Unavailability of deceased donors in the east has led to a rise in the popularity of living donor liver transplantation (LDLT). Preventing postoperative complications is a major concern in LDLT's. There are reports of fatal pulmonary embolism which has happened in Living Donors. Moreover, a study examining thromboelastogram (TEG) patterns in 10 liver donors in 2004 showed a significant development of hypercoagulability in 6 patients. We attempted to analyze the coagulation status of living donors through thromboelastography.
80 consecutive patients who underwent right donor hepatectomies were enrolled in the study. In addition to the baseline investigations and demographics, TEG analysis was done on the day of surgery and POD1, 3, 5 & 7 respectively. Postoperative blood counts, liver function tests & PT-INR were also recorded & statistically analyzed.
All patients were hypercoagulable at postoperative day (POD) 1 & 3. 98.8% of patients were hypercoagulable in POD5 & 67.5% of patients in POD7. None of the patients were hypo coagulable in spite of an increased prothrombin time/international normalized ratio (PT INR) & decreased platelet. R time & k-time had significantly shortened throughout the post-operative period whereas alpha angle and MA had significantly increased. The Coagulation Index (CI) demonstrated maximum hypercoagulability in POD1 (5.17 ± 0.75), gradually decreasing towards POD7 maintaining hypercoagulability throughout.
Despite routine laboratory investigations suggesting hypocoagulability, TEG monitoring showed significant hypercoagulability in all the patients who underwent right donor hepatectomies. These findings have a far-reaching impact on donor safety post living donor liver transplantation. We strongly recommend early postoperative mobilization and antithrombotic prophylaxis on all patients undergoing donor hepatectomies.
Living related liver transplantation, Post-operative complications, Thromboelastogram, Hypercoagulability
ASA: American Society of Anesthesiologists; BMI: Body Mass Index; CCT: Conventional Coagulation Tests; CI: Coagulation Index; DVT: Deep Vein Thrombosis; LDLT: Living Donor Liver Transplantation; MA: Maximal Amplitude; POD: Post-Operative Day; PT: Prothrombin Time; PTT: Partial Thromboplastin Time; TEG: Thromboelastogram; TRALI: Transfusion-Associated Lung Injury
Living related liver donations have become common practice in many Asian countries owing to the scarcity of cadaveric organs and lack of public awareness. Since this operation involves taking a part of the liver from a healthy donor, donor safety assumes paramount importance. In spite of surgical and anesthetic techniques being refined and post-operative monitoring becoming more efficient, there are a few reports of donor morbidity and mortality which require special attention. Fatal pulmonary embolisms have been attributed as causes of donor mortality by Emere, et al. [1] & Streneck, et al. [2] However, conventional coagulation tests tend to show a picture leaning towards hypocoagulability during the postoperative course of these patients. Hence, there seems to be an under-appreciation of the prothrombotic risks associated with Living related Liver donation.
Thromboprophylaxis, which is included in standard protocols of postoperative care of prolonged operative procedures, is largely withheld or delayed because of the risk of bleeding due to increased prothrombin time/international normalized ratio (PT/INR) [3-6]. However, reports of postoperative clinically significant bleeding strictly due to coagulopathy in living donors are lacking. On the other hand, there have been several reports of postoperative thrombotic events in living donors, some of them even lethal. Donor deaths have been reported related to deep venous thrombosis (DVT) and pulmonary embolism [7]. The reported incidence of pulmonary embolism is around 7% [8]. Additionally, thrombosis of splenic vein and portal vein has also been reported in these patients [8]. Overall, there has been an under appreciation of thromboembolic risks in donors. These patients do not undergo routine screening for postoperative coagulopathy despite relatively higher risks involved.
Apart from the risks related to bleeding, many centers either question or recommend cautious use of epidural analgesia due to the presence of coagulopathy [9]. Some institutes practice plasma transfusions prior to removal of the catheter in view of deranged PT/INR. Blood transfusion further increases the risks of infection, transfusion-associated lung injury (TRALI), fluid overload, anaphylaxis, etc. in this subset of patients [10,11].
The change in liver function tests usually varies, however it is considered to be directly proportional to the extent of liver resection. Liver parenchymal enzymes such as Serum Glutamic Oxaloacetic Transaminase (SGOT), Alkaline Phosphatase (ALP) and total bilirubin, showing derangements even with the smaller wedge resections, are bound to increase after major right donor hepatectomies, which invariably involves removal of up to 70% of hepatic tissue from donor [12]. Resection usually follows a decrease in the synthetic function of the liver, and thus is responsible for the impaired synthesis of clotting factors and regulatory proteins in the coagulation pathway.
Thromboelastogram (TEG) is a bedside blood test that provides a point of care measurements about the viscoelastic properties of blood. It is able to provide information about platelet activation, fibrin formation, and clot retraction. It has been used extensively to guide therapy of coagulation disorders in liver transplantation [3], cardiac surgery [13], orthopedic [14] and pre-eclamptic parturients and is also one of the most sensitive methods for detecting and monitoring postoperative hypercoagulability.
The reasons for the altered coagulation pathway in donor hepatectomies are not thoroughly understood and thus are difficult to predict. Impaired synthesis of both pro- and anticoagulants following hepatectomies is largely considered the reason for this alteration. Extensive tissue trauma, acute phase response, mild blood loss, and hemodilution all have been implicated as the reasons for hypercoagulability. TEG has been studied in liver donors but with very limited sample size. Most other studies have been performed on patients undergoing liver resections for various malignancies [4].
The study was designed as a prospective observational trial. After approval by the institutional Ethics Committee, 80 consecutive patients undergoing modified right donor hepatectomy were included in the trial. Written informed consent was taken. Demographic data was collected like age, sex, co-morbidities, history of smoking, oral contraceptives, BMI, ASA (American Society of Anesthesiologist) classification.
The anesthetic technique was standardized as follows: Upon admission to the operating room and after standard basic monitoring, midazolam (0.05 mg/kg) and fentanyl (2 mcg/kg) were given intravenously. The induction of anesthesia was done with propofol (2 mg/kg) and atracurium (0.5 mg/kg) to facilitate endotracheal intubation. Anesthesia was maintained with isoflurane in air: Oxygen mixture to maintain a MAC of 1.0 and atracurium (4-12 µg/kg/minute) infusion. An arterial line was routinely inserted into the left radial artery, central venous pressure was accessed with 14G cavafix in left median cubital vein. All patients received Ringer's acetate and 5% dextrose in normal saline as fluids to maintain the CVP between 4-8 mmHg. Low CVP was maintained during the hepatic resection. Gelatin was used to replace blood loss. Patients were provided mechanical thromboprophylaxis in the form of elastic stockings and intermittent pneumatic compression devices. All patients received 5000 IU unfractionated heparin 3 min prior to vascular clamping. Warming devices like bair hugger and fluid warmer were used to maintain the temperature around the normal range (35-37 ℃). Patients received multimodal analgesia intraoperatively as well as postoperatively. Intraoperative morphine was used as per the judgment of attending anesthesiologist. Paracetamol and diclofenac sodium were also used.
The surgical technique was standardized for all donors. Open Right hepatectomy along the cantles line was performed, including segments 5 and 8, without involving the middle hepatic vein. Briefly, an ultrasonic dissector, and argon laser were used to divide the liver parenchyma. No Pringle maneuver was performed. All liver grafts were perfused on the back-table with University of Wisconsin solution.
All living donors were extubated in the operating room. The aim was to achieve a visual analog pain score of 2 or less. Intravenous morphine sulfate (0.05 mg/kg) was administered for rescue analgesia. All patients received patient-controlled analgesia with morphine sulfate through pump postoperatively along with diclofenac sodium 75 mg twice a day. Thereafter patients were shifted to intensive care unit where they were monitored for the next 3 days before being shifted to the ward. Mechanical thromboprophylaxis was continued until the patient is in ICU. None of the patients received pharmacological thromboprophylaxis. We follow enhanced recovery protocol for all donors and thus mobilization out of bed is done on day 1. Perioperative crystalloid, colloid and blood transfusions (packed red cells, fresh frozen plasma, platelets, and cryoprecipitate) were recorded.
The TEG analysis was performed 5 times: Before the skin incision baseline (before surgical incision) (D0)], on postoperative day 1 (D1; 24 hours after surgery), and on a postoperative day 3 (D3; 72 hours after surgery), day 5 and day 7. The Conventional Coagulation Tests (PT, INR, and platelets) were also measured at the same time points. Apart from CCTs liver enzymes (SGOT, SGPT, ALP) were also checked at these time points. Thromboelastography analyzer 5000 (Haemoscope was used to perform TEG. The test was done using non-citrated fresh sample utilizing Kaolin to accelerate the clotting. The primary team was blinded to the results of TEG and thus were not allowed to change the clinical management based on the results so obtained.
Various parameters from TEG analysis were recorded. 5 main parameters include- reaction time (r-time normal range 4-8 minutes), clot formation time (k-time normal range 0-4 minutes), a-angle (normal range - 47 - 74 degrees), Maximal amplitude (MA normal range 54-72 mm) and lysis at 30 minutes (LY30 normal range 0-8%), TEG coagulation index (CI normal range +3 to -3) describes patients overall coagulation status as derived from these aforementioned parameters by means of a mathematical equation. Value of more than +3 indicates hypercoagulability and less than -3 indicates hypocoagulability.
CI = -0.3258R - 0.1886K + 0.1224MA + 0.0759a - 7.7922
SPSS v22 (IBM corporation) software was used to analyze the data. Continuous variable is represented as mean ± SD. Categorical variables are represented as number and proportion. Paired t-test with 2 tailed sig. is used to compare the change in continuous variable over time. Chi-square test is used to compare categorical data sets.
A total of 80 consecutive patients undergoing right modified hepatectomy were enrolled in the study from January 2017 to October 2018. Mean age was 30.4 years. There were 53.8% male and 46.2% female patients who underwent donor hepatectomy during this time period. Mean BMI was 27.5. Majority of the patients (72.5%) belonged to the American Society of Anesthesiologists ASA1 category. Mean duration of surgery was 586 minutes. Average blood loss of around 800 ml. Only 2 patients received intraoperative packed red cell transfusion. Mean CVP was maintained around 4 cm H20. Intraoperative variables are given in (Table 1). Mean liver remnant and graft weight was 38% and 761g, respectively.
Table 1: Patient demographics and operative findings. View Table 1
Preoperative r-time, time to initiation of clot formation, is a reflection of coagulation factor activity was within normal range i.e. 4-8 min. On subsequent days from day 1 to day 7, as shown in (Figure 1a) there is a progressive decrease in r-time and it is statistically significant with p-value < 0.005. k-time represents the time take to achieve a certain level of clot strength due to thrombin and activation of platelets usually it is between 0-4 min. (Figure 1b) depicts preoperative mean k-time in our study group is 2.76, it slowly decreases till day 7. The decrease in the k-time as compared to the baseline is also statistically significant.
 Figure 1a: R-time.
View Figure 1A
Figure 1a: R-time.
View Figure 1A
 Figure 1b: K-time.
View Figure 1B
Figure 1b: K-time.
View Figure 1B
α-angle that measures the speed at which fibrin builds up and cross linking takes place (clot strengthening) and thus, assesses the rate of clot formation remained in the normal range (47-74). However as seen in (Figure 1c), it kept rising progressively till day 3 and decreased only slightly thereafter by day 7. This hike was again significant as compared to the baseline values.
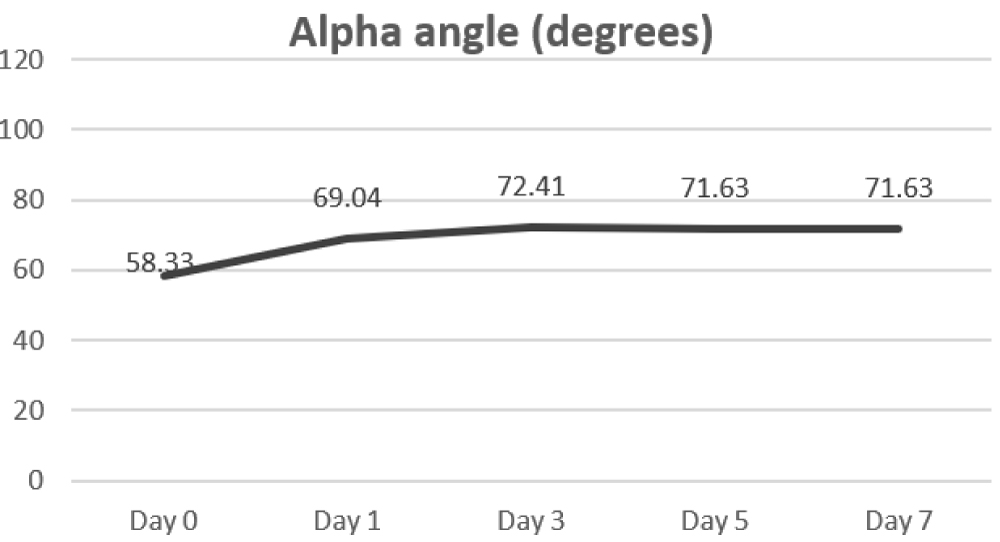 Figure 1c: Alpha angle.
View Figure 1C
Figure 1c: Alpha angle.
View Figure 1C
Maximum amplitude (MA) is a function of maximum dynamic properties of fibrin and platelet bonding via GPIIb/IIIa and represents the strongest point of the fibrin clot and correlates with platelet function. Change in MA over the course of 7 days is given in (Figure 1d), and it steadily increases from baseline of 50.48 to 67.75. LY 30 depicts the percentage fall in amplitude 30 min after MA and gives the idea about the degree of fibrinolysis. As shown in (Figure 1e), it was found to be increased significantly on day 1 and it slowly returned to normal by day 7.
 Figure 1d: MA.
View Figure 1D
Figure 1d: MA.
View Figure 1D
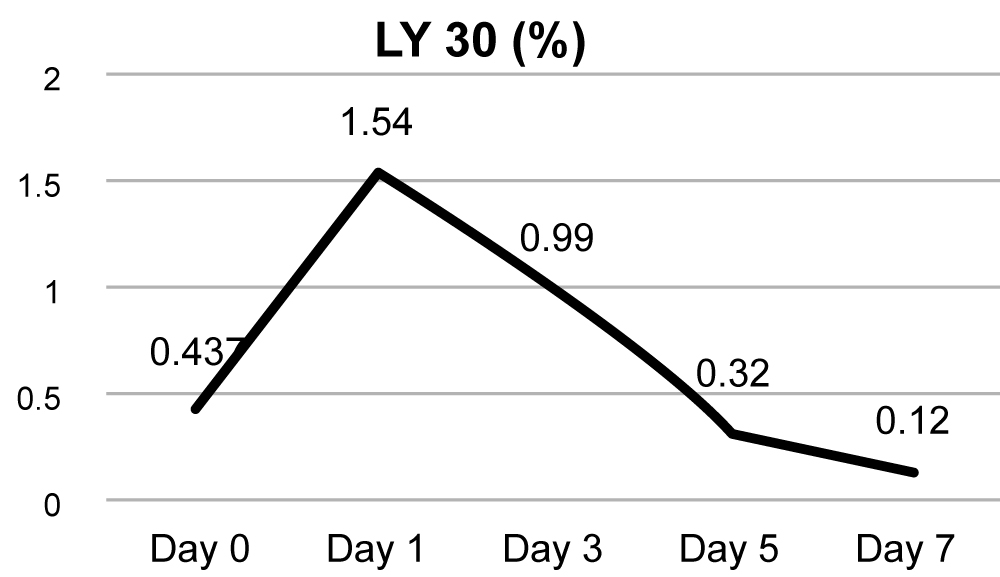 Figure 1e: LY-30.
View Figure 1E
Figure 1e: LY-30.
View Figure 1E
Overall coagulation index is given in (Figure 2a) Mean CI of the study population was normal prior to surgery on day 0 was 2.48, Coagulation index reaches its peak on Day 1- 5.17 and then slowly decreasing throughout the next week. However, it stays elevated from baseline even at the end of day 7. Evolution of coagulopathy is highlighted in (Figure 2b) All patients were in a hypercoagulable state as per TEG indices on day 1, 3 and 5, with the only total of 26 patients returning to baseline normocoagulable state on day 7.
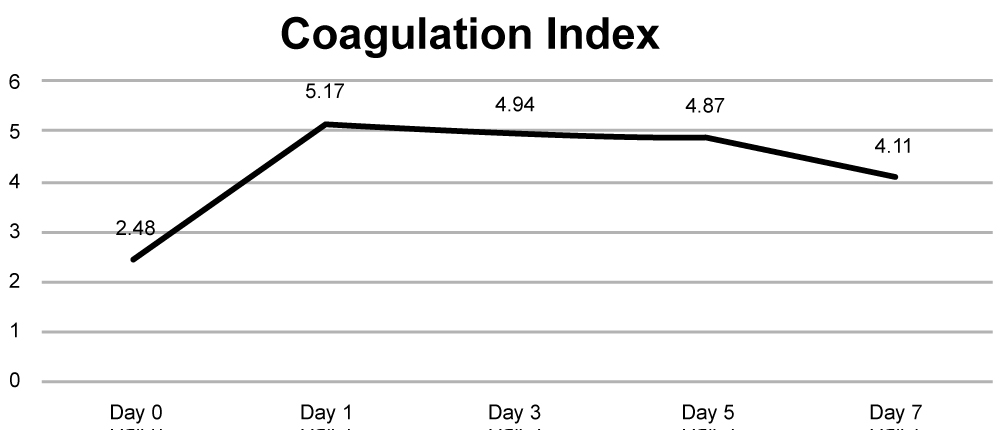 Figure 2a: Coagulation Index.
View Figure 2A
Figure 2a: Coagulation Index.
View Figure 2A
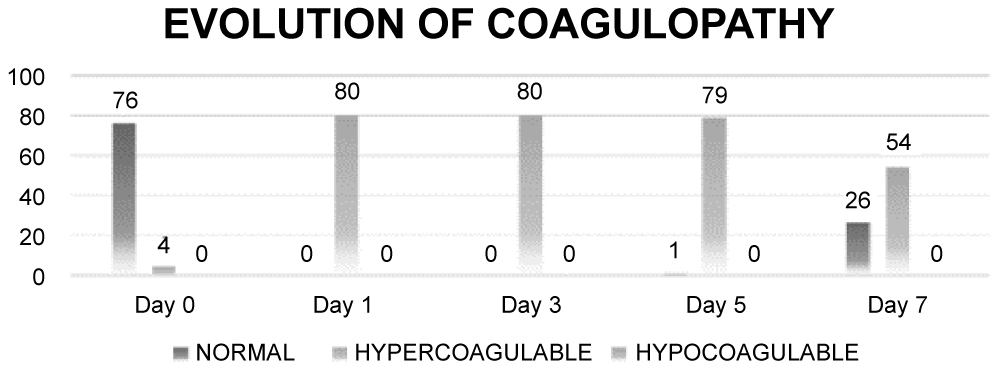 Figure 2b: Evolution of coagulopathy.
View Figure 2B
Figure 2b: Evolution of coagulopathy.
View Figure 2B
On contrary to these findings the conventional coagulation tests portray a very contradictory picture. As shown in (Figure 3a) prothrombin time and INR slowly increased and reached a peak on day 3 and starts to decrease after that, thus representing a highly hypocoagulable state. Liver enzymes (Figure 3b) follow the same pattern, peaking by day 3 before normalizing. Quantitative platelet count and hemoglobin (Figure 3c and Figure 3d respectively) remain in the normal range despite a slight fall in number from the baseline day 0 values.
 Figure 3a: INR.
View Figure 3A
Figure 3a: INR.
View Figure 3A
 Figure 3b: Liver enzymes.
View Figure 3B
Figure 3b: Liver enzymes.
View Figure 3B
 Figure 3c: Haemoglobin.
View Figure 3C
Figure 3c: Haemoglobin.
View Figure 3C
 Figure 3d: Platelets.
View Figure 3D
Figure 3d: Platelets.
View Figure 3D
There were no major surgical complications in the study group. After the surgery, all patients were managed in the Intensive care unit. No transfusion was required in ICU. Despite being hypercoagulable by thromboelastography, none of these patients showed any signs of DVT and or pulmonary embolism.
Living donor-related liver transplant in our country is a norm rather than the exception for patients with end-stage liver disease, especially due to the scarcity of cadaveric donors. Left hepatectomy that involves resection of only smaller or fewer segments can be used mainly for pediatric liver transplantation and is responsible for less severe effects on donor's hepatic function. Whereas, right hepatectomy involves resection of 60-70% of liver tissue, thus, amounting to dramatic derangement of hepatic function.
In this study, we found that there is significant hypercoagulability present in liver donors which is not seen with conventional coagulation tests. Previously, investigators have looked into this phenomenon. Cerutti, et al. in 2004 presented the first of its kind study showing the hypercoagulability in liver donors. They evaluated 10 patients undergoing right hepatectomy for LDLT [3]. They observed that despite increased PT/INR, normal APTT and decreased platelet TEG showed progressive hypercoagulability in 4 subjects on day 5 and 6 subjects on day 1. One of the donors from the study group developed DVT which was resolved with a therapeutic dose of Low Molecular weight heparin. Small sample size was a limitation of the study.
Another study done by Barton, et al. was published in 2012 which primarily looked into TEG profile of 40 patients undergoing hepatectomies for various malignancies [15]. They found that there was normal TEG with transient relative hypercoagulability, despite increased PT/INR in the postoperative period. However, this study group consisted of patients with malignancies and hence, the results cannot be directly extrapolated to the donor hepatectomy patients. In this study, there was also no standardization with regard to surgical procedures.
Procoagulants, as well as anticoagulants both, are manufactured in the liver. Apart from that, there is upregulation of non-hepatically synthesized coagulation factors like factor VIII and Von Willebrand factor, the liver also removes activated clotting factors from blood and thus, helping balance the procoagulant and anticoagulant mediators [8].
Despite rampant hypocoagulability often seen through the postoperative rise of PT/INR, clinically significant bleeding has hardly been reported. Even in the present study, we found that PT/INR of patients started rising on day 1 and reached a peak around day 3 (median INR 2.2) before returning to normal by day 5. We did not find any clinically significant bleeding in the postoperative period and none of the patients required blood transfusion in the ICU. However, there have been reports of pulmonary embolism within 1st few months. Deep vein thrombosis, splenic and portal vein thrombosis have also been reported [8].
TEG has been used for evaluation of coagulopathies- acquired or congenital when compared to healthy controls [16]. Various biomarkers like protein C, protein S, SPS protein, anti-thrombin III, have been speculated to be playing a pivotal role in the coagulation abnormalities following liver resection [17-19]. Further studies are required especially with regards to serum levels of these biomarkers to find out the reasons for the hypercoagulability in donors.
Apart from hypercoagulability seen on TEG, there were 2 more remarkable findings in this study. Firstly, most these patients remained hypercoagulable even on day 7. Only 26 patients returned to baseline normal coagulation index. Most patients are discharged on day 8 following the surgery. Secondly, we did not find any clinical evidence of DVT or PE in this group. However, long-term follow-up for the purpose of the study wasn't done.
To conclude, the best practice as of now would be to fully evaluate Living Related Liver donors for all acquired or inherited coagulation disorders. Postoperatively coagulation status must be monitored by thromboelastogram in addition to the conventional coagulation tests because there is discrepancy between the two aforementioned tests. It would be wise to put liver donors on both mechanical and pharmacological thromboembolic prophylaxis post operatively. Keeping in mind that majority of our patients demonstrated a hypercoagulable state even at the end of first week, we recommend close follow up of these patients after discharge for thromboembolic events.
No potential conflict of interest relevant to this article was reported.
No external funding was needed.