Summary: Anesthetic management must meet hemodynamic and pharmacological goals during renal transplantation; maintain intravascular volume; the perfused graft; control factors that influence its function. Objective: Determine if the handling of liquids; the transfusion; the hemodynamic values; the administration of diuretics and vasopressors significantly influence the optimal decrease in creatinine.
Material and methods: Case-control study of patients transplanted between 2012-2017; of the anesthetic record and clinical record were collected: Demographic variables, central venous pressure, mean arterial pressure, use and dose of diuretic, fluids, vasopressors and transfusion. It was classified into: Group A (without optimal creatinine decrease); Group B (with optimal decrease in creatinine); we collected creatinine value at 24, 48 and 72 hours after transplant; we divide each group according to the origin of the graft. Descriptive and inferential statistics (X2 and T of student, ANOVA).
Results: We included 391 transplant patients of 504; of 337 (86.1%) living donor receptors, 171 (43.7%) had optimal creatinine decrease (< 2.5 mg/dl) at 72 hours and 166 (42.4%) did not optimal decrease; of 54 transplants (13.8%) of cadaveric donors, 26 (6.6%) had optimal descent (< 3 mg/dl) and 28 (7.1%) had no optimal descent. Administer hydroxyethyl starch 6% (p = 0.048), blood transfusion (p = 0.008;) and furosemide (p = 0.0001;) were associated with no optimal decrease, and the administration of vasopressors and mannitol (p = 0.0002; 95%).
Conclusions: It is necessary to standardize the anesthetic management of the kidney transplant and to develop management guidelines that favor the prognosis and survival of the graft.
Kidney transplantation, Anesthesia for kidney transplantation
Kidney transplantation is the treatment of choice for end-stage chronic kidney disease [1]. According to Wolf, et al. [2], patients with chronic kidney disease on dialysis have mortality 2.6 times higher than patients on the waiting list, and patients on the list have mortality 1.7 times higher than post-transplant patients. In the same way, the mortality rate of the transplanted patient is 69% less than the dialysis patients, and the survival increases 4.3; 2.8 and 1 years in the age groups of 60-64, 65-69 and 70-74 years, respectively.
The management guides of the renal recipient patient during the transanesthetic vary from one hospital center to another, since there is no consensus on the ideal anesthetic management. It is preferred to use drugs with independent elimination of renal function and that their metabolites do not accumulate, since the pharmacokinetics will be altered since the intravascular volume is affected by dialysis and an inadequate intrinsic vasoconstrictor response is also generated [3]. The ideal anesthetic technique should maintain hemodynamic stability in all phases of kidney transplant surgery, along with water management, which is essential in the maintenance of tissue perfusion, with special attention to the renal graft, as it will directly determine the production of urine [4].
According to the American Society of Anesthesiology (ASA), standardized monitoring during major surgery includes central venous catheter and monitoring of invasive blood pressure, aimed at hemodynamic management and control of fluid therapy [3].
In 2015, Hendrikus, et al. reported that static parameters such as central venous pressure (CVP) should be maintained between 8 to 15 cmH2O [1], and be adjusted for heart failure up to 12 mmHg, without losing sight of the limitations of measuring only preload of the right ventricle; Mean arterial pressure (MAP), at levels greater than 90 mmHg during reperfusion and, in addition, the use of intravenous crystalloid solutions, with 250 ml boluses that gradually increase intravascular volume (0.9% sodium chloride is preferred), however, the use of large volumes (2-3 liters) can lead to hyperchloremic acidosis, resulting in a decrease in the glomerular filtration rate; the use of 0.9% saline solution and Ringer's lactate in a 1-1 proportion to also avoid the hyperkalemia that could be produced by reduced sodium solutions. Fluid therapy is a challenge for the anesthesiologist. The hemodynamic parameters and the intravascular volume loss are dynamic during surgery, and unexpected. The use of hydroxyethyl starch is not recommended in critically ill patients, since it has been observed that it increases the risk of the appearance of kidney damage [5] (Level of evidence 2B). The use of albumin should be limited, since it has not shown benefits compared to crystalloids for volume expansion [6].
Blood transfusion should be avoided as much as possible for the renal recipient, however it may be necessary in patients with significant surgical bleeding or hemoglobin values less than 7 mg/dl (less than 8 mg/dl in patients with significant cardiovascular disease). In case of transfusion, washed or allogeneic red blood cells reduced in leukocytes are recommended [7].
Hemodynamic management should guarantee perfusion of the graft; there is a period of hypotension that anticipates the clearing of the renal vessels and reperfusion of the graft. This has been explained by the sudden change of around 25% of cardiac output to the renal graft and the release of accumulated vasodilator mediators during the period of renal ischemia. The use of vasopressors is minimized, since alpha agonists such as noradrenaline can negatively interfere with renal perfusion and affect graft function. If a vasopressor is necessary for hypotension refractory to volume administration, ephedrine from 5 to 10 mg intravenous is preferred, with repeated doses as necessary. Dopamine is not beneficial for the function of the graft because it has harmful potential, however in cases of hypotension refractory to the previous measures, an infusion can be used at low doses (for example 3 to 5 mcg/kg/min) [8]. Dobutamine can be used as a positive inotropic agent for patients with low cardiac output, in whom advanced hemodynamic monitoring can optimize the use of inotropic and volume drugs.
Anesthetic management has not yet been standardized; there is a need to know the different drugs and anesthetic techniques that are used. General anesthesia has been the most used and has been combined with epidural analgesia, taking into account that patients who have undergone hemodialysis can maintain circulating heparin levels, in addition to the platelet dysfunction induced by uremia, reasons why the Neuraxial anesthesia is not often used in these patients [9].
The use of diuretics is not mandatory; there may be no benefit in using furosemide in patients with renal failure and oliguria, but more studies focused on renal recipient patients are needed. The administration of 200 to 250 ml of 20% mannitol immediately before reperfusion has shown an increase in renal perfusion pressure and has resulted in lesser occurrence of acute renal failure in kidney transplant surgery [8].
After the end of the anastomosis, the production of urine should be monitored, immediate uresis is achieved in 90% of the kidneys of living donors and 40-70% of cadaveric donors. Early uresis and a good urinary volume improves the viability of the graft (75% survival with immediate diuresis compared to 49% in the first 12 hours) and post-transplant survival (13% mortality rate in one year in patients with delayed diuresis versus 7% in patients without delay) [4].
By having variable recommendations regarding the transanesthetic management of fluids and medications, it is essential to analyze and evaluate the repercussions of the different approaches on the evolution of the patient.
The decrease in creatinine is expected in the first hours after transplantation, considering an optimal decrease in the live donor at 24 hours of 25% of the presurgical value, at 48 hours at least decrease of 30% more and at 72 hours levels below of 2.5 mg/dl. In the cadaveric donor recipient, the expected decrease at 24 hours is at least 10%, in 48 hours, 10% more and at 72 hours maximum creatinine levels of 3 mg/dl. In addition, in any type of donor, non-optimal levels of creatinine decrease are considered at values below 10% per day for 3 consecutive days or the increase in creatinine values on any day of the postoperative period, diuresis less than 1 liter in 24 hours on the first day or less than 75 ml/hr in the first 2 days, together with the urgency of dialysis in the first 72 hours [10].
The patient with end-stage chronic renal failure and who enters the operating room to undergo kidney transplantation, represents a challenge for anesthetic management; The phases of this surgery require the fulfilment of hemodynamic goals, the base acid, electrolyte and pharmacological equilibrium so that the outcome is the functioning of the graft.
Cases and controls study; retrospective, descriptive, transversal and comparative. It included all patients who underwent kidney transplantation.
In Mexico, despite the rise in kidney transplantation, survival and the culture of donation, we do not have exact data that explain the phenomena surrounding the short-term success of kidney transplantation; this hospital center performs more than 100 kidney transplants a year and it is necessary to know what factors intervene in the prognosis of the graft. The search of the data included patients who were transplanted in the period from January 2012 to January 2017, from a living or cadaveric donor, in the Specialty Hospital "Dr. Antonio Fraga Mouret "of the High Specialty Medical Unit of the National Medical Center La Raza.
All transplant patients were included in the study period; those who did not have a trans-anesthetic record or complete clinical file were excluded and all patients who had complications during transplantation or postsurgical in the first 72 hours after renal transplantation were eliminated from the study: Obstruction or leakage of the urinary tract, arterial or venous vascular dysfunction of the graft, rejection, re-surgical intervention.
It was authorized by the Local Research Committee.
All the variables were collected: age, gender, weight, height, BMI, physical state of the ASA, creatinine value in mg/dl at 24, 48 and 72 hours after transplantation; type of fluids administered; ischemia times, cold or hot; if there was spontaneous uresis; central venous pressure and invasive mean arterial pressure; use of vasopressor or inotropic; use of diuretics; and they concentrated on a data collection sheet designed for this purpose and a database for statistical analysis was developed.
Kidney receptors in the period January 2012 to January 2017.
Once the groups were established, all the values and variables were collected in the data collection sheet; descriptive and inferential statistics, multiple regression, X2 and Student's T, and NOVA were applied for the independent variables that influenced the outcome and a 95% confidence interval was sought, considering a p less than or equal to 0.05 as statistically significant. And they were analyzed with Graph Pad Prism 6 for Windows.
In the study period, 504 kidney transplants were performed. 391 patients entered the study because the rest had not completed their clinical file, of them 337 (86.1%) received kidney graft from a live donor and 54 (13.8%) from a cadaveric donor. In the case of live donor transplantation, 171 patients (43.7%) had optimal creatinine decrease translated as values below 2.5 mg/dl at 72 hours after surgery and 166 patients (42.4%) had a non optimal decrease. When the donor was cadaveric, 26 patients (6.6%) had optimal creatinine decrease, values below 3 mg/dl in the first 72 hours and 28 patients (7.1%) had no optimal decrease (Figure 1).
 Figure 1: General diagram of the study. * It is considered an optimal decrease of creatinine at 72 hours after kidney transplantation, at values of less than 2.5 mg/dl in a live donor patient and less than 3 mg/dl in a cadaveric donor.
View Figure 1
Figure 1: General diagram of the study. * It is considered an optimal decrease of creatinine at 72 hours after kidney transplantation, at values of less than 2.5 mg/dl in a live donor patient and less than 3 mg/dl in a cadaveric donor.
View Figure 1
Of the total number of patients included, 236 were male (60.3%) and 155 (39.5%) female (Table 1 and Table 2).
Table 1: Demographic data. View Table 1
Table 2: Demographic data. View Table 2
Of the 391 patients, 18 were in predialysis at the time of transplant and 373 (95.4%) already with renal function substitution therapy: 197 patients (50.3%) on hemodialysis and 176 (45.01%) in peritoneal dialysis. 5 patients (1.2%) had a previous transplant.
According to the anesthetic technique used, 349 (89.2%) patients were given balanced general anesthesia; 39 (9.9%) with total intravenous anesthesia (TIVA) and regional anesthesia 3 patients (0.7%).
The causes of Chronic Renal Insufficiency that were identified were: Bilateral Renal Hypoplasia 266 cases (68%), cause not determined in 53 patients (13.5%) and thirdly preeclampsia with 20 patients (5.11%). The remaining causes, 13%, were: Diabetes Mellitus, Polycystic Kidney Disease, Glomerulonephritis, Vesicoureteral Reflux, Systemic Lupus Erythematosus and Congenital Adrenal Hyperplasia.
When analyzing the final water balance, no statistically significant difference was found p = 0.53 between live or cadaveric donor transplants; the mean income was 73.13 ml/kg of weight (Figure 2).
 Figure 2: Milliliters administered per kilogram of body weight.
View Figure 2
Figure 2: Milliliters administered per kilogram of body weight.
View Figure 2
The use of 6% hydroxyethyl starch during the transanesthetic was statistically significant p = 0.048.
Of the 337 patients in the study, 161 patients were administered hydroxyethyl starch and it was observed that of all of them, those who did not have an optimal decrease in creatinine, when the donor was alive, were those who received an average of 10.99 ml/kg or more and 15.58 ml/kg of weight when the donor was cadaveric; in contrast, of this same group, patients who had optimal descent, received 9.22 ml/kg and 8.84 ml/kg, from live and cadaveric donors respectively (Figure 3 and Figure 4).
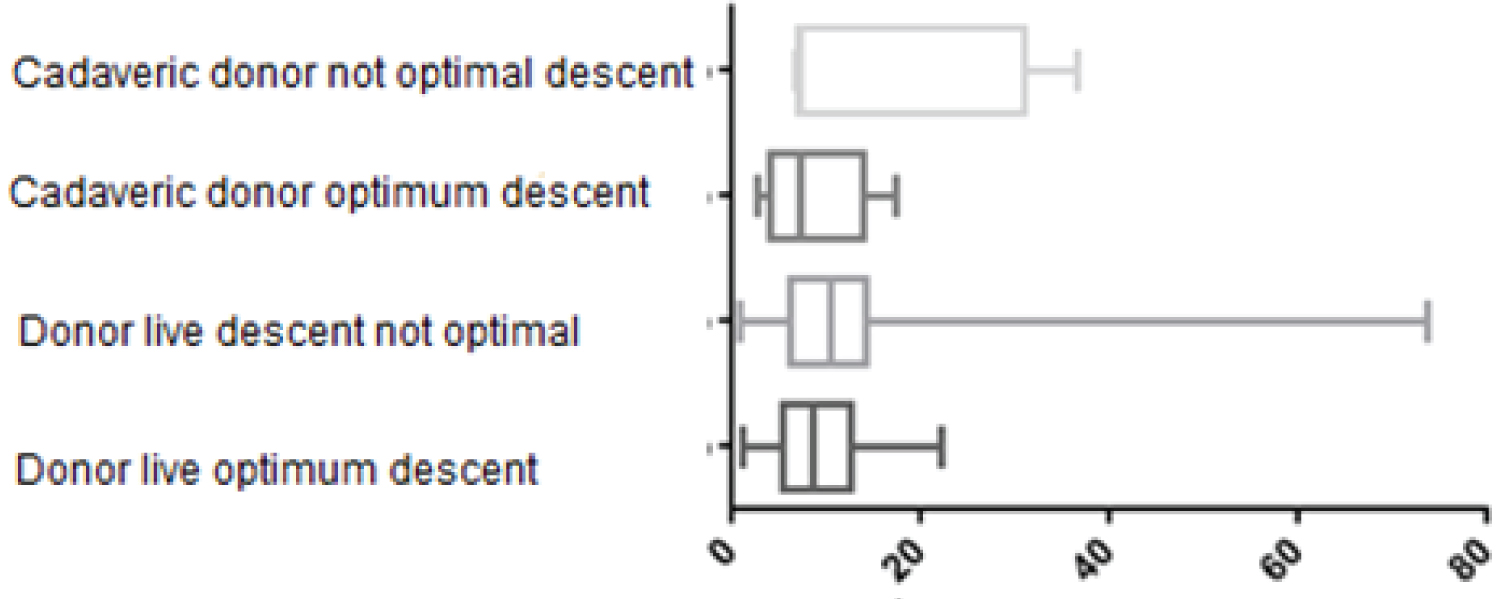 Figure 3: Use of 6% hydroxyethyl starch in ml/kg of weight.
View Figure 3
Figure 3: Use of 6% hydroxyethyl starch in ml/kg of weight.
View Figure 3
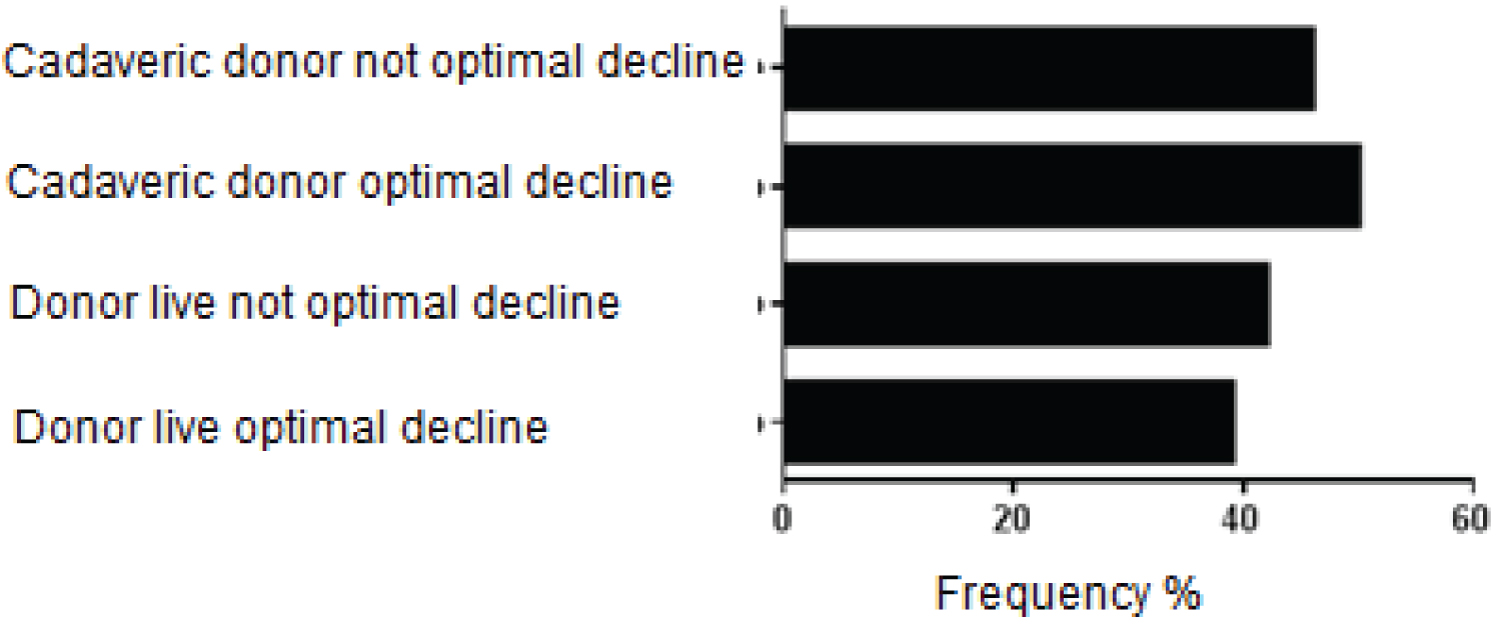 Figure 4: Percentage of patients who received hydroxyethyl starch according to the study groups.
View Figure 4
Figure 4: Percentage of patients who received hydroxyethyl starch according to the study groups.
View Figure 4
There was no significant difference with respect to bleeding during the surgical procedure (Figure 5).
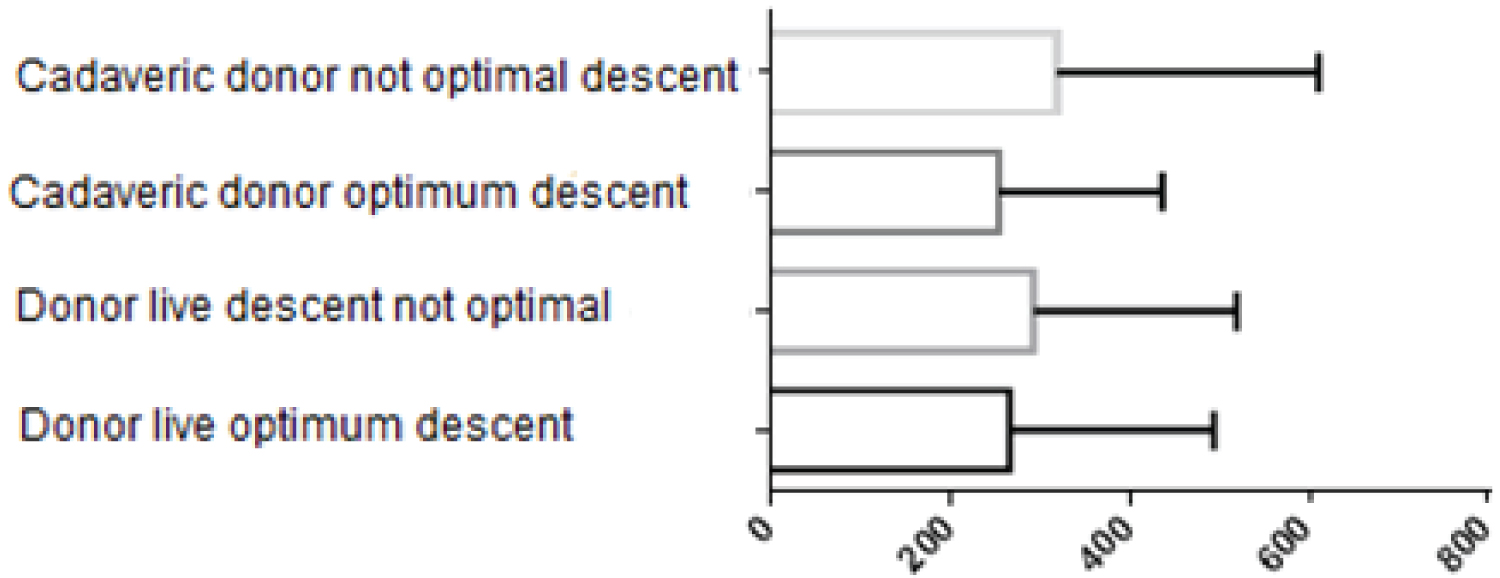 Figure 5: Difference of bleeding during the transplant. p = 0.59.
View Figure 5
Figure 5: Difference of bleeding during the transplant. p = 0.59.
View Figure 5
With regard to blood transfusion (113 patients transfused during the transanesthetic), it was statistically significant that there was a negative impact on the optimal decrease in creatinine (p = 0.008). That is, patients who were transfused with a greater amount of erythrocyte concentrate did not have an optimal decrease in creatinine (Figure 6).
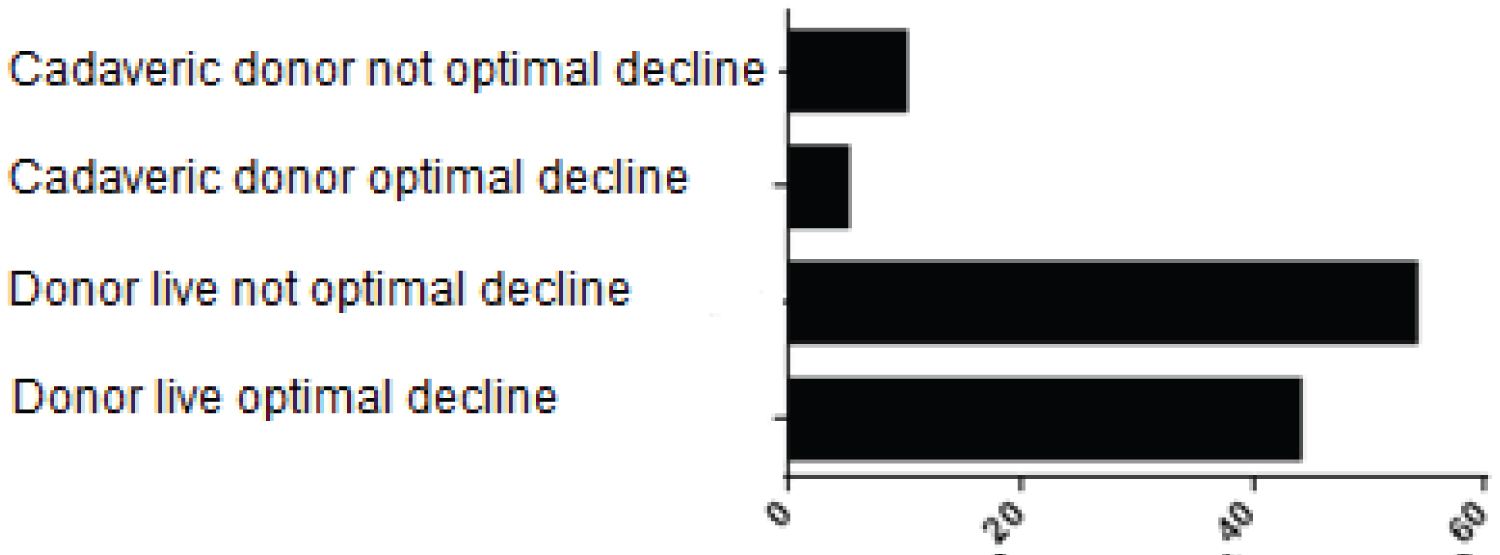 Figure 6: Patients who received blood transfusion during surgery, by study groups.
View Figure 6
Figure 6: Patients who received blood transfusion during surgery, by study groups.
View Figure 6
MAP during graft reperfusion had no significant difference (p = 0.1088) in the same way as CVP (p = 0.9) (Figure 7).
 Figure 7: Difference in hemodynamic variables in relation to creatinine levels (A) MAP; B) CVP.
View Figure 7
Figure 7: Difference in hemodynamic variables in relation to creatinine levels (A) MAP; B) CVP.
View Figure 7
Regarding the use of inotropics and vasopressors, the Chi-square test was applied and it was found that the groups that received inotropics had optimal creatinine decrease with confidence interval p = 0.0001, and in the non-optimal decrease of creatinine was observed, p = 0.0002, when inotropes were used in less quantity and time. Dopamine was the most used drug, then norepinephrine and dopamine (Figure 8).
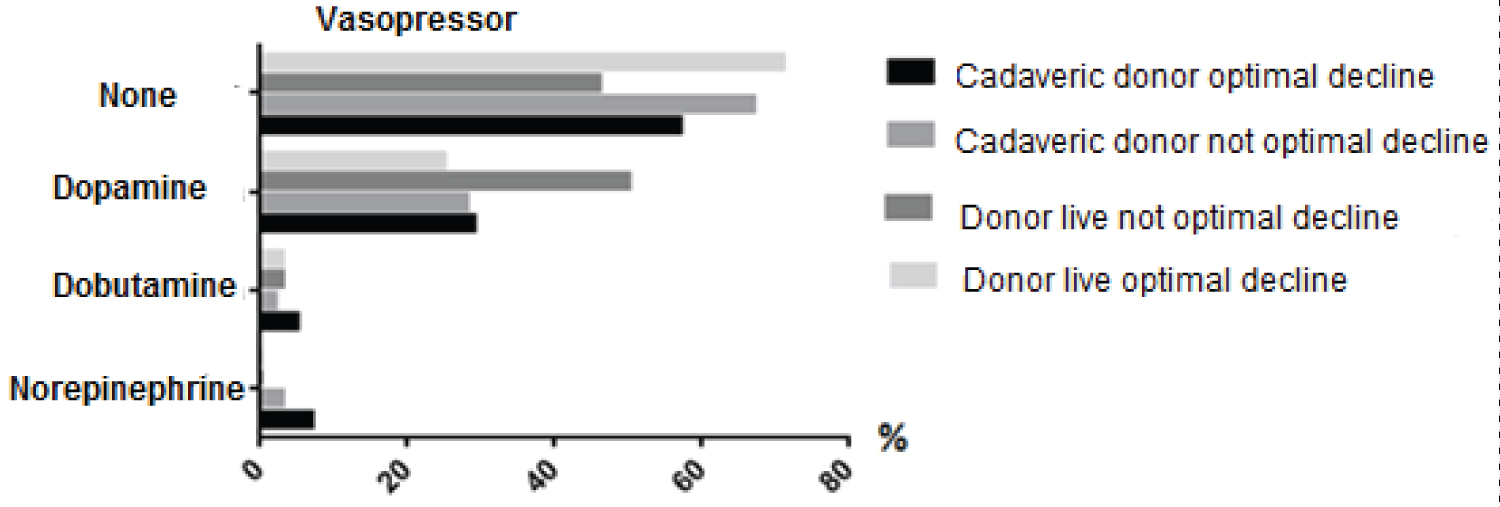 Figure 8: Main Vasopressors used during surgery in the different study groups.
View Figure 8
Figure 8: Main Vasopressors used during surgery in the different study groups.
View Figure 8
Regarding the use of diuretics, we found that 378 patients (96.93%) received diuretics, 76.2% used the combination of mannitol and furosemide. 17.1% received furosemide alone and 3.6% mannitol alone. The average dose per kilo of weight of furosemide that was administered was 1.95 mg and the average dose per kilogram of mannitol that was used was 0.4 mg (Figure 9).
 Figure 9: Ratio of the dose of furosemide mg/kg.
View Figure 9
Figure 9: Ratio of the dose of furosemide mg/kg.
View Figure 9
The administration of furosemide had a significant difference p = 0.0001 among the patients in the study who had or not an optimal decrease in creatinine, demonstrating that the use of this drug had an inversely proportional effect with the decrease in creatinine, that is, a higher dose, lower decrease in creatinine. On the other hand, the administration of mannitol had a positive and directly proportional influence on the decrease in creatinine since in the groups that received the highest dose of mannitol they had the best optimum decrease in creatinine, with statistical difference p = 0.0009 (Figure 10).
 Figure 10: Ratio of mannitol dose mg/kg.
View Figure 10
Figure 10: Ratio of mannitol dose mg/kg.
View Figure 10
Regarding spontaneous uresis, we obtained data from 291 patients, of which 221 (75%) presented spontaneous uresis in the operating room and 70 patients (25%) did not present it. Comparing the study groups, the patients receiving live donor had spontaneous uresis in 42.9%, while the cadaveric donor patients only 1.03% presented spontaneous uresis.
Kidney transplantation is a growing therapy of choice for patients with end-stage renal disease. However, several demographic factors could affect success after transplantation includes race, sex and age of the recipient [11]. In the same way, the race, gender and vital condition (cadaveric or living) of the donor could have a significant impact on the short and long term results [12]. Blood urea nitrogen (BUN) and serum creatinine are biomarkers that are typically measured over time for patients who undergo kidney transplants [13]. These biomarkers provide an assessment of how well the kidneys are after kidney transplantation. It is not uncommon for patients to experience kidney failure after a kidney transplant that is usually attributed to graft rejection. In this case, patients with graft failure return to dialysis. Anesthetic management is essential to allow an optimal functioning of the renal graft from the moment of reperfusion. Different anesthetic approaches have been proposed in the literature without reaching consensus on important aspects such as fluid administration, hemodynamic management (inotropic drugs or vasopressors), and the use of diuretics during surgery to improve perfusion of the newly grafted kidney.
With respect to the optimal decrease in creatinine, in our review, we found that the average fluid administration was 73.13 ml/kg, that is, fluid quantities greater than those recommended1 have been administered [1]. At a global level, reference has been made to the adverse effects of the use of 6% hydroxyethyl starch in critically ill patients with renal failure and the need to avoid its use (level of evidence 2B) [5]. In our study we found that there was a statistically significant difference p = 0.058 in the studied patients, who received higher doses per kilo of weight and who showed a non-optimal decrease in creatinine; Current evidence suggests that all hydroxyethyl starch products increase the risk of acute renal failure and the need for renal replacement therapy in all patient populations and a safe volume of any HES solution has yet to be determined [14]. In most clinical situations, these risks are likely to outweigh any benefits, and alternative volume replacement therapies should be used instead of the HES products.
However, no other studies have been performed in patients with renal receptors, but the results show that it is better not to administer it [6].
In the case of the transfusion of erythrocyte concentrates, the recommendation of the bibliography is to avoid it to the maximum [8] and it is necessary to mention that, in fact, all the patients who received the highest amount of erythrocyte concentrates were those who did not have an optimum decrease in creatinine, with p = 0.008. That is, there was an association with the non-optimal decrease in creatinine and blood transfusion.
In the world medical literature we do not find a consensus regarding the use of inotropic drugs or vasopressors in kidney transplantation; in 2010, Schmid, et al. discouraged the use of dopamine, based on two meta-analyzes that demonstrated a detrimental effect on renal function, in addition to being associated with greater mortality and longer hospitalization in patients who received it after surgery of kidney transplant [7]. However, in cases of refractory hypotension, it has been recommended to use low doses (3 to 5 mcg/kg/min) [8]. In contrast, it turns out that the vasopressor most used in patients transplanted from this hospital, is Dopamine and that there was also a statistically significant difference with respect to the optimal decrease in creatinine, between the groups that used inotropic drugs and those that did not use it, being a positive effect to have used said vasopressor p = 0.0002. As well as the chi-squared value with p = 0.0001, in which it was observed that the use of Dopamine was higher in the group of patients who had an optimal decrease in creatinine.
Hendrikus, et al. recommend that MAP be maintained above 90 mmHg during reperfusion of the renal graft to ensure filtration of the graft [1].
The MAP remained in the average values in the studied population without there being any significant difference since in all the patients, it remained on average during the reperfusion in values of 93 mmHg, that is, values recommended by the world literature. No statistical significance was found in the central venous pressure, but a mean value of 11 mmHg was observed with a standard deviation of 4 mmHg.
The use of diuretics is not mandatory, two randomized controlled studies showed that there is no benefit in using furosemide in patients with renal failure and oliguria, but more studies focused on renal recipient patients are needed [8]. In this study, we found that the use of furosemide had a negative effect on the optimal decrease in creatinine with statistical significance p = 0.0001 related to doses higher than 2 mg/kg; that is, the non-optimal decrease in creatinine was related to the use of doses above 2 mg/kg of furosemide.
Schmid, et al. also demonstrated that the administration of 200 to 250 ml of 20% mannitol immediately before reperfusion showed an increase in renal perfusion pressure [7], which translated into a lower occurrence of acute renal failure in kidney transplant surgery. This effect was clearly observed in our studies since it had a positive influence on the decrease in creatinine, since the patients who received the highest doses of mannitol had an optimum decrease in creatine with a statistical difference of p = 0.0009.
The anesthetic management of the renal recipient patient has not yet been standardized, there are several and diverse recommendations for the administration of fluids; For the time being, it is demonstrated that the use of hydroxyethyl starch results in a decrease in the optimal decrease in creatinine and that it could be associated with a reduction in graft survival, although studies are needed to demonstrate this.
The hemodynamic management of renal recipient patients should be individualized according to the current clinical status, polypharmacy, the results of previous echocardiography, and so on; taking into account the hemodynamic goals for reperfusion of the graft, and the use of vasopressor or inotropic drugs will depend on it. The management of intravascular volume is very important due to the alterations caused by peritoneal dialysis or hemodialysis and the time elapsed since the last time prior to transplantation; In addition, it is common that on admission to the operating room there are uncontrolled hypertension, with difficult handling of the intravascular volumes affected by dialysis; under this premise, the hemodynamic values must be managed under the control of adequate volume expansion.
With regard to the use of diuretics, mannitol, an osmotic diuretic, has been shown to increase the renal perfusion pressure, decrease secondary alterations to reperfusion and promote early graft function. However, furosemide could affect the early function of the graft when the dose exceeds 2 mg/kg, but there is still a lack of studies to prove it.