Bioactive peptides from collagen and gelatin with antioxidant and antihypertensive properties have become a topic of great interest for health and food preservation industries.
Thornback Ray Skin Gelatin Hydrolysate with an hydrolysis degree of 9.23% was prepared by treatment with Alcalase (TRGH-Alcalase). The hydrolysate was found to exhibit ACE inhibitory activity and 1, 1-Diphenyl-2-Picrylhydrazyl (DPPH) scavenging activity with an IC50 values of 0.92 and > 5 mg/mL, respectively. The molecular weight distribution of the hydrolysate showed the abundance of small-sized peptides with average molecular weight below 1000 Da (57%) contrary to the untreated gelatin. The hydrolysate and the gelatin did not show hemolytic activity at a concentration of 2 mg/ml.
TRGH-Alcalase was further fractionated by using size-exclusion chromatography. Fraction F3 showing the highest antioxidant and ACE inhibitory activity was further separated by Reversed-Phased High Performance Liquid Chromatography (RP-HPLC). Sub-fractions SF3 and SF9 had higher anti-ACE activity and SF5 had the highest antioxidant activity.
Taken together, the results suggest that the hydrolysate as well as the peptide fractions could offer potential uses as a functional food.
Alcalase gelatin hydrolysate, Gel filtration, Reverse-phase HPLC, Angiotensin converting enzyme, Antioxidant
Several fish processing by-products, that are usually eliminated as organic wastes, causing environmental problem, may contain high-quality functional compounds, such as collagen. The nutritional and medical benefits of these compounds suggest that they could be used, after processing, as ingredients in functional foods [1]. Recently, much attention has been focused on the identification and characterization of the structure, composition and, sequence of bioactive peptides. Biologically active peptides play an important role in metabolic regulation and modulation. These peptides can be used as functional food ingredients, or nutraceuticals and pharmaceuticals to improve human health and prevent disease. The importance of fish as a source of novel bioactive substances is growing rapidly [2].
Angiotensin Converting Enzyme (ACE) plays an important role in the regulation of blood pressure as well as fluid and salt balance in mammals. It is a dipeptidyl carboxypeptidase which converts the inactive decapeptide, angiotensin I, into a potent vasoconstrictor, the octapeptide angiotensin II. Moreover, ACE inactivates bradykinin, a vasodilatory peptide. Hence, ACE raises blood pressure [3]. Commercially available antihypertensive drugs such as captopril and enarapril are very potent ACE inhibitors. Recently, ACE inhibitory peptides derived from bovine collagen hydrolysate [4], rapeseed albumin hydrolysate [5], goby muscle hydrolysate [6], porcine skeletal muscle proteins [7] or Spanish dry-cured ham [8] were isolated. These peptides could be used as a potent functional food additive and represent a healthier and natural alternative to ACE inhibitor drugs.
Antioxidants are necessary to avert lipid oxidation in food, avoiding the formation of toxic compounds and unattractive odours and flavours. Furthermore, oxidative stress, caused by excessive free radicals from human-body metabolism, has also been related with the manifestation of numerous diseases, such as hypertension, cancer, diabetes, Alzheimer's and ageing [9]. Antioxidant peptides, a class of safe and widely distributed natural antioxidants, have been derived from different protein resources such as corn [10], cod [11] or soybean [12].
Fish skin gelatins have been reported to give rise to biologically active peptides with high ACE-inhibitory and antioxidant activities [1,13]. A number of commercial proteases have been used for the production of antioxidant and ACE inhibitory gelatin hydrolysates, including Protamex, Flavourzyme, Trypsin, Neutrase, Alcalase, Safinase and Esperase [13-15]. Alcalase, a commercial protease from Bacillus liqueniformis, has been shown to be very efficient in the hydrolysis of fish skin gelatin and the generation of ACE inhibitory [4,13,15-18] and antioxidant peptide hydrolysates [13,18].
Thornback ray skin is a processing by-product, which could be converted to value-added products, such as peptides, by bioconversion techniques. In a previous study, Thornback Ray Skin's Gelatin Hydrolysate obtained after treatment with Alcalase (TRGH-Alcalase), was characterized in terms of hydrophobicity, physico-chemical properties, and amino acid composition, as well as the comparative assessment of its antioxidant and ACE inhibitory capacities [19]. However, there is no information on the relationships between peptide physico-chemical characteristics and potential bioactive properties. The aim of the present work was to separate the TRGH-Alcalase by gel filtration and Reverse-Phase (RP) HPLC into fractions that differ in hydrophobicity followed by measurement of their antioxidant and ACE inhibition activities.
Common chemicals and solvents of analytical grade were obtained from different commercial sources. Alcalase® 2.4 L serine-protease from B. licheniformis was supplied by Novozymes® (Bagsvaerd, Denmark). Butylated Hydroxyanisole (BHA), captopril, 1,1-Diphenyl-2-Picrylhydrazyl (DPPH), Angiotensin-Converting Enzyme (ACE from rabbit lung) and Hippuryl-l-Histidyl-l-Leucine (HHL) were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Sephadex G-25 was purchased from Pharmacia (Uppsala, Sweden). Water was obtained from a Culligan system; the resistivity was approximately 18 MΩ. All other chemicals and reagents used were of analytical grade.
Fresh thornback rays (R. clavata) were obtained from the fish market of Sfax City, Tunisia. The samples were packed in polyethylene bags, placed in ice and transported to the laboratory within 30 min. Skins were collected and residual meat was removed manually. Cleaned skins were washed with tap water, cut with scissors into small pieces (0.5 × 0.5 cm) and placed in polyethylene bags. Skins were used immediately or stored at -80 ℃ until use less than three months.
The serine protease from B. licheniformis (Novozymes®) was used for the production of the hydrolysate. Protease activity was determined according to the method of Kembhavi, Kulkarni, and Pant [20] using casein as a substrate. One unit of protease activity was defined as the amount of enzyme required to liberate 1 μg of tyrosine per minute under the experimental conditions used.
Thornback Ray Gelatin (TRG) was extracted from the skin wastes following the method described by Lassoued, et al. [21]. Glycine-HCl buffer (100 mmol/l, pH 2.0) in combination with commercial pepsin (MP Biomedicals, France) were used in the extraction process.
Thornback Ray Gelatin Hydrolysate (TRGH) generated by Alcalase, was prepared according to Lassoued, et al. [19]. The degree of hydrolysis, defined as the percent ratio of the number of peptide bonds broken to the total number of peptide bonds per unit weight (htot), in each case, was calculated from the amount of base (NaOH) added to keep the pH constant during the hydrolysis [22].
The molecular weight distribution was determined by size-exclusion chromatography. The freeze-dried sample (1 g), was suspended in 5 ml of bidistilled water, then loaded onto a Sephadex G-25 gel filtration column (2.9 cm × 53 cm), pre-equilibrated and eluted with bidistilled water with a flow rate of 27 ml/h. The absorbance of fractions was measured at 280 nm. The column was calibrated with standard proteins, all standards were loaded separately: Albumin bovin serum fraction V (MW, 60000 Da, Biomatik corporation), human insulin (MW 5800 Da, Novo nordisk), vitamin-B12 (MW 1579 Da, SIPHAT), gluthation (307 Da, Sigma aldrich) and the Gly-Gly hydrochloride (168 Da, Sigma aldrich). The fitted linear equation between the log of the molecular weight (logMW) vs. the Kav (kav = Ve-V0/Vt-V0 where Ve: elution volume, V0: void volume and Vt: total volume) was calculated by the method of least squares, as log MW = -0.2278 Kav + 1.1162 (R2 = 0.9856).
The hemolytic activity was determined by inoculating the hydrolysate (2 mg/ml) on Mueller-Hinton agar supplemented with 5% (v/v) of horse blood (AES Laboratoires, France). The plates were incubated at 37 ℃ for 24 h. The development of a clear zone around the sample was considered as positive result.
The lyophilized hydrolysate (2.5 g), obtained by treatment with Alcalase, was suspended in 5 ml of Milli Q water, then separated onto a Sephadex G-25 gel filtration column (5 × 85 cm) pre-equilibrated and eluted with Milli Q water. Fractions (4 ml) were collected at a linear flow rate of 12 ml/h. Fractions (4 ml each) were collected and elution curves were obtained by measuring absorbance at 278 nm using an online spectrophotometer. Fractions were pooled, lyophilized and tested for their antiradicalar and anti-ACE activities.
The most active fraction was dissolved in Milli Q water, filtered through 0.22 μm filters, and then separated by RP-HPLC on a Waters C18 column (4.6 mm × 250 mm) (XBrideg™, Ireland). Sample was injected at a volume of 100 μl (10 mg/ml). Gradient elution was carried out with a mixture of solvant A (0.1%, Trifluoroacetic Acid (TFA), v/v) in deionized water and solvent B (0.1% TFA in acetonitrile, v/v). Sample was eluted with a linear gradient of solvent B (0-30%) for 60 min at a flow rate of 1 ml/min. On-line UV absorbance scans were performed between 200 and 300 nm at a rate of one spectrum per second with a resolution of 1.2 nm. Chromatographic analyses were completed with Millennium software. The antiradicalar and ACE inhibitory activities of the eluted peaks were determined. The liquid chromatographic system consisted of a Waters 600E automated gradient controller pump module, a Waters Wisp 717 automatic sampling device and a Waters 996 photodiode array detector. Spectral and chromatographic data were stored on a NEC image 466 computer. Millennium software was used to plot, acquire and analyse chromatographic data.
The DPPH free radical-scavenging activity was determined by the method of Kirby and Schmidt [23]. A volume of 500 µl of sample at different concentrations (1-5 mg/ml) was added to 375 µl of ethanol and 125 µl of DPPH solution (0.2 mM in ethanol) as free radical source. The mixtures were incubated for 1 h in the dark at room temperature. Scavenging capacity was measured spectrophotometrically (Labomed Inc, USA) by monitoring the decrease in absorbance at 517 nm. In its radical form, DPPH has an absorption band at 517 nm which disappears upon reduction by an antiradical compound.
In the presence of free radical scavengers, the violet-colored DPPH is reduced to yellow. Thus its absorbance decreases. DPPH radical-scavenging activity was calculated as:
Where A control, A blank and A sample are, respectively, the absorbance of the control reaction (containing all reagents except the sample), the sample without DPPH solution and the sample with DPPH solution. The test was carried out in triplicate and the results were mean values.
The ACE inhibitory activity was measured in triplicate as reported by Nakamura, et al. [24]. A sample solution (80 μl) was mixed with 200 μl of 5 mM HHL, and then preincubated for 3 min at 37 ℃. Samples and HHL were prepared in 100 mM borate buffer (pH 8.3), containing 300 mM NaCl. The reactions were then initiated by adding 20 μl of 0.1 U/ml ACE from rabbit lung prepared in the same buffer. After incubation for 30 min at 37 ℃, the enzyme reactions were stopped by the addition of 250 μl of 0.05 M HCl. The liberated Hippuric Acid (HA) was extracted with ethyl acetate (1.7 ml) and then evaporated at 90 ℃ for 10 min. The residue was dissolved in 1 ml of distilled water and the absorbance of the extract at 228 nm was determined using a UV-visible spectrophotometer.
The average value from three determinations at each concentration was used to calculate the ACE inhibition rate as follows:
Where A is the absorbance of HA generated in the presence of ACE inhibitor, B is the absorbance of HA generated without ACE inhibitors (100 mM borate buffer pH 8.3 was used instead of sample) and C is the absorbance of HA generated without ACE (corresponding to HHL autolysis in the course of enzymatic assay).
All experiments were carried out in triplicate, and average values with standard deviation errors are reported. Significant differences were analyzed using the SPSS software package (SPSS, Chicago, IL).
The enzymatic hydrolysis of proteins is an efficient method for producing peptides containing improved nutritional properties and bioactivities [25]. The nature of the bioactive peptides depends on the protein substrate, the specificity of the enzyme used in the proteolysis and the hydrolysis conditions, in particular the degree of hydrolysis [26]. In this study, thornback ray skin gelatin hydrolysate was prepared by treatment using Alcalase, a serine-protease from B. licheniformis. Alcalase has been widely used to produce bioactive peptides from varying fish skin gelatins [4,13,15-18].
The Degree of Hydrolysis (DH) of TRGH-Alcalase reached 9.23% after 240 min. This DH is lower than that described in squid gelatin (DH = 30%) [13] and similar to skin of sea bass [27].
Size-exclusion chromatography on Sephadex G-25 was performed to analyze the overall molecular weight distribution of the gelatin and the hydrolysate. The molecular weight distribution divided into four fractions is shown in Table 1. In the present study, the results showed that the gelatin, a high molecular weight polymer, contained more than 60% of peptides > 5000 Da. However, small-sized peptides with average molecular weight below 1000 Da are predominant in the hydrolysate (57%) which demonstrates the effectiveness of the Alcalase treatment on the gelatin.
Table1: Average Molecular Weight (AMW) distributions of gelatin (TRG) and Gelatin Hydrolysate by Alcalase (TRGH-Alcalase). View Table 1
The hemolytic activity of gelatin and hydrolysate was tested at a concentration of 2 mg/ml. No hemolysis was observed. These results confirmed that these peptides would be non-toxic. Previous studies reported also that no haemolytic activity was observed in peptides derived from RuBisCO purified from green juice alfalfa [28] or low haemolytic activity of peptides derived from palm kernel cake [29].
The ACE inhibitory activity of TRGH-Alcalase at different concentrations was investigated and reported in Figure 1A. As expected, the activity was concentration dependent. The captopril, a synthetic active inhibitor, showed the highest activities with an IC50 value of 0.02 µg/ml. The gelatin activity was very low comparing to that of the hydrolysate. In fact, the IC50 value of gelatin is 6.99 mg/ml against 0.92 mg/ml for TRGH-Alcalase. The activity increased 7.5-fold, suggesting that ACE inhibitory peptides are encrypted within the sequence of the parent protein. High ACE inhibitory activity of TRGH-Alcalase was attributed to the high level of hydrophobic peptides [19]. Byun, et al. [15] reported also the ACE inhibitory activity of Alaska pollack skin digested by Alcalase; the IC50 value is 0.629 mg/ml. The salmon skin collagen hydrolyzed with Alcalase and papain, and treated by multistage separation showed a lower activity than TRGH-Alcalase with an IC50 value of 1.165 mg/ml [30].
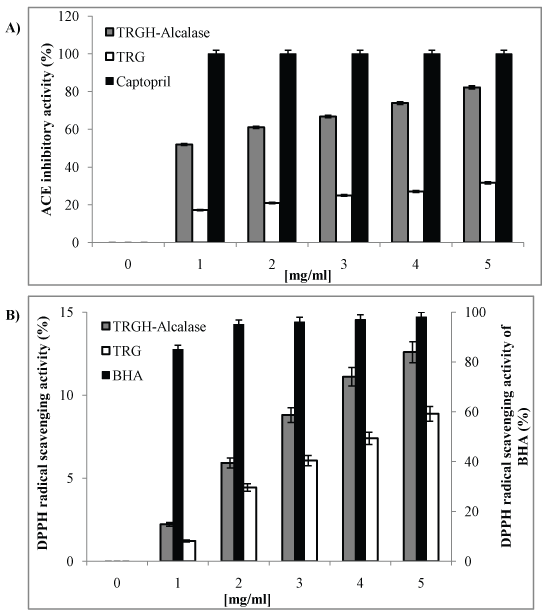 Figure 1A&B: A) Angiotensin-I Converting Enzyme (ACE) inhibitory activity; B) DPPH scavenging activity of Thornback Ray Gelatin (TRG) and Gelatin Hydrolysate (TRGH-Alcalase) at different concentrations. Captopril and BHA were used as standards, respectively. View Figure 1A&B
Figure 1A&B: A) Angiotensin-I Converting Enzyme (ACE) inhibitory activity; B) DPPH scavenging activity of Thornback Ray Gelatin (TRG) and Gelatin Hydrolysate (TRGH-Alcalase) at different concentrations. Captopril and BHA were used as standards, respectively. View Figure 1A&B
Figure 1B shows DPPH radical inhibitions of BHA, gelatin and TRGH-Alcalase at various concentrations. All of samples showed dose-dependent activity, and BHA showed the highest activity. Gelatin showed a lower activity comparing to TRGH-Alcalase. However, the IC50 value of TRGH-Alcalase is higher than 5 mg/ml. This low activity was attributed to the low content of amino acids that enhance the scavenging activities of peptides (Trp, Tyr and Met) [19]. Where does the interest of hydrolysate fractionation in order to get a concentrate of antioxidant peptides. Jridi, et al. [31] reported also the DPPH scavenging activities of cuttlefish skin gelatin hydrolysates which the Alcalase hydrolysate gives the best activity with 71% at 5 mg/ml. This high activity can be reported to the low molecular weight of generated peptides. In fact, the DH of cuttlefish skin gelatin Alcalase hydrolysate is 20.3%.
TRGH contained a mixture of active and inactive peptides with different amino acids sequences and various sizes. Therefore, for the separation and enrichment of antioxidant and ACE inhibitory peptides, TRGH-Alcalase was fractionated on a Sephadex G-25 gel filtration chromatography. As reported in Figure 2A, the chromatogram was separated into five fractions (F1 to F5). Each fraction was collected, freeze-dried and then evaluated for antioxidant and ACE-inhibitory activity at a concentration of 1 mg/ml (Figure 2B). Those fractions exhibited varying DPPH scavenging activity (from 12 to 38%). However, only F3 exhibited anti-ACE activity (87%) at 1 mg/ml. This high ACE inhibitory activity might presumably relate with the ability to chelate metal in zinc ligand of ACE [32]. F3 exhibited also the highest antioxidant activity which plausibly due to the ability in donating hydrogen of peptides [33].
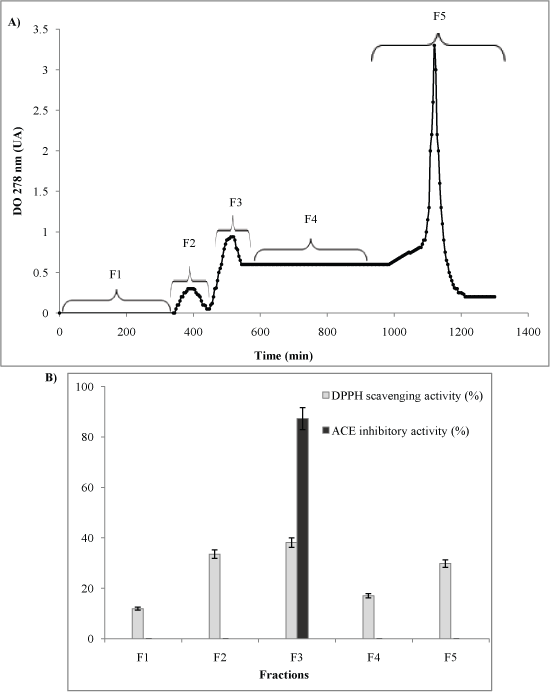 Figure 2A&B: A) Elution profile of Thornback Ray Gelatin Hydrolysate treated with Alcalase (TRGH-Alcalase) obtained after size exclusion chromatography on Sephadex G-25; B) Antioxidant and ACE inhibitory activities of fractions. View Figure 2A&B
Figure 2A&B: A) Elution profile of Thornback Ray Gelatin Hydrolysate treated with Alcalase (TRGH-Alcalase) obtained after size exclusion chromatography on Sephadex G-25; B) Antioxidant and ACE inhibitory activities of fractions. View Figure 2A&B
Thus, fraction F3 obtained from size-exclusion chromatography that displayed the highest activities was further separated by RP-HPLC on a C18 column using a linear gradient of acetonitrile. Fractions of 5 min (from SF1 to SF12) were automatically collected, and antioxidant and ACE-inhibitory activities were tested. The RP-HPLC profile reported in Figure 3A revealed a very large number of peaks relating to the abundance of peptides generated. Five major peptide sub-fractions (named SF3, SF4, SF9 and SF10) showed high ACE inhibitory activities varying from 60 to 88% at 1 mg/ml (Figure 3B). With increasing acetonitrile concentration, higher hydrophobic residues were eluted; the highest ACE inhibitory activity of SF9 and SF10 might be due to its high hydrophobicity Jung, et al. [34]. Similar result was reported by Intarasirisawat, et al. [35] when studying the ACE inhibitory activity of RP-HPLC peptide fractions of defatted skipjack roe hydrolysed by Alcalase.
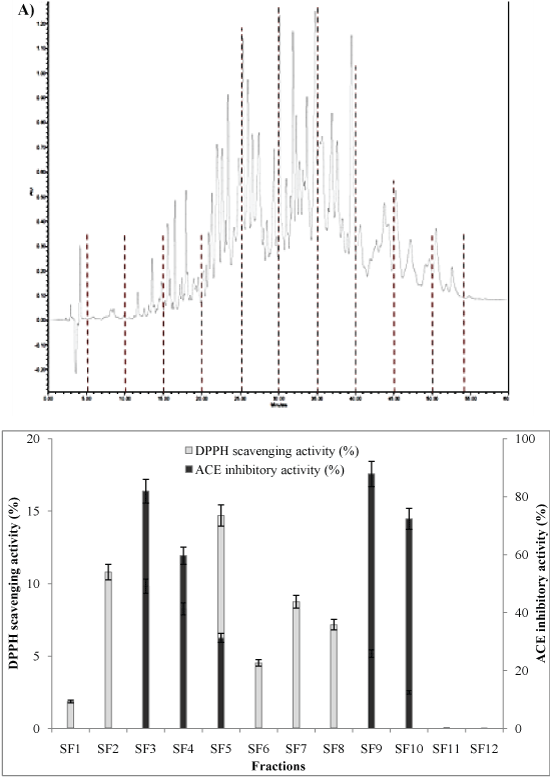 Figure 3A&B: A) Reversed-phase chromatographic separation of the selected pooled fraction (F3) obtained from size-exclusion chromatography; B) Fractions were automatically collected and assayed for their ACE inhibitory activity and DPPH radical scavenging activity. View Figure 3A&B
Figure 3A&B: A) Reversed-phase chromatographic separation of the selected pooled fraction (F3) obtained from size-exclusion chromatography; B) Fractions were automatically collected and assayed for their ACE inhibitory activity and DPPH radical scavenging activity. View Figure 3A&B
Regarding antioxidant activity, DPPH radical-scavenging activities of all fractions eluted from RP-HPLC are reported in Figure 3B. SF5 found to exhibit the highest activity with 15% at 20 µg/ml only, therefore, fractionation into peptides led to the improvement in DPPH radical scavenging activities as evident for SF5 peptides as well as SF2 peptides (Figure 3B). The results indicate also that the fractions that eluted late (SF9 to SF12) exhibited weak activity. Increased hydrophobic character of peptides derived from protein sources has been shown to correlate with higher DPPH other radical scavenging activities [36,37] when compared with peptide fractions of lower hydrophobic content. However, in the present work, there was no strong relationship between hydrophobic character of peptide fractions and DPPH scavenging activity. Similar results were found by Ajibola, et al. [38] when studying the DPPH scavenging activity of RP-HPLC fractions from African yam bean seed proteins treated with Alcalase. Hence, high DPPH scavenging activity exhibited by SF2 and SF5 could be attributed to the presence of specific amino acids and their positioning in the sequence rather than the overall hydrophobic character.
Results of this study showed that the thornback ray gelatin hydrolysate treated with Alcalase and their peptide fractions possess moderate antiradicalar activity against DPPH radicals. The antiradicalar activity exhibited by peptide sub-fractions was mostly independent of their hydrophobic character, suggesting that other peptide structural properties (peptide size, amino acid sequence…) are involved. The peptide sub-fractions showed also potential inhibition of ACE activity. Hydrophobic character of the peptides was not positively related to ACE inhibitory property. Overall, the fractionation of TRGH-Alcalase by gel filtration led to the production of some peptide fraction with better antiradicalar activity; and the fractionation by gel filtration followed by RP-HPLC releases peptide fractions with better ACE inhibitory activity. Thus, some of these peptide fractions may contribute to improve cardiovascular health or could be used as ingredient of functional foods with potential antihypertensive and antioxidant capacities. Further studies are required to test efficacy of these peptide fractions using model food systems or animal disease models.
This work was funded by the Ministry of Higher Education and Scientific Research, Tunisia.