In asthmatic patients, neutrophils are known to be hyperactive, characterized by a production of numerous inflammatory mediators. The present study aimed to evaluate the effect of total flavonoid extract (TFE) of Pseudocedrela kotschyi (P. kotschyi) on neutrophil activities and anaphylactic reactions.
Extract was tested on neutrophils isolated from blood of asthmatic patients activated by N-formyl-L-methionyl-L-leucyl-phenylalanine (fMLP) or histamine, and on anaphylactic reaction induced by horse serum and Bacillus Calmette-Guérin (BCG) vaccine intraperitoneal in albino Wistar rats. In vitro, fMLP-induced production of peroxidase activity, superoxide anion (O2-), tumor necrosis factor alpha (TNF-α) and leukotriene B4 (LTB4), and histamine-induced production of nitric oxide (NO) and hydrogen peroxide were evaluated, while blood total and differential leukocytes, and level of NF-α and LTB4 in bronchoalveolar lavage fluid were measured in rat.
Treatment with TFE of P. kotschyi exhibited significant reduction in the peroxidase activity (IC50, 76.03 µg/ml), O2- (IC50, 84.89 µg/ml), TNF-α (IC50, 54.07 µg/ml) and LTB4 (IC50, 48.97 µg/ml) production by fMLP-stimulated neutrophils. Moreover, TFE displayed markedly reduction in NO (IC50,37.31 µg/ml) and H2O2 (IC50, 32.05 µg/ml) production by the histamine-activated neutrophils. In addition, the treatment of rat with TFE brought to normalcy, the increased total white blood cells, neutrophil and eosinophils in blood, TNF-α and LTB4 in bronchoalveolar lavage fluid.
Results of study indicated that stem bark lavonoids of P. kotschyi could be effective in the treatment of anaphylaxis and asthma.
Asthma, Anaphylaxis, Flavonoids, Pseudocedrela kotschyi
Asthma is one of the most common chronic diseases worldwide affecting approximately 300 million individuals [1]. Asthma or anaphylaxis are allergic diseases essentially characterized by eosinophilia and mast cells release histamine which is responsible for the production of IgE. Nevertheless, neutrophils are highly implicated and it is demonstrated in about 50% of asthma cases [2]. Consecutive to bronchial allergen challenge for example, there is an influx of neutrophils in proximal and distal airways. In asthmatic patients, neutrophils are known to be hyperactive, characterized by a production of numerous inflammatory mediators including leukotriene, platelet activating factor, tumor necrosis factor (TNF)-α [3].
Because of low adequacy or related unfavourable events, the treatment choices for various respiratory tract hypersensitive disorders have significant impediments [3]. Nowadays, drug treatment of asthma is mainly based on the administration of anti-inflammatory medications preventing symptoms and relief medication during exacerbations [4]. There is also a particularly high frequency of use of herbal plants, even among adults with access to specific treatment for asthma. In the management of asthma and allergy, traditional Cameroonian system of medicine is represented by a few medications from the traditional indigenous plants among which Pseudocedrela kotschyi.
The medicinal plant Pseudocedrela kotschyi (Schweinf) Harms (Miliaceae) has numerous uses in traditional medicine. Root or root bark preparations are administered as powerful diuretic, to treat asthma, fever, dysentery, oedema, to facilitate childbirth, and are applied externally to ulcers, mastitis, haemorrhoids, fractures, rheumatism, caries and gingivitis, and as an aphrodisiac etc [5]. The plant also showed an important immunomodulatory activities with respect to neutrophil activities [6]. Despite the traditional use in allergy treatment, no study has been done with respect to the bioactive compounds.
The present study is considered to be the first to investigate as potential anti-asthmatic and anti-anaphylaxis drugs the bioactive compounds of P. kotschyi. Hence, this study was conducted to evaluate the inhibitory effect of total flavonoid extract of stem barks of P. kotschyi on activity of neutrophil in asthmatic diseases and anaphylaxis as possible phytotherapy controlling asthma.
Plant samples were collected from outfield of Garoua city, Head quarter of the North region, Cameroon. The plant was identified as Pseudocedrela kotschyi (Schweinf) Harms (Miliaceae) by comparison with the plant sample No: 7009/SRF/Cam at the National Herbaruim, Yaounde, Cameroon.
Stem barks of the plant were collected and washed thoroughly in tap water, shade dried and pulverized. TFE was obtained as described by Lavanya, et al. [7]. In summary, 20 g of the powder of the plant material were extracted with 200 ml of petroleum ether (40-60 ℃) in a shaker for 24 hours. The solid residue obtained was then extracted with 155 ml of ethyl acetate for 24 hours and filtered. The resulting filtrate was concentrated using flash evaporator for complete solvent removal. The freeze dried material was extracted with boiling acetone and the residue was concentrated at atmospheric pressure. This concentrated residue was extracted successively with light petroleum ether (40-60 ℃) and benzene to remove non flavonoid and other matter.
The reaction mixture consists of 1 ml of plant extracts, 0.6 ml of sodium nitrite (5% w/v), 0.5 ml of aluminum chloride (10% w/v), 3 ml of sodium hydroxide (4.3% w/v), and distilled water is added to make the volume to 10 ml. The reaction mixture was allowed to stand for 15 minutes before reading the absorbance. Absorbance was measured at 500 nm in a UV-Vis spectrophotometer. Quercetin was used as a standard and results were calculated as quercetin equivalents (Quercetin eq., mg/ml) of P. kotschyi. Total flavonoid content of P. kotschyi (mg/g stem bark) was found to be equal to 1.67.
Total flavonoid extract was tested at a range of concentrations from 128 µg/ml to 8 µg/ml. These concentrations was selected based on our previous study which showed no adverse effects when the extract at the concentration less than 512 mg.ml-1.
Blood sample was collected sample from adult atopic patients with bronchial asthma admitted in Regional Hospital of Bamenda. The procedure of blood collection has been read and approved by the ethical committee board of the faculty of health science. The research has received the okay of the Regional Hospital of Bamenda where the patients were recruited and collected by the trained personnel of the hospital. The patients have read and signed the informed consent. These patients received no specific hyposensitization (ie,.No immunotherapy) and are not taken corticosteroids or other drugs in the previous week.
Heparinized venous blood (5 to 10 ml) obtained from patients was mixed with an equal volume of dextran 70 and an additional half volume of phosphate buffer saline (PBS), and allowed to stand at room temperature for 1 hour in order to permit the erythrocytes to sediment. Neutrophils were purified following the methods of Böyum [8] as reported by Oumar, et al. [9]. The leukocyte-rich supernatant (buffy coat) was washed with RMPI-1640 (Sigma, Germany), and then centrifuged on a Ficoll-Paque density gradient at 1800 tr/min for 30 min at room temperature. After the granulocyte-rich erythrocyte layer at the bottom was washed with RMPI-1640, the residual erythrocytes were lysed by the addition of cold 0.2% NaCl. The mixture was then vortexed for 30 s followed immediately by the addition of an equal volume of cold 1.6% NaCl. The residual leukocytes were washed twice, unless otherwise stated, re-suspended in RPMI 1640 containing 5% FCS (Fetal calf serum), and immediately placed at 4 ℃. The suspension was found to contain approximately 93% neutrophils whom the viability was evaluated using the trypan blue [10].
Healthy adult male Wistar albino rats (150 -200 g), were raised in standard polypropylene cages with all standard laboratory conditions in the animal house of the laboratory of Biological Sciences. The rats were fed a standard rat pellet diet and had free access to water ad libitum. The protocol was read and received the okay the scientific committee of Department of Biological Sciences (Faculty of Science, University of Bamenda).
fLMP-activation of neutrophils: Following isolation, neutrophils were cultured in triplicate with RMPI-1640 medium in 96 microplate (NUNC) at 37 ℃, 5% CO2. Cells were cultured at 1 × 106/ml and stimulated with fMLP (10 mM.ml-1) in presence of the extract. After incubation for 45 min at 37 ℃, 50 µl of supernatants were collected to assess the production of myeloperoxidase.
To measure the superoxide production, ferricytochrome-c (60 µM) and sodium azide (2 mM) was added to the culture and incubated for 10 min at 37 ℃. Later 50 µl of the supernatant was collected to assess O2- production. Negative control cells received PBS only, while positive control were activated and not treated with extract.
Measurement of neutrophil peroxidase release: Enzymatic activity of myeloperoxidase (MPO) was measured in a colorimetric assay [9] where 100 ml of substrate buffer (50 ml of citrate-phosphate buffer, pH 5; 20 ml of 30% H2O2; 20 mg of orthophenylenediamine) was added to 20 ml of supernatant of PMNs culture. The reaction was stopped with H2SO4 and the absorbance measured with a microplate reader at 490 nm. The optical density were used to determine the percentage inhibition as follows:
% Inhibition = 100 × [(ODcontrol - ODsample)/ODcontrol]
Measurement of superoxide anion production: The production of O2- by neutrophils was measured with the ferricytochrome-c reduction assay [11]. The optical density of the collected supernatants were at 550 nm using ELISA reader. The optical density were used to determine the percentage inhibition as follows:
% Inhibition = 100 × [(ODcontrol - ODsample)/ODcontrol]
Evaluation of tumor necrosis alpha and leukotriene B4 production: Neutrophils were cultured at 1 × 106 in RMPI 1640 and treated with TFE and fMLP (10 mM.ml-1) at 37 ℃, 5% CO2. After 24 h, 50 µl aliquot of supernatant was transferred to a 96-well plate for either TNF-α or LTB4 determination by enzyme immunoassay (ELISA) as described by the manufacturer of the kit (Sigma). Each sample was assayed in triplicate. The percentage inhibition was then calculated as follows:
Histamine-activation of neutrophils: Neutrophils were cultured in 96-well plate at 1 × 105 cells.ml-1 for 2 hours in RMPI 1640, with histamine (16 mM.l-1). Later, TFE at different concentrations (8-128 µg.ml-1) was added (untreated was considered as a positive control, free histamine culture as negative control) for a final volume per well of 200 µl. Next, the plates were incubated for 16-20 hrs. Thereafter, the supernatant was collected to determine nitrite (NO2-) as NO production and hydrogen peroxide.
Assessment of NO production: The amount of NO2−, the end product NO, released in the culture medium was estimated to appreciate the NO production. Briefly, 50 µl of culture supernatants were mixed with an equal volume of Griess reagent (1% sulfanilamide, 0.1% naphthylethylenediamine in 5% phosphoric acid) in a 96-well flat-bottom microplate and incubated at room temperature for 10 min [9,12]. The resulting colour development was measured at 550 nm with a microplate reader. The absorbance values were compared to a standard sodium nitrite curve and the absorbance values were converted to corresponding nitrite concentrations (µM). The percentage of NO inhibition were calculated as follows:
Measurement of hydrogen peroxide production: Hydrogen peroxide (H2O2) production was analyzed using the colorimetric method [12]. To 200 µl of culture supernatant, 50 µl 10 mM FeSO4•7H2O (dissolved in 1 mMHCl) and 25 µl 2.5 mMKSCN (dissolved in PBS) were added and the optical densities were measured in a microplate reader at 490 nm wavelength. Additionally calibration curve were assessed. Optical densities of RPMI 1640 containing various H2O2-concentrations (0-200 µM f.c.) and 20 µl TCA (50%) were measured and used for standard curve for determination of the H2O2 release in tested well. The percentages of H2O2 inhibition were calculated as follows:
Induction and treatment of anaphylaxis in rat: Twenty five rats were divided into 5 groups: Negative control received normal diet and water only and served as normal control (Group I); Sensitized control received 1 ml/kg PBS p.o.(Group II); Positive control was administered standard dexamethasone (10 mg/kgp.o.) (Group III); Test groups were given 50 mg/kg and 100 mg/kg TFE(p.o.) (Group IV and V respectively). Extract and dexamethasone were given in PBS through oral gavages for 14 days. Before treatment, mice were sensitized by sub-cutaneous injection of 0.5 ml of horse serum followed by 0.5 ml of bacillus calmette-guérin (BCG) vaccine containing 3 × 1010 organisms per ml in all groups except normal control group. At the end of treatment, 0.25 ml of horse serum in saline was challenged via intravenous injection in tail vein.
Collection of blood and bronchoalveolar lavage fluid: Thereafter, the blood was collected from retro-orbital plexus. Part of the blood was used to prepared serum and other part was used for analysis for total leukocyte, eosinophil count, and % polymorphs count using an automatic blood cell counter (DxH800; Beckman Coulter, USA). A tracheal cannula was inserted via mid cervical incision and lavage four times with 1 ml of cold PBS, pH = 7.4. The bronchoalveolar lavage fluid was centrifuged for 10 min and the supernatant was analyses for TNF-α and LTB4 according to the manufacturer's instructions and guidelines using ELISA kits (eBioscience, USA).
A statistic software Graph Pad Prism version 6 was used to evaluate the statistical difference between different groups. The values are expressed as the Mean ± SD of four independent experiments using four different patients released in triplicate. The statistical significance was performed using One-Way ANOVA followed by Student's t-test. A P-value < 0.05 was considered to be statistically significant.
In vitro culture, the study indicated that fMLP increased the peroxidase activity and superoxide anion generation in neutrophils for about 13.52 ± 3.71 and 27.26 ± 5.26 percent respectively. TFE therefore scarcely inhibited this neutrophils peroxidase release as well as the superoxide production (Table 1).
Table 1: Concentration-dependent inhibition percent of superoxide anion generation and neutrophil peroxidase (NPO) release of asthmatic patient neutrophil by total flavonoid extract of P. kotschyi. View Table 1
Data are mean of % inhibition values ± SD of four independent experiments of three replicates in each. P value denotes the significance difference as compared with the effect of fMLP (considered as zero).
Results indicated that treatment of asthmatic patient neutrophils with Fmlp resulted in high increase of the production of TNF-α and LTB4 compared to unstimulated cells with an augmentation percent of 77.43 ± 12.81 and 86.33 ± 6.30 respectively. TFE of P. kotschyi were therefore found to inhibit this production of TNF-α and LTB4 in a dose-dependent manner (Table 2).
Table 2: Effect of total flavonoids extract of P. kotschyi on production of TNF-α and LTB4 by asthmatic patient neutrophil. View Table 2
Data are mean of % inhibition values ± SD of four independent experiments of three replicates in each. P value denotes the significance difference as compared with the effect of fMLP (considered as zero).
Neutrophils of patients with asthma were found to be very sensitive to histamine lead to an increase introduction of NO and H2O2, with an increase percent of 40 ± 6.55 and 58.61 ± 7.56 compared to untreated neutrophils. When exposed to TFE of P. kotschyi, this production of NO and H2O2 in response of histamine decreased compared to the counterpart control. TFE of P. kotschyi inhibited this production of NO and H2O2 in a concentration-dependent manner with IC50 = 37.31 ± 4.11 and 32.05 ± 7.62 (Table 3).
Table 3: Inhibitory effect of total flavonoid extract of P. kotschyi on histamine-induced nitric oxide and hydrogen peroxide production by asthmatic patient neutrophil. View Table 3
Data are mean of % inhibition values ± SD of four independent experiments of three replicates in each. P value denotes the significance difference as compared with the effect of histamine (considered as zero).
The number of total and differential leukocyte in blood was evaluated in untreated-rats, horse serum-sensitized rats (sensitized control) and sensitized rats treated with dexamethasone (positive control), 50 mg/kg TFE and 100 mg/kg at 5 h after challenging with horse serum followed by BCG vaccine (Table 4). In the absence of treatment, results demonstrated that the total white blood cell number of total leukocytes and the percentage of eosinophil, neutrophil and monocytes increased in horse serum sensitized rats, while that of lymphocytes decreased when compared to untreated animal (negative control). Treatment with dexamethasone, the reference drug (10 mg/kg), resulted in significant decrease of the number of total and differential leukocyte. Similarly, TFE of P. kotchyi (50 mg/kg and 100 mg/kg), instigated significant decrease in the number of total and differential leukocyte in blood (excluding monocytes) in horse serum sensitized rats compared to sensitized control.
Table 4: Effect of total flavonoids extract of P. kotschyi on total blood leukocyte and differential leukocytes blood count in horse serum-induced anaphylaxis in rats. View Table 4
TWBC, Total white blood cells. Positive control, animal sensitized and received dexamethasone. Negative control, untreated animal. Sensitized control, animal challenged with horse serum. Results are Mean ± SD. (n = 5). Asterisks denoted the significance difference between drug/extract treated rats and sensitized control (p < 0.05).
Results indicated that the increase of TNF-α and LTB4 level in the bronchoalveolar lavage fluid that was observed in horse serum challenged rats was suppressed by treatment with 50 mg/kg and 100 mg/kg TFE compared untreated sensitized rats. The inhibitory efficacy of TFE treatment was concentration-dependent, and that of 100 mg/kg TFE was almost comparable to that of standard drug, dexamethasone 10 mg/kg b.w (Figure 1).
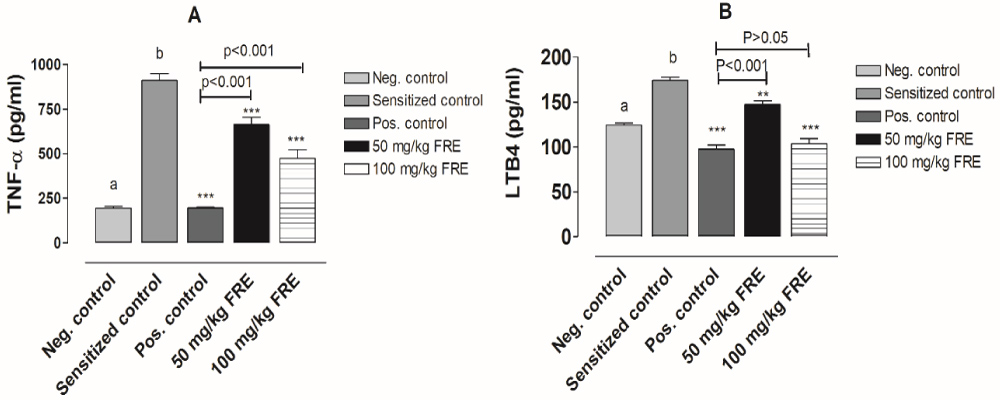 Figure 1: Effect of treatment with total flavonoid extract of P. kotschyi on TNF-α and LTB4 concentration of bronchoalveolar lavage fluid of horse serum-induced anaphylaxis in rats. Results are mean values ± SD (n = 5). Neg. control is untreated animal group. Sensitized control is untreated sensitized animal group. Pos. control is dexamethasone-treated sensitized animal group. a,b) P < 0.05 comparison between negative control and sensitized control. **P < 0.01, ***P < 0.05 compared with sensitized control.
View Figure 1
Figure 1: Effect of treatment with total flavonoid extract of P. kotschyi on TNF-α and LTB4 concentration of bronchoalveolar lavage fluid of horse serum-induced anaphylaxis in rats. Results are mean values ± SD (n = 5). Neg. control is untreated animal group. Sensitized control is untreated sensitized animal group. Pos. control is dexamethasone-treated sensitized animal group. a,b) P < 0.05 comparison between negative control and sensitized control. **P < 0.01, ***P < 0.05 compared with sensitized control.
View Figure 1
The immune system is highly implicated in bronchial asthma, characterized by complex reactions between inflammatory cells and the release of granule proteins [13] due to the hyperactive neutrophils implicated. Numerous bioactivities described for anti-asthmatic drugs are also known for flavonoids. Phytochemical study of P. kotschyi showed the presence of flavonoids which may be effective as antiasthmatic or antiallergic drugs. Here we examined the flavonoid extract of this plant on neutrophil activities.
Neutrophils are major source of O2-, H2O2, and HOCl, which result in the severity of tissue damage in allergic diseases [14,15]. They have been described to produce more superoxide in individuals with atopic diseases than those did not [15]. Our results demonstrated that TFE of P. kotschyi displayed a significant reduction in production of O2- by neutrophils isolated from patients with asthma. This might demonstrate the effectiveness of the flavonoids of P. kotschyi in treatment of asthma. The incorporation of the 2 cytoplasmic components of the NADPH oxidase, p47 and p67, into the cell membrane to form the active NADPH oxidase represent one of the major factor leading to the generation O2- in neutrophils. TFE of P. kotschyi might be act by blocking the incorporation p47 and p67 to inhibit the production of O2-. In addition, treatment of neutrophil of asthmatic patients with TFE of P. kotschyi decreased the neutrophil peroxidase release. TFE might block the synthesis or activities of myeloperoxidase in neutrophils, and this might also justify its use in asthma treatment as peroxidase activity of neutrophils is known to be high in individual with asthma as result of degranulation of primary granules [16].
TNF-α is an important cytokine playing a key role in immediate host defense before activation of the adaptive immune system. It also contribute in inflammatory response as demonstrated by the high TNF-α levels seen in allergic asthma [17]. In addition, TNF-α has been described to mediate the first phase of late airway hyper responsiveness [18] inducing the release of histamine from mast cells [19]. Here, the total flavonoid extract of stem barks of P. kotschyi exhibited effective inhibition of TNF-α release by fMLP-stimulated neutrophils of asthmatic patients. This demonstrated that TFE could be effective in treatment of asthma. Moreover, leukotriene B4 (LTB4), is also a potent proinflammatory mediator inducing inflammatory cell chemotaxis, adherence, and stimulation in the pathophysiology of asthma were decreased by TFE in neutrophils. LTB4 is instrumental in recruiting neutrophils to the lung in IgE-mediated reactions [20] which partially mediates the acute and chronic responses to antigen [21], therefore its inhibition by TFE of P. kotschyi is also an evidence of effectiveness in asthma.
In allergic reactions, histamine is highly produced by mast cell in response to various substances such as TNF-α. In various ways including production of NO and H2O2, the produced histamine contribute to the aggravation of asthma. Results of this study indicated that the treatment of neutrophils with TFE of P. kotschyi caused a dose-dependently inhibition of the production of nitric oxide and hydrogen superoxide induced by histamine demonstrating once more the possible efficacy of TFE of P. kotschyi in treatment of allergic reactions. Production of abundant amounts of NO in response of histamine stimulation was described to be due to high expression of iNOS [22], therefore, the TFE of P. kotschyi might be inhibited the expression of this enzyme.
In the in vivo investigation, as is known that the euglobulin fraction of horse serum is known to be responsible of initiation of the allergic response resulting in anaphylactic shock [23]. The consequence of this anaphylactic shock is manifested by increase in total leukocytes volume with increase in percentage of neutrophil, eosinophil and monocytes, but decrease in that of lymphocyte. Results of the present study indicated that the TFE of P. kotschyi exhibited a significant reduction in neutrophil and eosinophil, and significant increase in lymphocyte. This was after injection of horse serum suggesting that TFE of P. kotschyi may correct the anaphylaxis, during allergic reactions. Additionally, treatment with TFE of P. kotschyi appeared to have a significant inhibitory effect on level of TNF-α and LTB4 levels in bronchoalveolar lavage fluid of rats treated with horse serum. These results therefore demonstrated in in vivo study the beneficial effect of TFE in treatment of allergic reactions. Decrease in leukotriene level might be due to the low level of neutrophils and eosinophils in the blood as observed in white blood cells count [24,25].
Results of this study indicated that flavonoids of P. kotschyi are potent natural products to treat asthma, and they warrant further testing of this compound in preclinical and clinical models of asthma.
We thankful to the Director of IRAD, Bambui-Cameroon for providing necessary facilities to carry out this research work. Authors thanks Miss. Hakoueu Flora for his helpful contribution for the realization of this study.
Authors declare that there has been no financial support for this work that could have influenced its outcome.
Authors declare that there are no conflicts of interest associated with this publication.
All participants gave their written consent.
OM and GKO made substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data. OM drafted the manuscript and revised it critically for important intellectual content. TC and KA gave final approval of the version to be published. Each author agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.