventricle fistula (large arrow). View Figure 2
Table 1: Summary of invasive coronary hemodynamic evaluation of the LAD. View Table 1
At 6 months post-transplant, follow-up surveillance coronary angiography showed negative remodeling and occlusion of the LAD distal to the fistula. Filling of the distal LAD occurred after administration of intracoronary glyceryl trinitrate and intravenous adenosine. Coronary flow reserve was again reduced distal to the fistula, measuring 0.6. Dipyridamole rubidium-82 cardiac positron emission tomography showed mildly reduced myocardial flow reserve at the left ventricular apex. Due to these findings, elective percutaneous coronary intervention was scheduled and undertaken 8 months after transplant. A covered bare-metal stent was placed to seal off the fistula (Figure 3). Subsequent surveillance echocardiograms and myocardial perfusion/flow imaging did not demonstrate recurrence of abnormal flow. However, on follow-up coronary angiography at 13 months post-transplant (5 months after stenting), there was 90% in-stent restenosis and evidence of early cardiac allograft vasculopathy with non-obstructive disease evident in the all three major coronary arteries. The LAD in-stent restenosis was treated by drug eluting balloon angioplasty. Sirolimus was added to his immunosuppression regimen to delay progression of cardiac allograft vasculopathy. The patient remains well, currently at 30 months post-transplant.
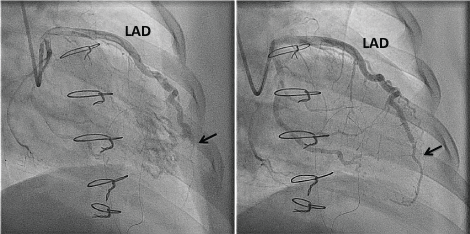 Figure 3: A) Occlusion of distal Left Anterior Descending Artery (LAD) at fistula (arrow); B) Improved flow in distal LAD following insertion of covered stent (arrow). View Figure 3
Figure 3: A) Occlusion of distal Left Anterior Descending Artery (LAD) at fistula (arrow); B) Improved flow in distal LAD following insertion of covered stent (arrow). View Figure 3
Coronary Artery Fistulas (CAF) occur in the general population but are rare with a reported 0.2% incidence [1]. Large, hemodynamically significant fistulas are detected at an early age and usually treated by surgical intervention. Congenital CAF occur due to embryologic arrest leading to additional communications within the heart or major vessels. Acquired CAF are commonly due to trauma or iatrogenic, related to endomyocardial biopsies or less commonly, inadvertent bypass of the vein instead of the artery during coronary bypass. Most CAFs are found incidentally during coronary angiography. Patients are usually asymptomatic but occasionally exhibit chest pain, shortness of breath and palpitations.
Case series report up to a 21% incidence of acquired CAF in heart transplant patients (Appendix Table 1). The majority of reported CAFs were detected on angiography at 1-year post-transplant, and are believed to be iatrogenic. Interestingly, angiogenesis secondary to the post-cardiac transplant inflammatory state has also been proposed to contribute to the formation of fistulas. This is supported by the detection of CAFs that do not involve the RV, which is unexpected for fistulas complicating RV endomyocardial biopsies [2-5]. The true incidence of CAF post-transplant is likely underestimated as most patients are asymptomatic and surveillance coronary angiography is not routine for all transplant programs. Similarly, our centre utilizes noninvasive echocardiography and nuclear myocardial perfusion imaging for allograft surveillance, which do not enable accurate evaluation for CAF post-transplant.
There is currently no consensus on the ideal management of post-cardiac transplant CAF as their natural history is unclear. Theoretical concerns include: [1] coronary steal leading to ischemia and arrhythmia, [2] a source of embolization, and [3] the risk of infectious endocarditis. A conservative management approach is common, but follow-up data is short ranging from 2 to 4 years after initial diagnosis [6-8]. Variable outcomes have been reported with some fistulas increasing in size, some remaining unchanged in appearance and others resolving spontaneously. Furthermore, despite the abovementioned theoretical complications of CAFs, few have been confirmed in the literature.
Data on treatment intervention for CAF is relatively sparse. The majority of available long-term data is for congenital CAF. Congenital CAFs that are found incidentally in adulthood have been treated with ligation, coils, occluder devices, vascular plugs and even bypass. Invasive surgical intervention is difficult to justify in asymptomatic patients. In a case series of 58 patients with treated congenital CAFs, all patients' survived surgery but 8 patients had ischemia in the peri-operative period and 1 had a significant arrhythmia leading to a stroke [9].
Appendix Table 2 summarizes published outcomes of treatment interventions for post-cardiac transplant CAF. The decision to treat was based on the development of symptoms for the majority of patients. Oreglia, et al. reported a case of a LAD to RV fistula noted on 1-year surveillance angiogram [10]. The patient was initially asymptomatic with a normal echocardiogram. At 10 years post-transplant the patient developed apical hypokinesis on echocardiogram, a continuous murmur and a dilated LAD. A Jo stent was inserted in the distal LAD to cover the fistula. Allograft function remained preserved before and after the procedure and 1-year follow-up angiography showed a patent stent. A similar successful treatment strategy with a Jo stent to cover a LAD CAF was reported in a patient presenting with an acute myocardial infarction immediately post-endomyocardial biopsy (Figure 4) [11]. Other percutaneous options have also been explored including detachable balloons and coils [12,13]. Uchida, et al. reported a single case of coronary artery surgical bypass for a CAF post-transplant [14]. A saphenous vein graft was performed on the LAD with suturing of the fistula. Long-term data was not reported for any of the above studies.
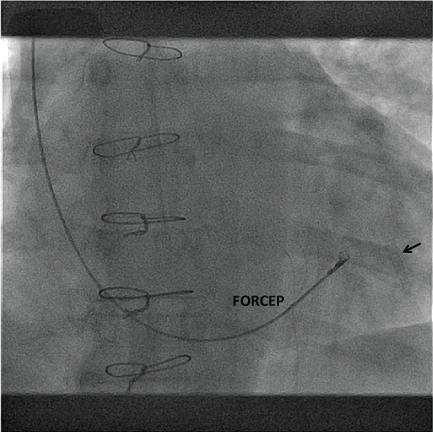 Figure 4: Fluoroscopic image obtained during subsequent endomyocardial biopsy demonstrating the close proximity of the biopsy forceps to the inserted covered stent in left anterior descending artery (arrow). View Figure 4
Figure 4: Fluoroscopic image obtained during subsequent endomyocardial biopsy demonstrating the close proximity of the biopsy forceps to the inserted covered stent in left anterior descending artery (arrow). View Figure 4
Post-transplant CAFs are potentially a more common complication of endomyocardial biopsies than recognized and routine screening should be considered. In our patient, the decision to intervene was based on the presence of symptoms, demonstrable coronary steal on hemodynamic evaluation and progressive reduction of distal coronary flow on serial angiograms. To our knowledge, this is the first reported case of post-transplant CAF with detailed longitudinal hemodynamic, angiographic and cardiac imaging evaluation before and after treatment. The rapid development of in-stent restenosis 5 months postcoronary intervention may be related to the pro-inflammatory state post-cardiac transplantation. Alternatively, underlying pathogenic mechanisms of early cardiac allograft vasculopathy in our patient may have played an important role in early restenosis. The long-term outcome of this clinical approach remains to be determined. Long-term data on CAFs is needed to better understand the natural history of this unique post-transplant phenomenon, and to guide recommendations for screening, monitoring and management.
The authors would like to thank the nurses, technologists and staff at the University of Ottawa Heart Institute for their assistance in obtaining data for this article. A special thanks also to our patient for permission to report his case.
The authors have no disclosures.