Ocular candidiasis is one of the major complications of disseminated candidiasis in liver transplantation (LT) recipients. In this case report, it is aimed to present a patient developing candida retinitis in LT recipient in the first month after LT and successfully recovered with liposomal amphotericin B therapy.
A 52-year-old male patient had been undergone right lobe LT from a living donor. He had diabetes mellitus and underwent serial erythrocyte and thrombocyte transfusions, the score of models for end-stage liver disease (MELD) was 23. On the 14th day, biliary leakage and infected bilioma determined in multislice abdominal tomography. On the 25th day, sepsis symptoms such as fever, tachycardia and hypotension developed. Leukocytosis, elevated C-reactive protein (CRP) and procalcitonin were also present. It was started empirically imipenem/cilastatin, teicoplanin, and fluconazole intravenous therapy. In the three blood cultures taken from fever presence, Enterobacter spp. Enterococcus faecium and Candida albicans (caspofungin-sensitive, fluconazole-resistant, MIK level > 256 µg/ml) respectively were identified. According to the antimicrobial susceptibility test, fluconazole was discontinued and anidulafungin treatment was initiated. On the third day of anidulafungin treatment, the patient having the complaints of headache, decrease in visual acuity. Ophthalmology consultation was requested, as a result of ocular examination candida retinitis was diagnosed in the right eye. There were two focal yellow retinal lesions thought to be candidal plaques in retina and macula. Because of not successful to reach higher concentrations in cerebral tissues, the anidulafungin treatment was altered to liposomal amphotericin B. The antifungal treatment was also administrated intravitreal. The blood culture was sterile at the day of fourteen. All of the complaints downgraded, and the retinitis symptoms recovered at the end of 28-day treatment. So, antifungal treatment was discontinued because of normal ocular examination.
LT recipients have a high risk of disseminated candidiasis, the ophthalmological examination should never be neglected because candida retinitis is a common variant of disseminated candidiasis.
Liver transplantation, Disseminated candidiasis, Retinitis, Ocular complication
Liver transplantation (LT) is a highly effective and cost-beneficial method for the treatment of acute liver failure and chronic end-stage liver diseases [1]. Invasive fungal infections (IFIs) frequently occur after liver, lung, heart and pancreas transplantation, the incidence of IFI's vary from 1.4 to 42% [2]. The common risk factors of IFI's were reported as prolonged and/or recurrent surgeries, retransplantation, haemodialysis, Continuous Ambulatory Peritoneal Dialysis (CAPD), Continue Veno-Venous Haemofiltration (CVVH), irrational use of antibiotics, long-term Intensive Care Unit (ICU) and prolonged stay in hospital, candidal colonization and cytomegalovirus (CMV) disease [3]. Candida albicans is the most common seen candida species, however, Candida glabrata, Candida krusei, Candida parapsilosis and Candida tropicalis, respectively, have also been reported as non-albican species [4].
Disseminated candidiasis is named as a combination of several clinical manifestations such as surgical site infections, peritonitis, intraabdominal abscesses (like liver, spleen, etc.), ocular infections (like endophthalmitis, chorioretinitis etc.), esophagitis and urinary tract infections. Candidemia is known to bring on metastatic ocular infections with potential horrible outcome. Clinical manifestations of ocular infections in LT recipients are often atypical; mixed infections or noninfectious complications may coexist [5]. Ocular candidiasis is a sight-threatening ocular infection that most frequently occurs as a result of complications from disseminated candidiasis. Ophthalmoscopy should be performed in all patients with suspected candidiasis. The real incidence, risk factors, and outcome of eye involvement during disseminated candidiasis are completely unknown [6]. While amphotericin B is considered the gold standard for the treatment of most IFI's, the optimal antifungal management of ocular candidal infections has not been clear yet [7].
In this case report, it is aimed to present the recipient developing C. retinitis and successfully recovered with liposomal amphotericin B within the first month of LT.
A 52-year-old male patient was followed-up for two years in a medical center with the diagnosis of chronic liver failure. He had been diagnosed with diabetes for the last five years. Having a model for end-stage liver disease (MELD) score of 23, the patient has undergone right lobe liver transplantation from a living donor in our institute in April 2019.
On the first day, the standard immunosuppressive treatment protocol (tacrolimus, mycophenolate mofetile, and prednisolone). The serial erythrocyte and thrombocyte replacements were performed in the first week. On the seventh day, trimethoprim/sulfamethoxazole 80/400 mg tablet in a day for antibacterial prophylaxis, and 900 mg oral valganciclovir for antiviral prophylaxis were started. On the 25th day, the patient had fever, tachycardia, and hypotension. Blood analysis was carried out and the results were interpreted as follows leukocytosis (white blood cell (WBC): 12200 109/L), C-reactive protein (CRP): 11.2 mg/dL, and procalcitonin (PCT): 4.51 ng/ml. Therefore, the patient empirically started receiving treatment of 4 × 500 mg imipenem/cilastatin, 1 × 400 mg teicoplanin, and 1 × 400 mg fluconazole intravenous therapy. Enterobacter sp., Enterococcus faecium and Candida albicans (Caspofungin-sensitive and Fluconazole-resistant, MIK level > 256 µg/ml) were grown in the three blood cultures, respectively. All the blood cultures were studied by using the BACTEC 860 system (Becton-Dickinson, Inc., Sparks, MD) and the BacT/Alert 3D (Bio Merieux, Marcy-l'Etoile- France). An antifungal sensitivity test was performed by using the E-test method, and the reference values of the European Committee on Antimicrobial Susceptibility Testing (EUCAST) were taken as a base. As soon as the blood culture was reported by growing Candida, we performed an eyes examination for retinitis exclusion and the result was considered normal. No modification was made in the antibacterial treatment, we, therefore, wanted to switch to liposomal amphotericin B treatment. The antifungal treatment was also administered through intravitreal.
However, liposomal amphotericin B could not be obtained at that time, anidulafungin therapy was initiated to the patient because it was sensitive to all echinocandins. In order to determine the infection focus, portal vein Doppler USG, T-tube cholangiography, and multiple thin section abdominal tomography were studied. The portal vein Doppler USG findings were found to be normal. In T-tube cholangiography, a biliary leakage was detected from the internal segment of the feeding abdomen. In abdominal tomography, partly hyperechogenic liquid (compatible with intraabdominal abscess or infected bilioma) was observed in the form of the perihepatic region. The patient underwent transthoracic echocardiography for the probability of invasive candidiasis, but there was no sign detected for infective endocarditis.
On the 3rd day of anidulafungin treatment, the patient was having complaints of headache and a decrease in visual acuity as a result of this, an ophthalmology consultation was requested, with an ocular examination, candida retinitis was diagnosed with the right eye. There were two focal yellow retinal lesions thought to be candidal plaques in the centre of right retina and macula. The retinal examination images of the patient after the diagnosis are shown in (Figure 1).
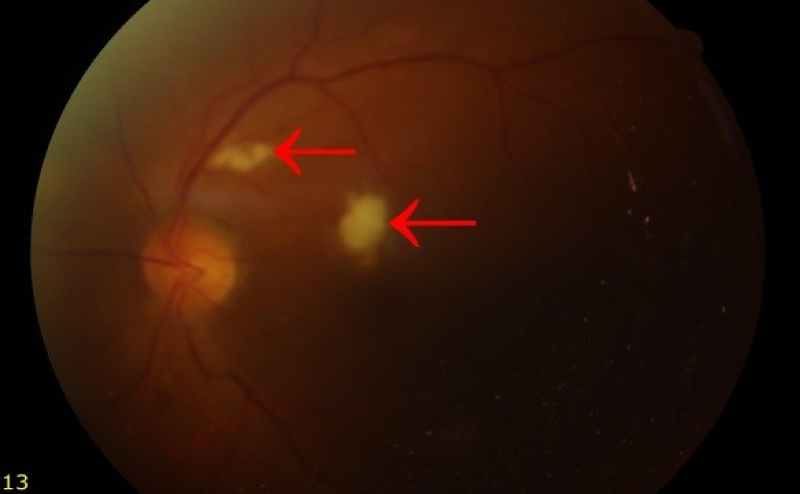 Figure 1: The retinal candida plaques in right eye during the diagnosis.
View Figure 1
Figure 1: The retinal candida plaques in right eye during the diagnosis.
View Figure 1
Since it well passes the brain-blood barrier and reaches high concentrations in ophthalmic tissues, the anidulafungin treatment was stopped and altered to 250 mg/d IV liposomal amphotericin B therapy. The blood culture was sterile at the fourteen-day of treatment, and the antifungal treatment was stopped after achieving the clinical and radiological improvement. During the liposomal amphotericin B treatment, the count blood cell (CBC), liver and kidney function tests were performed on a daily basis and the ophthalmological examinations were performed once in a week. No complication related to liposomal amphotericin B treatment was observed. At the end of the 28-day treatment, all the complaints of the patient were diminished and the retinitis condition recovered. After the treatment, the patient was discharged with full recovery. The retinal examination images of the patient at the end of the antifungal therapy are shown in (Figure 2).
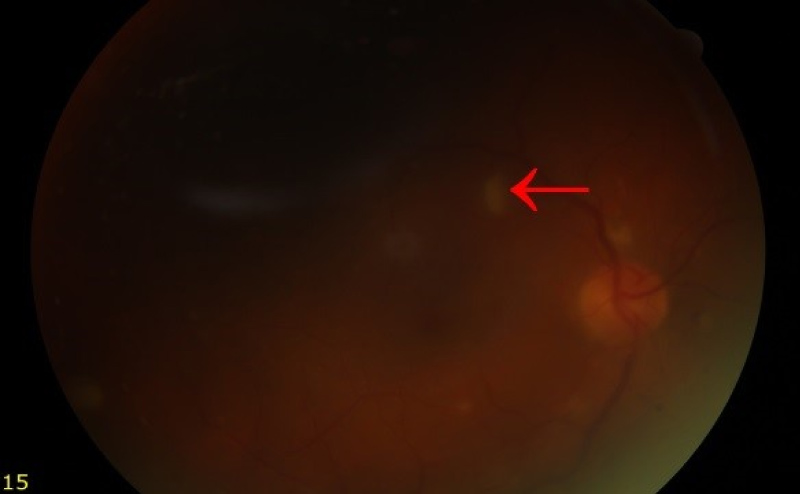 Figure 2: The retinal plaques disappeared after the treatment.
View Figure 2
Figure 2: The retinal plaques disappeared after the treatment.
View Figure 2
Although CMV is the most common opportunistic infectious agent of ocular involvement in LT recipients, ocular candidiasis is also a major complication of disseminated candidiasis [5]. Papanicolaou, et al. reported a 1.3% incidence of ocular infections in LT recipients and declared that 684 patients undergone LT at Mount Sinai Hospital in New York had fungal infections earlier (mean, 42 days) than viral infections (mean, 128 days) [8]. Husain, et al. found that the risk factors for invasive candidiasis include the use of antibiotics to prevent spontaneous bacterial peritonitis, post-transplantation dialysis, and retransplantation [9]. In our case study, the patient had diabetes mellitus and high Model for End-Stage Liver Disease (MELD) score and they underwent serial erythrocyte/thrombocyte transfusions at perioperative and postoperative period. Additionally, biliary leakage, infected bilioma and bacterial sepsis were developed, then broad-spectrum antibiotics were performed. C. retinitis appeared on the 25th day after LT.
Retinal lesions are said to be associated with an episode of candidemia, if they appeared in the first eye examination that was carried out within seven days following a positive blood culture. Ophthalmoscopy has been recommended for diagnosing frequently nondescript diagnosis of disseminated candidiasis. The optic fundus may be affected by an independent opportunistic infection focus or may be part of systemic infection [10]. While the appearance of CMV retinitis is typical with a characteristic vascular sheath, edema and actively extending bleeding borders and lesions of a pale necrotic center, there are extensive haemorrhages and larger lesions in the retinitis associated with disseminated candidiasis or toxoplasmosis. Ocular candidiasis usually presents with distinctive signs demonstrated by funduscopic examination [11]. Libsen, et al. prospectively evaluated five studies about candidemia patients for Candida endophthalmitis, and they declared that the rates from 0-78% of lesions consistent with candidal endophthalmitis in immunocompromised patients. In the same study, 180 out of 206 patients diagnosed with candidemia underwent an ophthalmologic examination in the first week, 27 (15%) patients had identified with retinal lesions such as retinal haemorrhage or a cotton wool spot [12,13]. In our case, it was demonstrated with two focal yellow retinal lesions thought to be candidal plaques extending from inferotemporal to the optic nerve and center of macula in the right eye.
There are several treatment options such as systemic antifungal therapy, or with intravitreal injection of an antifungal agent, rarely combined with vitrectomy for ocular candidiasis [14]. C. retinitis can be treated in two ways: Liposomal amphotericin B formulation combined with flucytosine is used for advances lesions, cases with less serious severity can be treated with fluconazole. The recommended duration of therapy is at least 4 to 6 weeks [15]. When our case had developed sepsis, findings shown that there are no growing candida species in the blood cultures, however, we initiated empirically fluconazole along with antibacterial therapies. Therefore, we had to switch the therapy with liposomal amphotericin B and administrated the drug for a total of 28 days according to guidelines.
The LT recipients having candida species growing in the blood cultures should never be neglected. Retinal examination by an ophthalmologist in the first week of culture positivity and the eyes screening must be performed. Determining the involvement of candidemia in the eyes in early period and implementing the appropriate treatment strategy is of significant importance. However, further studies including more transplant group of patients are needed.
I would like to thanks to Professor Doctor Abuzer Gündüz, who allowed me to use his retinal images.
The authors have no conflicts of interest to disclose.
This research was not support by any firm or person.
The data that support the findings of this study are available from the corresponding author upon reasonable request.